Advanced Oxidation Technology for High-Purity Formylcyclopropanecarboxylate Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and efficient pathways for synthesizing critical intermediates, particularly those containing sensitive functional groups like the cyclopropane ring. Patent CN100341842C introduces a groundbreaking method for the production of formylcyclopropanecarboxylate compounds, which serve as vital precursors in the synthesis of pyrethroid insecticides and various pharmaceutical agents. This technology represents a significant departure from traditional oxidation methods by utilizing a nitroxyl radical compound in conjunction with mild oxidizers such as hypohalites or N-halosuccinimides. The core innovation lies in the ability to achieve high conversion rates under mild conditions while completely avoiding the use of toxic heavy metals and malodorous sulfur by-products. For R&D directors and process chemists, this patent offers a robust solution to the longstanding challenges of waste management and process safety associated with older oxidation technologies. By leveraging this specific catalytic system, manufacturers can achieve superior purity profiles essential for downstream applications in high-value agrochemical formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of hydroxymethyl cyclopropane derivatives to their corresponding aldehydes has relied heavily on reagents such as pyridinium chlorochromate (PCC) or the Swern oxidation protocol. The use of PCC presents severe environmental and regulatory hurdles due to the generation of hexavalent chromium waste, which requires complex and costly disposal procedures to meet modern environmental standards. Furthermore, the Swern oxidation, while effective, involves the use of dimethyl sulfoxide and oxalyl chloride at cryogenic temperatures, leading to the formation of dimethyl sulfide, a compound notorious for its extremely unpleasant odor and potential safety hazards in large-scale operations. These conventional methods often suffer from exothermic risks that make temperature control difficult, potentially leading to the degradation of the thermally sensitive cyclopropane ring structure. Consequently, the overall process efficiency is compromised by the need for extensive purification steps to remove metal residues or sulfur-containing impurities, driving up both operational costs and production lead times for key intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN100341842C employs a catalytic amount of a nitroxyl radical compound, such as 2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPO), combined with inexpensive stoichiometric oxidants like sodium hypochlorite. This approach operates effectively at temperatures ranging from -5°C to 50°C, eliminating the need for energy-intensive cryogenic cooling. The reaction proceeds through a selective oxidation mechanism that preserves the stereochemical integrity of the cyclopropane ring, ensuring that both cis and trans isomers, as well as optical isomers, are maintained without racemization. By avoiding heavy metals and sulfur reagents, the process generates a much cleaner crude product profile, significantly simplifying the downstream workup and purification stages. This technological shift not only enhances the safety profile of the manufacturing facility but also aligns perfectly with the growing global demand for green chemistry solutions in the production of reliable agrochemical intermediate supplies.
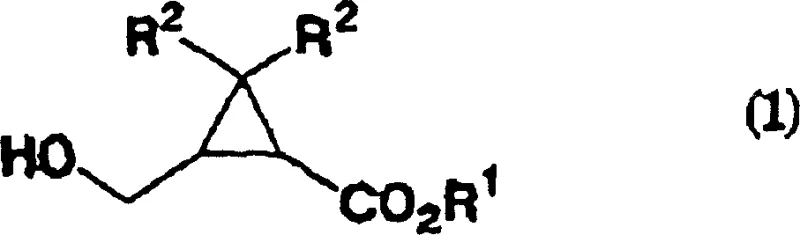
The starting material for this transformation is the cyclopropanecarboxylate compound of formula (1), where the hydroxymethyl group is selectively oxidized. As illustrated in the structure, the molecule contains a strained three-membered ring which is susceptible to ring-opening under harsh acidic or basic conditions. The novelty of this patent lies in the buffered reaction conditions that protect this sensitive motif. The use of buffers such as sodium bicarbonate or potassium phosphate maintains the pH within a narrow window of 6 to 13, preferably 8 to 10. This precise control prevents the acid-catalyzed hydrolysis of the ester group or the ring-opening of the cyclopropane, which are common side reactions in non-optimized oxidation processes. The versatility of the method allows for a wide range of R1 groups, including linear, branched, or cyclic alkyl groups, as well as substituted aryl and aralkyl groups, making it applicable to a diverse library of substrates used in fine chemical synthesis.
Mechanistic Insights into Nitroxyl Radical-Catalyzed Oxidation
The mechanistic pathway of this reaction involves the generation of an oxoammonium cation from the nitroxyl radical precursor by the action of the primary oxidant, such as sodium hypochlorite. This highly reactive oxoammonium species acts as the actual oxidizing agent that abstracts a hydride from the alpha-carbon of the alcohol substrate, converting the hydroxymethyl group into an aldehyde functionality while being reduced back to the hydroxylamine form. The hydroxylamine is subsequently re-oxidized by the stoichiometric oxidant to regenerate the active nitroxyl radical or oxoammonium species, completing the catalytic cycle. This redox cycle is highly efficient and allows for the use of the nitroxyl compound in catalytic quantities, typically ranging from 0.01 to 10 mol%, which drastically reduces the cost of goods compared to stoichiometric oxidants like Dess-Martin periodinane. The presence of phase transfer catalysts, such as tetrabutylammonium bromide, can further enhance the reaction rate in biphasic systems by facilitating the transport of the hypohalite anion into the organic phase where the substrate resides.
