Scaling High-Purity Troxerutin Production: A Novel Catalytic Approach for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic viability, particularly for cardiovascular agents like Troxerutin. Patent CN106892950B, published in March 2020, introduces a transformative preparation method for high-content Troxerutin that addresses longstanding challenges in selectivity and impurity control. This technical disclosure outlines a sophisticated three-stage process that leverages natural macromolecular sodium alginate as a catalytic mediator, diverging significantly from conventional strong alkali methodologies. By integrating a rigorous pre-purification step for the raw material rutin, achieving purity levels exceeding 99.0%, the subsequent etherification reaction is optimized to favor the formation of 7,3′,4′-trihydroxyethylrutin. This innovation is pivotal for manufacturers aiming to supply reliable pharmaceutical intermediate supplier networks with material suitable for sensitive parenteral applications, where impurity profiles are scrutinized under intense regulatory magnification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of Troxerutin typically relies on the Williamson etherification of rutin with ethylene oxide under basic conditions using catalysts such as sodium hydroxide or potassium hydroxide. However, the flavonoid structure of rutin possesses four active hydroxyl groups with relatively similar reactivities, making selective tri-substitution exceptionally difficult. Consequently, conventional processes invariably generate a complex mixture comprising monohydroxyethylrutin, dihydroxyethylrutin, trihydroxyethylrutin, and tetrahydroxyethylrutin. Separating the desired tri-substituted product from this congener mixture often necessitates energy-intensive chromatographic techniques or repeated recrystallizations, which drastically inflate production costs and reduce overall yield. Furthermore, residual impurities in standard commercial grades, often around 80.0% to 88.0% purity, pose significant safety risks for injectable formulations, including potential allergic reactions and vascular toxicity.
The Novel Approach
In stark contrast, the methodology described in CN106892950B employs a strategic pre-refinement of the starting material followed by a catalytic system based on natural sodium alginate. This biopolymer catalyst appears to modulate the reaction environment, potentially through steric hindrance or specific interactions with the hydroxyl groups, thereby enhancing the selectivity towards the tri-hydroxyethyl derivative. The process utilizes anhydrous methanol as a solvent in a high-pressure autoclave, allowing for precise control over reaction parameters such as temperature and pressure. This refined approach eliminates the need for complex protective group strategies or expensive chromatographic separations, enabling the direct isolation of high-purity crystals through simple acidification and cooling. Such a streamlined workflow represents a significant advancement in cost reduction in pharmaceutical intermediate manufacturing, offering a scalable alternative to legacy processes.
Mechanistic Insights into Sodium Alginate-Catalyzed Etherification
The core chemical transformation involves the nucleophilic attack of the deprotonated hydroxyl groups on the rutin scaffold by ethylene oxide. In standard alkaline media, the lack of differentiation between the 7-OH, 3'-OH, 4'-OH, and other hydroxyl positions leads to random substitution patterns. The introduction of sodium alginate, a linear copolymer of β-D-mannuronic acid and α-L-guluronic acid, introduces a unique microenvironment. While the exact mechanistic interaction remains a subject of detailed kinetic study, it is hypothesized that the polyanionic nature of the alginate chain may create a localized pH gradient or coordinate with the cationic species, effectively shielding certain hydroxyl positions or stabilizing the transition state for the desired tri-substitution. This selectivity is crucial for minimizing the formation of the tetra-hydroxyethyl byproduct, which is notoriously difficult to remove due to its structural similarity to the target molecule.
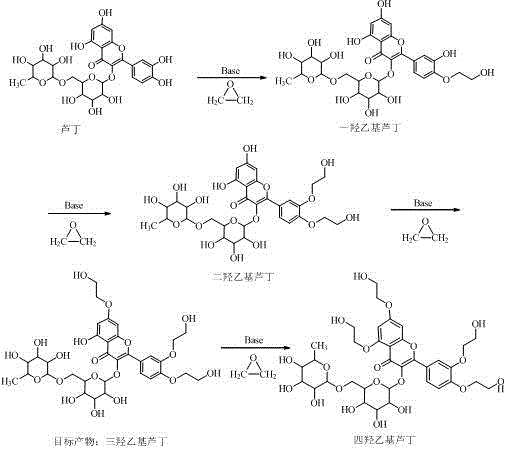
Furthermore, the impurity control mechanism is deeply rooted in the quality of the starting material. By refining crude rutin to >99.0% purity using an aqueous alkaline dissolution followed by diatomaceous earth adsorption, the process removes flavonoid congeners and degradation products that could otherwise participate in side reactions. This upstream purification ensures that the catalytic cycle operates on a homogeneous substrate, reducing the burden on downstream processing. The final crystallization step, aided by activated carbon adsorption, effectively scavenges any remaining colored impurities or trace catalyst residues, ensuring the final high-purity pharmaceutical intermediate meets the stringent specifications required for global regulatory compliance.
