Revolutionizing Phenolic Intermediate Production via Stable Ketone Oxidation Pathways
Revolutionizing Phenolic Intermediate Production via Stable Ketone Oxidation Pathways
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more robust, scalable, and environmentally conscious synthetic routes. A pivotal advancement in this domain is documented in patent CN1777572A, which outlines a sophisticated process for the preparation of alkoxyphenols and aryloxyphenols. These compounds, including critical intermediates like sesamol, serve as foundational building blocks for a vast array of pharmaceutical products, antioxidants, and agrochemical agents. The core innovation lies in shifting the synthetic paradigm from the oxidation of aromatic aldehydes to the oxidation of aromatic ketones. This strategic modification addresses long-standing stability issues associated with traditional methods, offering a pathway that is not only chemically superior but also commercially viable for large-scale operations. By leveraging the inherent stability of ketone-derived esters, manufacturers can achieve higher purity profiles and more consistent yields, making this technology a cornerstone for any reliable pharmaceutical intermediates supplier aiming to optimize their production capabilities.
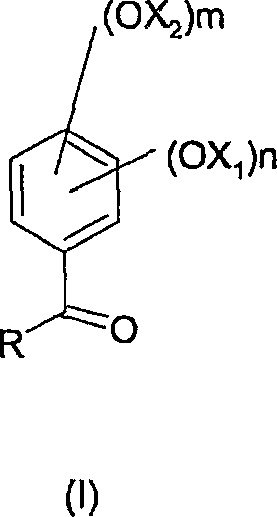
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenolic derivatives has heavily relied on the Baeyer-Villiger oxidation of aromatic aldehydes. While conceptually straightforward, this conventional approach is plagued by significant chemical inefficiencies that hinder cost reduction in pharmaceutical intermediates manufacturing. The primary drawback stems from the nature of the intermediate formed: the oxidation of an aldehyde yields a formate ester. These formate esters are notoriously unstable and highly reactive towards hydrolysis, even under mildly acidic conditions. This instability necessitates strictly anhydrous reaction environments to prevent premature decomposition, which drastically increases operational complexity and cost. Furthermore, the susceptibility of these intermediates to hydrolysis often leads to the formation of complex mixtures of by-products, complicating downstream purification and reducing the overall yield of the desired high-purity OLED material or API precursor. The reliance on harsh conditions and the generation of substantial waste streams render these older methods increasingly obsolete in a modern regulatory environment.
The Novel Approach
In stark contrast, the methodology disclosed in CN1777572A introduces a transformative solution by utilizing aromatic ketones of Formula (I) as the starting substrates. 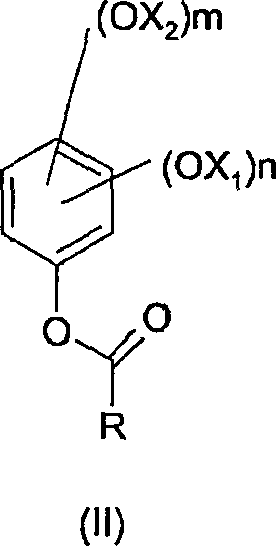
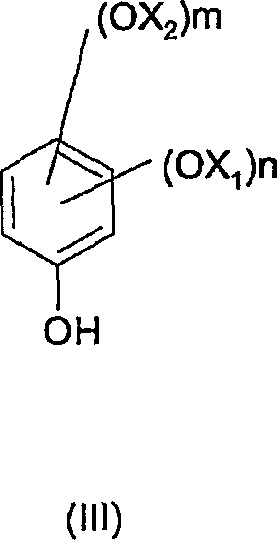 When these ketones undergo oxidation with a peracid, they yield the corresponding esters of Formula (II), which possess markedly different chemical properties compared to their formate counterparts. Crucially, these ketone-derived esters are significantly less reactive towards acidic hydrolysis. This enhanced stability allows the reaction to proceed in non-anhydrous environments, effectively eliminating the need for rigorous moisture control and expensive drying protocols. The result is a streamlined process where side reactions are minimized, and the integrity of the intermediate is preserved until the deliberate hydrolysis step. This shift not only simplifies the operational workflow but also ensures a cleaner reaction profile, facilitating the commercial scale-up of complex fine chemicals with greater predictability and safety.
