Advanced Three-Step Synthesis of Sesamol for Commercial Scale-Up and High Purity
The global demand for high-purity 3,4-methylenedioxyphenol, commonly known as sesamol, has surged due to its critical role as an antioxidant in food preservation and as a key building block in the synthesis of antihypertensive and cardiovascular pharmaceuticals. Recent intellectual property developments, specifically patent CN113563301A published in late 2021, have introduced a transformative synthesis process that addresses long-standing inefficiencies in the production of this valuable fine chemical. This novel methodology utilizes piperonyl as the starting material, executing a streamlined three-step reaction sequence that includes acylation, Baeyer-Villiger oxidation, and hydrolysis. Unlike traditional extraction methods from sesame oil which are plagued by high solvent consumption and inconsistent quality, or older synthetic routes involving hazardous diazotization, this new approach offers a robust pathway for reliable sesamol supplier operations. The technical breakthrough lies in the meticulous control of reaction conditions and the strategic selection of catalysts, which collectively ensure high conversion rates and exceptional product purity. For R&D directors and procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing, understanding the nuances of this patent is essential for optimizing supply chains and ensuring the continuous availability of this critical API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of sesamol has been hindered by significant technical and economic barriers inherent in conventional methodologies. The extraction of sesamol directly from sesame oil, while natural, is economically unviable for large-scale industrial applications due to the massive volumes of solvents required and the relatively low yield obtained from the raw biomass. Furthermore, synthetic routes relying on piperonyl amine precursors often necessitate diazotization and hydrolysis reactions, which are not only hazardous but also generate coupling side reactions that produce difficult-to-remove pigment substances, resulting in poor product color and limited application scope. Another prevalent method involving glyoxylic acid and piperonyl suffers from incomplete conversion of raw materials, leading to increased wastewater treatment costs and higher unit consumption of expensive reagents. Additionally, the use of nitric acid in piperonal preparation often causes discoloration, and subsequent Baeyer-Villiger oxidation steps in older patents frequently struggle with the separation of byproduct piperic acid, creating a bottleneck in the purification process that compromises the final quality of the sesamol.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in patent CN113563301A presents a streamlined and highly efficient alternative that fundamentally restructures the synthesis workflow. By initiating the process with piperonyl and employing a direct acylation step to form 3,4-methylenedioxyacetophenone, the route bypasses the need for complex catalyst preparation or hazardous diazotization reagents. The core innovation is the subsequent Baeyer-Villiger oxidation, which is conducted under mild conditions using a carefully selected solvent and oxidation system that maximizes efficiency while minimizing safety risks. A critical advantage of this novel approach is the integration of a solvent recovery step immediately following the oxidation reaction, which effectively separates unreacted ketone intermediates from the mixture before the final hydrolysis stage. This strategic separation ensures that the crude sesamol entering the rectification phase already possesses a purity exceeding 98 percent, drastically simplifying the final purification and ensuring a finished product quality of 99.9 percent. This process not only enhances the commercial scale-up of complex pharmaceutical intermediates but also significantly reduces the environmental footprint by lowering three-waste generation.
Mechanistic Insights into Baeyer-Villiger Oxidation and Impurity Control
The chemical elegance of this synthesis lies in the precise execution of the Baeyer-Villiger oxidation, a pivotal transformation that converts the ketone intermediate into the corresponding ester with high fidelity. In this specific embodiment, the reaction is facilitated by the use of oxidants such as hydrogen peroxide or MCPBA in the presence of acidic catalysts like sulfuric acid within solvents such as dichloroethane or toluene. When hydrogen peroxide is utilized, it reacts with acetic acid within the system to generate peroxyacetic acid in situ, a mechanism that prevents the dangerous accumulation of unreacted hydrogen peroxide and eliminates safety hazards associated with oxidant stability. The reaction is maintained at a controlled temperature range of 25-50°C, which is sufficiently mild to prevent thermal degradation of the sensitive methylenedioxy ring structure while providing enough energy to drive the oxidation to completion. The molar ratio of the ketone to the oxidant is tightly controlled between 0.8:1 and 1.2:1, ensuring that the oxidant is neither in excessive surplus, which would complicate waste treatment, nor in deficit, which would leave unreacted starting material. This precise stoichiometric balance is crucial for maintaining the integrity of the reaction mixture and setting the stage for high-yield downstream processing.
