Advanced Microwave-Assisted Synthesis of Ganciclovir for Commercial Scale Production
Advanced Microwave-Assisted Synthesis of Ganciclovir for Commercial Scale Production
The pharmaceutical industry constantly seeks robust synthetic routes for critical antiviral agents, and the preparation of ganciclovir stands as a prime example where process innovation drives commercial viability. Patent CN108467396B, published in July 2021, introduces a transformative methodology that leverages microwave reactor technology to synthesize ganciclovir with exceptional efficiency and purity. This technical breakthrough addresses long-standing challenges in nucleoside analog manufacturing, specifically targeting the issues of low conversion rates, difficult purification, and hazardous reagent usage that have plagued conventional production lines. By shifting from traditional thermal heating to dielectric heating mechanisms, the process achieves a dramatic reduction in reaction times while simultaneously enhancing selectivity. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards greener, more cost-effective manufacturing protocols that do not compromise on the stringent quality standards required for active pharmaceutical ingredients.
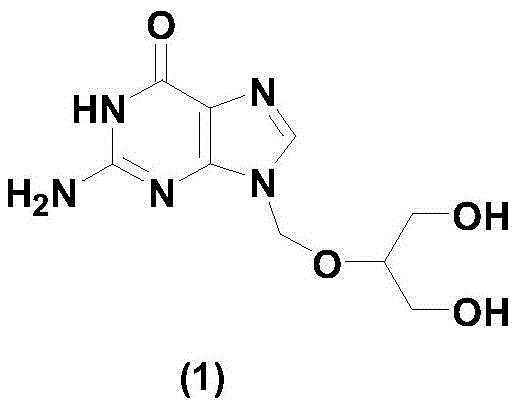
Ganciclovir, chemically known as 9-(1,3-dihydroxy-2-propoxymethyl)-guanine, is a vital therapeutic agent used globally for treating cytomegalovirus retinitis and other severe infections in immunocompromised patients. The structural complexity of this molecule necessitates precise synthetic control to ensure the final product meets pharmacopeial specifications. The innovation detailed in the patent focuses on optimizing the critical intermediate, triacetyl ganciclovir, which serves as the precursor to the final drug substance. By improving the quality of this intermediate, the entire downstream process benefits from higher yields and reduced waste, making it an attractive proposition for any reliable antiviral intermediate supplier looking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ganciclovir has been fraught with significant technical hurdles that impact both cost and scalability. Traditional routes, such as those described in European patent EP49072, rely heavily on silanization reagents and highly toxic mercury cyanide catalysts, posing severe environmental and safety risks that are increasingly unacceptable in modern regulatory landscapes. Furthermore, these legacy processes often require column chromatography to separate isomers, a technique that is notoriously difficult to scale beyond the laboratory bench and results in substantial product loss. Another prevalent method reported in US2005176955 utilizes strong acids like sulfuric acid or p-toluenesulfonic acid, which unfortunately leads to poor selectivity. In these acid-catalyzed reactions, the ratio of the desired triacetyl ganciclovir isomer (2) to the unwanted isomer (3) is often as unfavorable as 3:6, meaning the majority of the reaction mass is waste. This high isomer content necessitates complex and expensive purification steps, drastically inflating the cost of goods sold and extending lead times for high-purity pharmaceutical intermediates.