When these ketones undergo oxidation with a peracid, they yield the corresponding esters of Formula (II), which possess markedly different chemical properties compared to their formate counterparts. Crucially, these ketone-derived esters are significantly less reactive towards acidic hydrolysis. This enhanced stability allows the reaction to proceed in non-anhydrous environments, effectively eliminating the need for rigorous moisture control and expensive drying protocols. The result is a streamlined process where side reactions are minimized, and the integrity of the intermediate is preserved until the deliberate hydrolysis step. This shift not only simplifies the operational workflow but also ensures a cleaner reaction profile, facilitating the commercial scale-up of complex fine chemicals with greater predictability and safety.
Mechanistic Insights into Baeyer-Villiger Oxidation of Ketones
The heart of this technological breakthrough is the precise control over the Baeyer-Villiger oxidation mechanism. In this process, the ketone substrate is treated with a peracid, which is preferably generated in situ through the reaction of hydrogen peroxide with an organic acid such as formic acid. The use of in situ generation is particularly advantageous as it avoids the handling and storage hazards associated with concentrated peracids. The reaction typically proceeds in an inert organic solvent, with dichloromethane being the preferred medium due to its excellent solubility characteristics for both the ketone and the peracid species. The temperature is carefully maintained between 30°C and 50°C, a range that balances reaction kinetics with thermal safety. During this oxidative transformation, the peracid attacks the carbonyl carbon of the ketone, leading to the migration of an alkyl or aryl group and the insertion of an oxygen atom. The specific migration aptitude is governed by the electronic and steric nature of the substituents on the aromatic ring, ensuring high regioselectivity for the desired ester isomer.
Following the oxidation, the resulting ester undergoes hydrolysis to release the final phenolic product. The patent elucidates that while acidic hydrolysis is feasible, alkaline hydrolysis offers superior performance, particularly when augmented with phase transfer catalysts. The addition of catalysts such as tetrabutylammonium chloride facilitates the transport of hydroxide ions into the organic phase, dramatically accelerating the hydrolysis rate without requiring extreme temperatures. This mechanistic nuance is critical for impurity control; by optimizing the hydrolysis conditions, manufacturers can ensure that unreacted ketones remain intact in the organic phase, allowing for their easy separation and recycling. This level of control over the reaction mechanism is what enables the production of high-purity alkoxyphenols with minimal contamination from starting materials or degradation products, meeting the stringent quality standards required by global regulatory bodies.
How to Synthesize Sesamol Efficiently
The practical application of this patented technology is best exemplified by the synthesis of sesamol, a valuable antioxidant and pharmaceutical intermediate. The process begins with the oxidation of 1,3-benzodioxol-5-propan-1-one using performic acid generated in situ. Detailed operational parameters, including reagent stoichiometry and temperature profiles, are critical for maximizing yield. Following the oxidation, the crude ester is subjected to alkaline hydrolysis in the presence of a phase transfer catalyst. For a comprehensive understanding of the specific laboratory procedures and safety precautions required to replicate this high-efficiency synthesis, please refer to the standardized protocol outlined below.
- Oxidize the aromatic ketone (Formula I) using a peracid, preferably performic acid generated in situ, in an inert solvent like dichloromethane at 30-50°C.
- Separate the organic phase containing the intermediate ester (Formula II) and wash to remove residual peroxides.