Following the oxidation, the process incorporates a sophisticated impurity control mechanism that distinguishes it from prior art. The post-treatment stage involves the recovery of the first solvent through phase separation or rotary evaporation, a step that serves the dual purpose of recycling valuable solvents and removing unreacted 3,4-methylenedioxyacetophenone from the reaction mixture. By eliminating the unreacted ketone prior to the hydrolysis step, the process prevents the co-elution of starting materials with the final product during rectification, a common issue in less optimized routes. Consequently, the crude sesamol obtained after hydrolysis and pH adjustment to 3-4 exhibits a purity level of more than 98 percent, which is exceptionally high for a crude intermediate. This high initial purity reduces the load on the final rectification column, minimizes energy consumption, and ensures that the final distilled product consistently meets the stringent 99.9 percent purity specification required for pharmaceutical applications. The ability to achieve such high purity without complex chromatographic separation or extensive washing procedures underscores the robustness of this synthetic design.
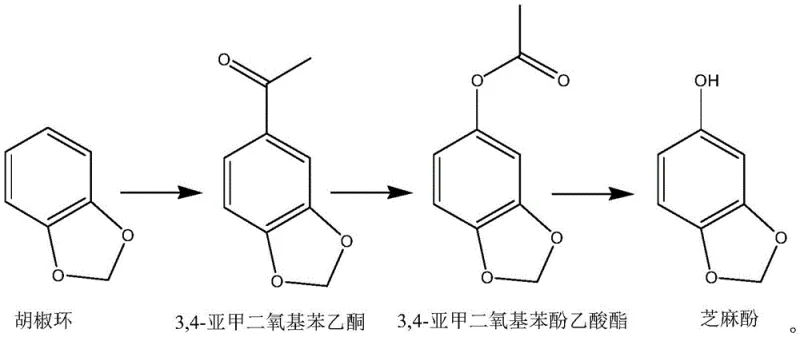
How to Synthesize 3,4-Methylenedioxyphenol Efficiently
The practical implementation of this synthesis route requires careful attention to the sequential addition of reagents and the maintenance of specific thermal profiles to ensure optimal yield and safety. The process begins with the acylation of piperonyl using acetic anhydride and a Lewis acid catalyst such as aluminum chloride or zinc chloride at temperatures between 50-90°C, followed by the critical oxidation step where the ketone intermediate is treated with the oxidant system at 25-50°C. The final hydrolysis is conducted using sodium hydroxide solution at 20-60°C, followed by neutralization and solvent recovery. While the general parameters are well-defined, the exact scaling of these steps from laboratory to production scale requires precise engineering to manage exotherms and solvent loads effectively. For detailed operational protocols, the standardized synthesis steps see the guide below which outlines the specific molar ratios and workup procedures validated in the patent examples.
- Acylation of piperonyl with acetic anhydride using Lewis acid catalysts at 50-90°C to form 3,4-methylenedioxyacetophenone.
- Baeyer-Villiger oxidation of the ketone intermediate using hydrogen peroxide or MCPBA at 25-50°C to generate the acetate ester.
