Advanced Resin-Assisted Hydroxyethylation for High-Content Troxerutin Manufacturing
Advanced Resin-Assisted Hydroxyethylation for High-Content Troxerutin Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes for vascular protectants, and the preparation of troxerutin (3',4',7-trihydroxyethyl rutin) remains a critical process for producing high-quality API intermediates. Patent CN100503627C introduces a groundbreaking methodology that fundamentally alters the hydroxyethylation landscape by integrating cation exchange resins directly into the reaction matrix. This innovation addresses the longstanding challenge of controlling side reactions in highly alkaline environments, offering a pathway to crude products with 3',4',7-troxerutin content exceeding 78%, and optimally surpassing 85%. For R&D directors and procurement specialists, this patent represents a significant leap forward in process reliability, demonstrating how precise stoichiometric control of water and sodium hydroxide, combined with dynamic pH regulation via resin, can drastically minimize impurity profiles while enhancing overall yield efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis protocols for troxerutin have historically relied on excessive solvent volumes and aggressive alkaline conditions, creating a precarious environment for the flavonoid backbone of rutin. As documented in prior art, conventional methods often utilize water ratios up to five times the weight of the rutin substrate, which not only dilutes the reaction mixture but also introduces substantial amounts of dissolved oxygen that drive oxidative degradation. Furthermore, the reliance on high concentrations of sodium hydroxide without adequate buffering mechanisms leads to rapid pH spikes that exceed the stability threshold of the glycosidic bond. Under these harsh conditions, rutin is prone to hydrolysis, cleaving the sugar moiety to form quercetin aglycone, which subsequently undergoes oxidative ring opening. This degradation pathway is visually represented in the following reaction scheme, illustrating how the valuable starting material is lost to non-pharmacologically active by-products.
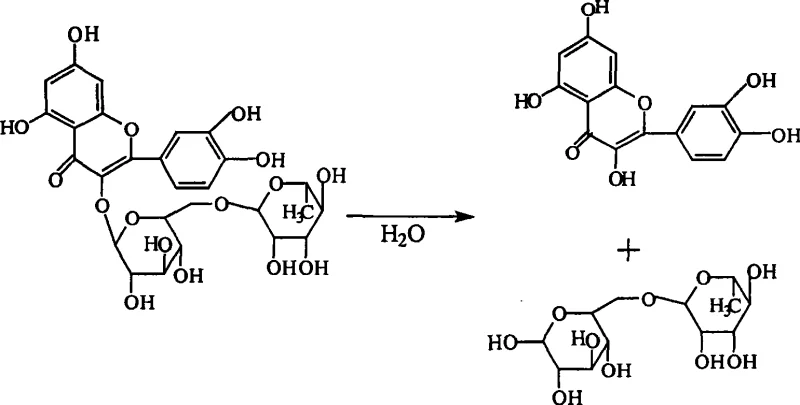
The consequences of this hydrolysis are severe for downstream processing, as the resulting aglycones and their oxidation products, such as phenols and aromatic acids, possess structural similarities to the target molecule that make purification arduous and costly. These impurities not only reduce the overall yield of the desired 3',4',7-substituted product but also pose potential safety hazards if carried through to the final API. Moreover, the high water content necessitates energy-intensive evaporation steps to isolate the solid product, inflating utility costs and extending production cycles. The inability to effectively sequester trace transition metal ions, which are naturally present in plant-derived rutin extracts, further exacerbates the problem by catalyzing unwanted oxidation reactions, leading to a final product that often struggles to meet stringent heavy metal specifications required for injectable grades.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN100503627C employs a minimalist solvent strategy and a sophisticated in-situ purification mechanism to preserve the integrity of the rutin scaffold. By strictly limiting the water-to-rutin weight ratio to a range of 1:1 to 1:3, preferably 1:1 to 1:1.5, the process significantly reduces the total volume of dissolved oxygen available for side reactions, thereby kinetically favoring the desired nucleophilic attack of ethylene oxide on the phenolic hydroxyl groups. Crucially, the introduction of a hydrogen-type cation exchange resin at a specific pH window of 9.0 to 9.5 serves a dual function: it acts as a physical buffer to slow the rate of pH increase, preventing the localized high-alkalinity zones that trigger hydrolysis, and simultaneously adsorbs trace heavy metal ions like copper and iron. This strategic intervention ensures that the reaction proceeds smoothly to the target endpoint pH of 9.5 to 10.3 without the catastrophic degradation seen in conventional high-water processes, resulting in a crude product of exceptional quality.