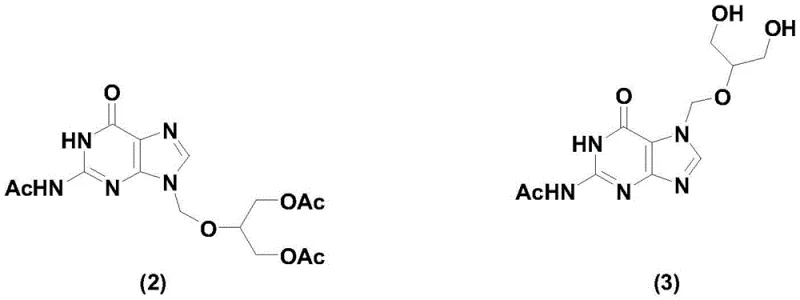
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach outlined in CN108467396B utilizes a microwave reactor to drive the condensation and hydrolysis reactions with unprecedented efficiency. This method replaces hazardous heavy metal catalysts with a boron trifluoride complex, which acts as a highly selective Lewis acid catalyst under microwave irradiation. The result is a profound improvement in reaction selectivity, where the content of the troublesome isomer (3) is reduced from the typical 30-35% range down to a mere 5-8%. This drastic reduction in impurity load means that the intermediate can be purified through simple crystallization rather than labor-intensive chromatography. Additionally, the microwave technology allows for rapid and uniform heating, which shortens the condensation reaction time from the conventional 24-48 hours down to just 10-12 hours. This acceleration not only boosts throughput but also minimizes energy consumption, aligning perfectly with the goals of cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Boron Trifluoride-Catalyzed Condensation
The core of this technological advancement lies in the synergistic effect between the boron trifluoride catalyst and microwave energy during the condensation of 2,9-diacetylguanine and 1,3-diacetoxy-2-(acetoxymethoxy)propane. Unlike conventional conductive heating, which heats the vessel walls and relies on slow thermal conduction, microwave irradiation directly couples with the polar molecules in the reaction mixture, generating heat instantaneously throughout the bulk volume. This rapid heating profile helps to overcome the activation energy barrier more efficiently, promoting the formation of the N9-glycosidic bond over the N7-isomer. The boron trifluoride catalyst, whether used as the diethyl etherate, tetrahydrofuran, or dimethyl ether complex, coordinates with the acetoxy groups of the side chain, increasing the electrophilicity of the methylene carbon. This activation facilitates a nucleophilic attack by the N9 nitrogen of the guanine base, leading to the desired triacetyl ganciclovir structure. The precision of this mechanism ensures that side reactions, such as the formation of monoacetyl guanine or degradation products, are minimized, resulting in a cleaner reaction profile.
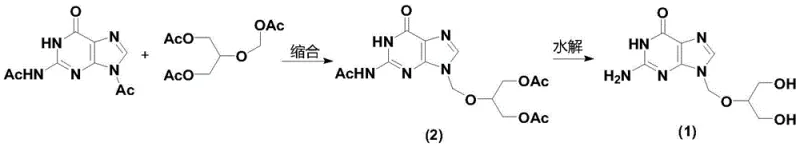
Furthermore, the mechanistic pathway includes a streamlined hydrolysis step that capitalizes on the stability of the intermediate produced. Once the triacetyl ganciclovir is formed, it undergoes hydrolysis in a microwave reactor using a mild alkaline solution, typically 5-10% potassium hydroxide. The microwave energy ensures that the deprotection of the acetyl groups occurs rapidly at temperatures between 50-70°C, completing the transformation in just 0.5 to 1 hour. This is a significant improvement over traditional hydrolysis methods that might require prolonged heating at higher temperatures, which can lead to product degradation. The combination of selective condensation and rapid, controlled hydrolysis creates a robust process window where impurity carryover is negligible. For quality control teams, this translates to a final product with purity levels consistently exceeding 99%, meeting the rigorous demands of global regulatory bodies without the need for excessive reprocessing.
How to Synthesize Triacetyl Ganciclovir Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and stoichiometric ratios to maximize the benefits of the microwave technology. The process begins with the dissolution of the guanine derivative in a polar aprotic solvent such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), which are excellent absorbers of microwave energy. The detailed standardized synthesis steps involve precise temperature ramping and holding periods to ensure complete conversion while preventing thermal runaway. Operators must adhere to strict protocols regarding the addition of the boron trifluoride catalyst and the subsequent workup procedures involving ethyl acetate crystallization. Following these optimized parameters ensures that the commercial scale-up of complex nucleoside analogs proceeds smoothly, yielding a high-quality intermediate ready for the final hydrolysis and purification stages.
- Condense 2,9-diacetylguanine with 1,3-diacetoxy-2-(acetoxymethoxy)propane using a boron trifluoride catalyst in a microwave reactor at 80-100°C.
- Hydrolyze the resulting triacetyl ganciclovir intermediate in a microwave reactor using potassium hydroxide solution at 50-70°C.