- Hydrolyze the ester under basic conditions using sodium hydroxide and a phase transfer catalyst to yield the final phenol (Formula III).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ketone-based oxidation technology translates into tangible strategic benefits that extend far beyond simple chemical yield. The robustness of the process directly addresses key pain points in the sourcing of critical intermediates, offering a more resilient supply chain model. By eliminating the need for strictly anhydrous conditions and expensive stabilization protocols, the overall manufacturing overhead is significantly reduced. Furthermore, the ability to recycle unreacted starting materials creates a closed-loop system that minimizes raw material consumption and waste disposal costs. These factors combine to create a more sustainable and economically attractive production model, ensuring that partners can secure a steady flow of materials without the volatility associated with less efficient synthetic routes.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by fundamental chemical efficiencies rather than arbitrary price cuts. By utilizing ketones instead of aldehydes, the process avoids the formation of unstable formate esters, thereby eliminating the need for costly anhydrous solvents and rigorous moisture exclusion systems. Additionally, the in situ generation of peracids from commodity chemicals like hydrogen peroxide and formic acid reduces the dependency on specialized, high-cost oxidizing agents. The ability to recover and recycle unreacted ketones from the organic phase further amplifies these savings, as it effectively lowers the net consumption of starting materials per unit of product. This holistic approach to cost optimization ensures long-term price stability for buyers seeking a reliable pharmaceutical intermediates supplier.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that are sensitive to minor variations in conditions. The tolerance of this ketone oxidation method to non-anhydrous environments makes it inherently more robust against environmental fluctuations and equipment limitations. This resilience reduces the risk of batch failures and production delays, ensuring consistent delivery schedules. Moreover, the starting materials, such as substituted acetophenones and propiophenones, are widely available from established chemical vendors, reducing the risk of raw material shortages. The simplified workup procedures, which do not require complex distillation setups for solvent removal prior to hydrolysis, also shorten the overall cycle time, contributing to reducing lead time for high-purity phenolic derivatives.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often exposes hidden inefficiencies, but this technology is designed with scalability in mind. The reaction operates at mild temperatures (30-50°C) and atmospheric pressure, removing the need for high-energy heating or pressurized reactors. From an environmental perspective, the minimization of by-products and the ability to recycle organic phases significantly reduce the volume of hazardous waste generated. The use of dichloromethane, while requiring careful management, allows for efficient phase separation and solvent recovery, aligning with modern green chemistry principles. This combination of operational simplicity and waste reduction facilitates easier regulatory approval and supports the growing demand for environmentally responsible manufacturing practices in the fine chemical sector.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis route is essential for stakeholders evaluating its potential impact on their supply chains. The following questions address common inquiries regarding the process mechanics, safety, and quality implications derived directly from the patent specifications. These insights are intended to provide clarity on how this technology differentiates itself from legacy methods and what it means for product quality assurance.
Q: Why is the ketone-based route superior to traditional aldehyde oxidation?
A: Traditional aldehyde oxidation produces formate esters which are highly unstable and prone to hydrolysis, leading to significant by-product formation. The ketone-based route described in CN1777572A generates esters that are significantly less reactive towards acidic hydrolysis, allowing for non-anhydrous reaction environments and minimizing impurities.
Q: Can the unreacted starting materials be recovered in this process?
A: Yes, the process is designed for high efficiency. Unreacted ketones and non-hydrolyzed esters remaining in the organic phase after hydrolysis can be easily separated and recycled back into the oxidation step, substantially reducing raw material waste.
Q: What are the optimal conditions for the hydrolysis step?
A: While acidic hydrolysis is possible, the patent highlights that alkaline hydrolysis is preferred. Specifically, using sodium hydroxide in the presence of a phase transfer catalyst like tetrabutylammonium chloride significantly enhances conversion rates and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sesamol Supplier
The technological advancements detailed in CN1777572A represent a significant leap forward in the production of alkoxyphenols, yet realizing their full potential requires a partner with deep technical expertise and industrial capacity. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards. We understand that in the pharmaceutical and fine chemical industries, consistency is paramount, and our state-of-the-art facilities are designed to deliver the reliability that global enterprises demand.
We invite you to explore how our advanced manufacturing capabilities can enhance your supply chain efficiency and product quality. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of ketone oxidation technology can drive value and innovation for your organization, securing your position in a competitive global market.