Mechanistic Insights into Resin-Assisted Hydroxyethylation and Impurity Control
To fully appreciate the technical superiority of this method, one must understand the intricate interplay between the resin's adsorption capacity and the reaction kinetics of ethylene oxide. The cation exchange resin possesses a higher affinity for polyvalent transition metal ions than for monovalent sodium ions, allowing it to effectively scavenge catalytic impurities from the plant-derived rutin feedstock before they can initiate radical oxidation chains. By removing these metal catalysts, the rate of oxidative degradation of the quercetin nucleus is dramatically suppressed, preserving the core structure necessary for pharmacological activity. Simultaneously, the resin's hydrogen ions provide a localized buffering effect that moderates the alkalinity of the microenvironment around the reacting molecules. This prevents the pH from rising too rapidly, which is critical because the hydroxyethylation reaction is exothermic and autocatalytic; without this buffer, the reaction could runaway, pushing the pH into a range where the glycosidic bond becomes unstable and hydrolysis dominates over etherification.
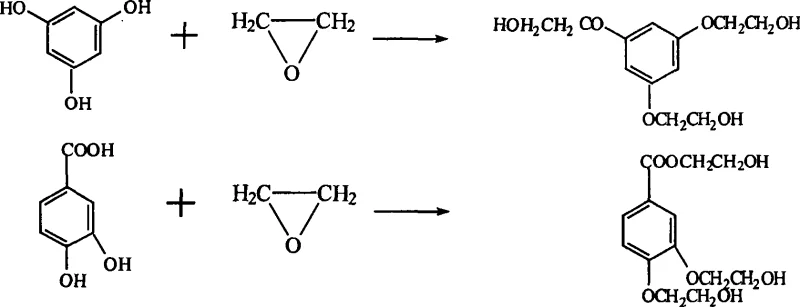
Furthermore, the control of impurity formation extends beyond simple hydrolysis prevention to the suppression of secondary reactions involving degradation products. If rutin were to hydrolyze and oxidize into phenolic acids, these species would still contain reactive hydroxyl groups capable of reacting with ethylene oxide, generating hydroxyethylated impurities that are structurally analogous to troxerutin and difficult to separate. The reaction pathway for these troublesome by-products is depicted below, highlighting the complexity of the impurity profile that arises when the primary substrate is compromised. By maintaining the integrity of the rutin molecule through precise pH control and metal ion removal, the novel process effectively shuts down this parallel reaction channel, ensuring that the ethylene oxide reagent is consumed primarily for the intended 3', 4', and 7-position substitution rather than being wasted on scavenging degradation fragments.
How to Synthesize 3',4',7-Trihydroxyethyl Rutin Efficiently
The implementation of this synthesis route requires careful attention to the timing of reagent addition and the monitoring of physicochemical parameters to maximize the benefits of the resin assistance. The process begins with the formation of the rutin sodium salt in a minimal amount of water, followed by the controlled introduction of ethylene oxide gas. The critical operational pivot point occurs when the reaction mixture reaches a pH of approximately 9.1, at which stage the temperature is lowered, and the resin is introduced to stabilize the system for the remainder of the etherification. This specific sequence ensures that the initial deprotonation is complete while preventing the subsequent pH surge from damaging the product. For a comprehensive breakdown of the exact temperatures, stirring rates, and workup procedures required to replicate these results, please refer to the standardized synthesis guide provided below.