- Purify the crude ganciclovir by dissolving in DMSO or DMF, adding methanol or ethanol for crystallization, and filtering to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microwave-assisted protocol offers tangible strategic advantages that extend far beyond simple yield improvements. The elimination of toxic reagents like mercury cyanide and precious metal catalysts like palladium on carbon removes significant supply chain risks associated with the sourcing and disposal of hazardous materials. This simplification of the bill of materials leads to substantial cost savings, as the expensive and logistically complex steps of heavy metal removal and specialized waste treatment are rendered unnecessary. Moreover, the drastic reduction in reaction time—from days to hours—means that manufacturing assets can be turned over much more frequently, effectively increasing production capacity without the need for capital investment in new reactors. This enhanced asset utilization directly contributes to a more resilient supply chain capable of responding quickly to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the purification workflow. By reducing the isomer content to single-digit percentages, the need for preparative column chromatography is eliminated, which is one of the most expensive unit operations in fine chemical synthesis. This avoids the massive solvent consumption and silica gel costs associated with chromatography, leading to significantly reduced operating expenses. Additionally, the high conversion rate means that less raw material is wasted, further driving down the variable cost per kilogram of the final API. The qualitative shift from a multi-step purification nightmare to a simple crystallization process represents a fundamental optimization of the cost structure.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical antiviral intermediates is paramount, and this method enhances stability by removing dependency on scarce or regulated reagents. Traditional methods relying on mercury salts face increasing regulatory scrutiny and potential supply disruptions due to environmental bans. By switching to a boron-based catalyst system, manufacturers secure a more stable and compliant supply chain. Furthermore, the shortened production cycle time reduces the lead time for high-purity pharmaceutical intermediates, allowing suppliers to maintain lower inventory levels while still meeting delivery commitments. This agility is crucial in a post-pandemic landscape where supply chain continuity is a top priority for global health organizations.
- Scalability and Environmental Compliance: Scaling microwave chemistry from the lab to industrial production has historically been a challenge, but modern continuous flow microwave reactors have made this feasible for large-volume manufacturing. The process described is inherently scalable because it avoids the bottlenecks of batch chromatography. From an environmental perspective, the reduction in solvent usage and the elimination of heavy metal waste align with Green Chemistry principles, facilitating easier permitting and compliance with strict environmental regulations. This sustainability angle is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet ESG (Environmental, Social, and Governance) targets.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its potential for integration into existing production lines. The following questions address common concerns regarding safety, scalability, and quality control, providing clarity on how this patent differentiates itself from prior art. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring that decision-makers have access to accurate and actionable information.
Q: How does the microwave method improve isomer control compared to traditional acid catalysis?
A: Traditional methods often result in isomer ratios of 30-35%, requiring difficult chromatographic separation. The patented microwave method using boron trifluoride reduces the isomer content to 5-8%, significantly simplifying downstream purification.
Q: What are the specific reaction conditions for the condensation step?
A: The condensation is performed at 80-100°C for 10-12 hours using solvents like DMF or DMSO, with a boron trifluoride catalyst dosage of 5-50% molar weight relative to the guanine derivative.
Q: Why is this method considered more environmentally friendly?
A: This process eliminates the need for toxic mercury cyanide reagents and precious metal palladium-carbon catalysts used in older routes, while also reducing solvent consumption and energy usage through efficient microwave heating.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ganciclovir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the microwave-assisted synthesis of ganciclovir can be successfully implemented at an industrial level. Our facilities are equipped with state-of-the-art reactor systems and rigorous QC labs capable of handling complex nucleoside chemistry while maintaining stringent purity specifications. We understand that consistency is key in the pharmaceutical supply chain, and our quality management systems are designed to deliver batch-after-batch reproducibility that meets the highest international standards.
We invite potential partners to engage with our technical team to explore how this advanced manufacturing route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume requirements. We encourage you to contact our technical procurement team to discuss specific COA data and route feasibility assessments, ensuring that your project moves forward with confidence and precision. Together, we can drive the next generation of antiviral therapeutics to market faster and more efficiently.