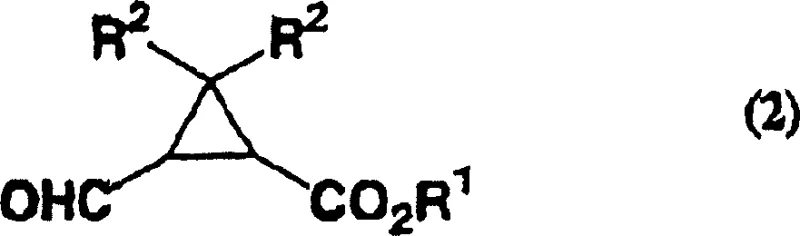
Impurity control is a critical aspect of this mechanism, particularly regarding the prevention of over-oxidation to the corresponding carboxylic acid. The selectivity of the nitroxyl radical system is inherently high for primary alcohols, stopping at the aldehyde stage under the specified conditions. However, the patent emphasizes the importance of quenching residual oxidants immediately after the reaction is complete, typically using reducing agents like sodium thiosulfate. This step is crucial to prevent any post-reaction oxidation that could degrade the product quality during workup. Furthermore, the mild pH conditions ensure that the ester linkage remains intact, avoiding saponification which would complicate the isolation of the desired ester product. For R&D teams focusing on high-purity OLED material or pharmaceutical intermediate synthesis, understanding these mechanistic nuances is essential for troubleshooting and optimizing the process for specific substrate variations, ensuring consistent batch-to-batch quality.
How to Synthesize Formylcyclopropanecarboxylate Efficiently
To implement this synthesis effectively, process engineers must carefully manage the addition rate of the oxidant to control the exotherm and maintain the optimal pH throughout the reaction course. The patent provides detailed examples demonstrating that slow addition of the oxidant over a period of one to two hours, coupled with efficient stirring, leads to superior yields compared to rapid bolus addition. It is recommended to monitor the reaction progress using gas chromatography to determine the exact endpoint, thereby minimizing the exposure of the product to oxidative conditions. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by dissolving the cyclopropanecarboxylate alcohol substrate in a suitable organic solvent such as toluene or dichloromethane, adding a nitroxyl radical catalyst like TEMPO, and introducing an aqueous buffer system to maintain pH between 8 and 10.
- Slowly add the oxidizing agent, preferably an aqueous solution of sodium hypochlorite or N-halosuccinimide, while maintaining the reaction temperature between -5°C and 50°C to ensure selective oxidation without over-oxidation.
- Upon completion, quench residual oxidants with a reducing agent like sodium thiosulfate, separate the organic layer, and purify the resulting formylcyclopropanecarboxylate ester through distillation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this nitroxyl radical oxidation technology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of chromium-based reagents removes a significant regulatory burden and reduces the costs associated with hazardous waste disposal, which can be a major hidden expense in traditional manufacturing. Additionally, the avoidance of malodorous by-products like dimethyl sulfide improves the working environment and reduces the need for specialized scrubbing equipment, leading to lower capital expenditure for facility upgrades. The use of commodity chemicals such as sodium hypochlorite and sodium bicarbonate ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. This robustness translates directly into enhanced supply continuity for customers relying on these intermediates for their own production schedules.
- Cost Reduction in Manufacturing: The shift to catalytic oxidation using inexpensive oxidants significantly lowers the raw material costs per kilogram of product. By removing the need for expensive heavy metal oxidants and the subsequent purification steps required to meet strict metal residue specifications, the overall cost of goods sold is drastically reduced. The simplified workup procedure, which often requires only phase separation and concentration rather than complex chromatography, further decreases processing time and solvent consumption. These efficiencies accumulate to provide a competitive pricing advantage in the market for agrochemical intermediates, allowing manufacturers to offer more attractive terms to downstream clients without compromising margin.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals like bleach and buffer salts ensures that production is not vulnerable to the supply disruptions that often plague specialty reagent markets. This stability is crucial for maintaining consistent delivery schedules, especially for high-volume contracts in the agrochemical sector where seasonal demand peaks must be met without fail. The mild reaction conditions also reduce the risk of batch failures due to thermal runaways or equipment corrosion, further securing the supply pipeline. For supply chain heads, this means a more predictable lead time for high-purity intermediates and a reduced need for safety stock inventory.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in standard glass-lined reactors without the need for exotic materials of construction. The absence of heavy metals simplifies the environmental permitting process and reduces the liability associated with long-term site contamination. Wastewater treatment becomes more straightforward as the effluent primarily contains benign salts and organic by-products that are easier to biodegrade. This alignment with green chemistry principles not only future-proofs the manufacturing asset against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner in the global value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of the nitroxyl radical oxidation method over traditional PCC oxidation?
A: The nitroxyl radical method eliminates the use of toxic heavy metals like chromium found in PCC, significantly reducing hazardous waste disposal costs and environmental compliance burdens while avoiding the generation of malodorous by-products associated with Swern oxidation.
Q: How does pH control impact the yield and selectivity of this oxidation process?
A: Maintaining the reaction pH within the range of 6 to 13, optimally between 8 and 10 using buffers like sodium bicarbonate or potassium phosphate, is critical for stabilizing the active oxoammonium species and preventing acid-catalyzed degradation of the sensitive cyclopropane ring.
Q: Is this synthesis method scalable for industrial production of pyrethroid intermediates?
A: Yes, the process utilizes inexpensive and commercially available oxidants like sodium hypochlorite and operates under mild temperature conditions (-5°C to 50°C), making it highly suitable for large-scale commercial manufacturing with robust safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formylcyclopropanecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of our clients' final products. Our technical team has extensively analyzed the methodology described in CN100341842C and possesses the expertise to translate this laboratory-scale innovation into robust commercial processes. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green oxidation technology are realized at an industrial level. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of formylcyclopropanecarboxylate meets the exacting standards required for pyrethroid synthesis and pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a reliable source for these essential chemical building blocks.