- Alkaline hydrolysis of the ester followed by pH adjustment and rectification to isolate 99.9% pure 3,4-methylenedioxyphenol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis technology offers profound advantages for procurement managers and supply chain heads who are tasked with securing reliable sources of high-purity fine chemicals while managing budgetary constraints. The elimination of hazardous diazotization steps and the avoidance of complex supported catalyst preparations significantly simplify the raw material sourcing strategy, reducing dependency on specialized or hard-to-find reagents. Furthermore, the ability to recover and reuse solvents like dichloroethane and dichloromethane directly within the process flow creates a closed-loop system that drastically lowers the unit cost of production by minimizing raw material waste. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to substantial cost savings in utility expenses over the lifecycle of the production campaign. For supply chain planners, the robustness of this three-step route ensures greater predictability in production schedules, as the process is less susceptible to the variability and side-reactions that often cause delays in more complex synthetic pathways.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the strategic removal of expensive and hazardous processing steps found in legacy methods. By avoiding the use of modified supported catalysts and eliminating the need for extensive wastewater treatment associated with glyoxylic acid residues, the overall operational expenditure is significantly reduced. The in situ generation of peroxyacetic acid prevents the accumulation of unstable oxidants, thereby reducing safety compliance costs and insurance premiums associated with hazardous material storage. Additionally, the high conversion rate of the raw material means that less starting material is wasted, directly improving the material yield and lowering the cost of goods sold. These factors combine to create a manufacturing profile that is highly competitive in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The simplicity of the reaction sequence enhances the reliability of the supply chain by reducing the number of potential failure points in the manufacturing process. Since the process does not rely on biological extraction which is subject to agricultural variability, or complex multi-step sequences with low yields, production continuity is greatly improved. The use of common industrial solvents and reagents ensures that raw material availability is not a bottleneck, allowing for consistent production runs throughout the year. This stability is crucial for downstream pharmaceutical manufacturers who require just-in-time delivery of critical intermediates to maintain their own production schedules without interruption. The robust nature of the chemistry also allows for flexible batch sizing, accommodating both small-scale clinical trial needs and large-scale commercial demands.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the use of standard reaction equipment and the absence of extreme pressure or temperature requirements. The efficient solvent recovery system not only lowers costs but also aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions. By minimizing the generation of hazardous byproducts and ensuring that unreacted materials are separated and potentially recycled, the process supports a greener manufacturing footprint. This environmental compliance is a key asset for companies aiming to meet corporate sustainability goals and satisfy the auditing requirements of major multinational pharmaceutical clients who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and beneficial effects. These answers are designed to provide clarity on the operational feasibility and quality advantages of the new route compared to existing market standards. Understanding these details is vital for technical teams evaluating the adoption of this process for their own manufacturing lines or for procurement teams assessing supplier capabilities. The responses reflect the specific data points regarding purity, yield, and safety mechanisms outlined in the intellectual property documentation.
Q: How does this new process improve impurity control compared to traditional extraction?
A: Unlike sesame oil extraction which suffers from high solvent consumption and variable quality, this synthetic route utilizes a specific Baeyer-Villiger oxidation followed by solvent recovery. This step effectively removes unreacted ketone intermediates before hydrolysis, ensuring crude product purity exceeds 98% and final rectification yields 99.9% purity without pigment contamination.
Q: What are the safety advantages of using hydrogen peroxide in this oxidation system?
A: The patent specifies a system where hydrogen peroxide reacts with acetic acid to generate peroxyacetic acid in situ. This prevents the accumulation of unstable hydrogen peroxide in the reaction system, significantly mitigating safety risks associated with oxidant storage and handling while maintaining high oxidation efficiency at mild temperatures between 25-50°C.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids complex catalyst preparation and hazardous diazotization steps found in prior art. The use of standard solvents like dichloroethane and recoverable reagents, combined with mild reaction conditions, allows for commercial scale-up of complex pharmaceutical intermediates with reduced three-waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sesamol Supplier
The synthesis process detailed in patent CN113563301A represents a significant leap forward in the production of high-purity sesamol, offering a pathway that is safer, more efficient, and economically superior to traditional methods. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in an industrial setting. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 3,4-methylenedioxyphenol meets the exacting standards required for pharmaceutical and food grade applications. We understand the critical nature of API intermediates in the global supply chain and are committed to delivering consistent quality and reliability to our partners.
We invite you to collaborate with us to optimize your supply chain for sesamol and related fine chemicals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our implementation of this advanced synthesis technology can drive value for your organization. By partnering with us, you gain access to a supply source that combines cutting-edge chemical innovation with the operational excellence of a leading CDMO provider.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