How to Synthesize Troxerutin Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for reproducing these high-yield results in a pilot or production setting. The procedure emphasizes the critical importance of maintaining strict stoichiometric ratios between the high-purity rutin, ethylene oxide, and the sodium alginate catalyst within an anhydrous methanol medium. Operators must carefully monitor the reaction progress via HPLC to terminate the process at the optimal conversion point, typically when the trihydroxyethylrutin ratio reaches approximately 86%, to prevent over-alkylation. For a comprehensive breakdown of the specific operational parameters, including temperature ramps and workup procedures, please refer to the standardized guide below.
- Purify crude rutin using an alkaline aqueous solution and diatomaceous earth adsorption to achieve >99.0% purity.
- React high-purity rutin with ethylene oxide in anhydrous methanol using natural sodium alginate as a catalyst at 60-90°C.
- Purify the resulting trihydroxyethylrutin via dissolution, activated carbon adsorption, and recrystallization to obtain >98.0% Troxerutin.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards this sodium alginate-catalyzed process offers compelling economic arguments beyond mere yield improvements. The elimination of complex chromatographic purification steps, which are often cited in prior art as necessary for achieving >98% purity, translates directly into substantial cost savings. The patent explicitly notes a production cost range of 400-600 yuan/Kg for this method, which is markedly lower than the 2000-3000 yuan/Kg associated with certain high-purity prior art methods relying on intricate separation techniques. This drastic reduction in processing complexity allows for more predictable budgeting and reduced capital expenditure on specialized separation equipment.
- Cost Reduction in Manufacturing: The utilization of inexpensive, naturally derived sodium alginate replaces costly synthetic catalysts or complex multi-stage protection-deprotection sequences. By simplifying the purification to a straightforward crystallization and adsorption process, manufacturers can significantly lower utility consumption and solvent waste disposal costs. This efficiency gain ensures that the final high-purity Troxerutin is commercially viable for mass-market cardiovascular therapies without compromising on quality standards.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically rutin and ethylene oxide, are commodity chemicals with well-established global supply chains. Unlike processes dependent on exotic ligands or scarce transition metals, this method mitigates the risk of supply disruptions. The robustness of the reaction conditions, operating at moderate temperatures of 60-90°C, further ensures consistent batch-to-batch reproducibility, which is essential for maintaining long-term contracts with generic drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard high-pressure autoclaves common in fine chemical facilities. Moreover, the initial purification of rutin uses water as a solvent, aligning with green chemistry initiatives to reduce volatile organic compound (VOC) emissions. The non-toxic nature of the sodium alginate catalyst simplifies waste treatment protocols, facilitating easier compliance with increasingly stringent environmental regulations in major manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical rationale behind the innovation.
Q: Why is sodium alginate preferred over traditional alkali catalysts for Troxerutin synthesis?
A: Sodium alginate offers superior selectivity in the Williamson etherification of rutin, minimizing the formation of unwanted tetra-hydroxyethyl byproducts and simplifying downstream purification compared to harsh bases like NaOH.
Q: What is the achievable purity level using this novel preparation method?
A: The patented process consistently yields Troxerutin with a content higher than 98.0%, meeting stringent requirements for injectable pharmaceutical grades.
Q: How does this method address environmental concerns in API manufacturing?
A: By utilizing water for the initial rutin purification and avoiding heavy metal catalysts, the process significantly reduces toxic waste generation and aligns with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Troxerutin Supplier
The technical advancements detailed in CN106892950B underscore the potential for producing high-quality Troxerutin that meets the rigorous demands of modern pharmacopeia. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity intermediates. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Troxerutin delivered is free from critical impurities and ready for formulation into life-saving anticoagulant and thrombolytic therapies.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can evaluate the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring a seamless transition to a more efficient and cost-effective sourcing strategy for your cardiovascular drug portfolio.