- Charge rutin, water (1: 1 to 1:3 ratio), and sodium hydroxide (100:0.82 ratio) into a reactor at 75°C under nitrogen protection.
- Introduce ethylene oxide and monitor pH; when pH reaches 9.0-9.5, add cation exchange resin and lower temperature to 60°C.
- Continue reaction until pH reaches 9.5-10.3, terminate with hydrochloric acid to pH 4.0-5.0, then hot filter, concentrate, and vacuum dry.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this resin-assisted hydroxyethylation technology offers profound advantages for supply chain stability and cost management, particularly for large-scale manufacturers of vascular therapeutic agents. The most immediate impact is observed in the drastic reduction of solvent usage, where the water ratio is cut from the traditional 5:1 down to nearly 1:1. This reduction translates directly into significant energy savings during the concentration and drying phases, as there is substantially less water to evaporate, thereby lowering the carbon footprint and utility costs associated with each batch. Additionally, the improved selectivity of the reaction means that less raw material is lost to degradation by-products, effectively increasing the mass yield per kilogram of rutin input and reducing the cost of goods sold without requiring expensive raw material substitutions.
- Cost Reduction in Manufacturing: The elimination of excessive solvent volumes and the reduction in reaction time due to higher concentration lead to a more efficient use of reactor capacity and energy resources. By avoiding the need for complex downstream purification steps to remove hydrolysis by-products, the overall processing time is shortened, allowing for faster batch turnover and reduced labor costs. The ability to achieve high crude purity (>78%) means that subsequent recrystallization steps are more efficient, requiring less solvent and yielding higher recovery rates of the final API, which cumulatively drives down the manufacturing cost per unit.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in raw material quality, specifically regarding trace metal content in plant-derived rutin, ensures consistent batch-to-batch performance. The resin acts as a safeguard, absorbing fluctuations in feedstock quality that might otherwise lead to failed batches or off-specification products in conventional processes. This reliability minimizes the risk of production delays and supply interruptions, providing procurement managers with greater confidence in meeting delivery schedules for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process generates significantly less wastewater due to the reduced water input, simplifying effluent treatment and lowering environmental compliance costs. The use of a solid resin that can be filtered off eliminates the need for liquid-liquid extraction steps that often generate large volumes of organic waste. Furthermore, the final product consistently meets stringent heavy metal limits (<10ppm for injectable grade) without requiring additional chelation treatments, streamlining the regulatory approval process for new drug filings and ensuring continuous market access.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route, drawing directly from the experimental data and mechanistic explanations provided in the source documentation. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for commercial production or for quality assurance personnel verifying the purity profiles of incoming troxerutin supplies. The answers reflect the specific advantages of the resin-assisted approach over traditional alkaline hydroxyethylation methods.
Q: How does the resin improve troxerutin purity?
A: The cation exchange resin acts as a buffer to control pH rise and adsorbs transition metal ions that catalyze oxidation, reducing heavy metals to <10ppm and preventing oxidative degradation.
Q: What is the achievable content of 3',4',7-troxerutin?
A: The crude product achieves >78% content (optimally >85%), and single recrystallization with methanol can reach 95%-99% purity.
Q: Why is water ratio critical in this synthesis?
A: Reducing water from traditional 5x ratios to 1:1-1:3 minimizes dissolved oxygen and hydrolysis risks, significantly lowering oxidation by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Troxerutin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality oversight. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of pH and temperature required for this resin-assisted synthesis is maintained perfectly at an industrial scale. We are committed to delivering high-purity troxerutin that adheres to stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the >78% crude content benchmark and the <10ppm heavy metal standard essential for parenteral applications.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain for vascular health medications. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how the reduced solvent and energy consumption of this method translates to bottom-line value. We encourage you to request specific COA data and route feasibility assessments to validate the superior quality and consistency of our troxerutin intermediates against your internal standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
