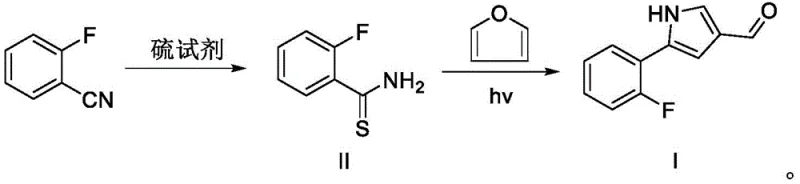
Scalable Production of Vonoprazan Fumarate Intermediate via Novel Photochemical Route
The pharmaceutical landscape for gastric acid suppression has been significantly reshaped by the introduction of Potassium-Competitive Acid Blockers (P-CABs), with Vonoprazan fumarate standing as a cornerstone molecule in this therapeutic class. As global demand for effective treatments against erosive esophagitis and Helicobacter pylori infections continues to surge, the supply chain security for its critical precursors has become a paramount concern for multinational pharmaceutical corporations. Patent CN112961092B, published in early 2023, introduces a groundbreaking preparation method for the key Vonoprazan fumarate intermediate, specifically 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde. This technical disclosure represents a paradigm shift from traditional multi-step syntheses to a streamlined, two-step process that leverages abundant starting materials and mild reaction conditions. For R&D directors and procurement strategists, this patent offers a viable pathway to de-risk the supply chain by reducing dependency on scarce noble metal catalysts and complex high-pressure equipment. The innovation lies not merely in the chemical transformation but in the holistic redesign of the manufacturing workflow to prioritize industrial feasibility, safety, and cost-efficiency without compromising the stringent purity standards required for API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for this critical pyrrole aldehyde intermediate have been plagued by significant operational inefficiencies and economic burdens that hinder large-scale commercialization. Prior art, such as the methodology described in EP2327692, relies on a lengthy sequence starting from o-fluoroacetophenone, necessitating bromination, substitution condensation, palladium-carbon dechlorination, and multiple reduction-oxidation steps. This convoluted pathway not only accumulates yield losses at every stage but also introduces expensive reagents like DIBAL-H and TPAP, which are cost-prohibitive for ton-scale production. Furthermore, alternative routes documented in WO2010098351 and CN106187852 attempt to shorten the sequence but introduce severe safety hazards or reliance on precious metal catalysts like silver and gold salts. The use of high-pressure hydrogenation with Raney nickel poses substantial safety risks in a plant environment, while the requirement for stoichiometric amounts of noble metals creates a volatile cost structure tied to fluctuating commodity markets. These conventional methods collectively suffer from poor atom economy, complex waste streams requiring extensive treatment, and rigorous operational conditions that demand specialized equipment, thereby creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the method disclosed in CN112961092B offers a remarkably concise and robust synthetic strategy that directly addresses the pain points of industrial manufacturing. The novel route initiates with the readily available and inexpensive o-fluorobenzonitrile, converting it into a thioamide intermediate through a simple nucleophilic addition with a sulfur reagent such as ammonium sulfide. This first step proceeds under mild thermal conditions in common solvents like 1,4-dioxane, achieving near-quantitative conversion without the need for exotic catalysts. The subsequent transformation involves a photochemical cyclization with furan, a reaction that elegantly constructs the pyrrole core under illumination at moderate temperatures. This approach completely bypasses the need for high-pressure hydrogenation or expensive noble metal promoters, replacing them with abundant reagents and light energy. By reducing the synthetic sequence to just two main steps from a commercial starting material, the process drastically minimizes unit operations, solvent consumption, and processing time. This streamlined workflow not only enhances the overall yield but also significantly simplifies the purification protocol, making it an ideal candidate for continuous manufacturing and large-scale batch production in a GMP-compliant environment.
Mechanistic Insights into Photochemical Pyrrole Cyclization
The core innovation of this patent lies in the mechanistic elegance of the photochemical cyclization step, which transforms the thioamide intermediate and furan into the target pyrrole aldehyde with high regioselectivity. Under illumination, typically using a mercury lamp, the thioamide undergoes a photo-induced activation that facilitates the nucleophilic attack by the electron-rich furan ring. This reaction pathway avoids the harsh acidic or basic conditions often required for traditional Paal-Knorr pyrrole synthesis, thereby preserving sensitive functional groups such as the ortho-fluorine substituent which is critical for the biological activity of the final API. The use of isopropanol as the solvent in this step is particularly advantageous, as it acts not only as a reaction medium but also potentially participates in proton transfer processes that stabilize the transition state. The reaction temperature is maintained between 30°C and 40°C, a range that is low enough to prevent thermal decomposition of the reactants yet sufficient to drive the photochemical kinetics efficiently. This mild thermal profile is a significant departure from the high-temperature cyclizations seen in other routes, reducing the energy footprint of the process and minimizing the formation of thermal byproducts that complicate downstream purification.
From an impurity control perspective, this mechanism offers superior selectivity compared to metal-catalyzed alternatives. The absence of transition metals eliminates the risk of metal leaching into the product, a critical quality attribute for pharmaceutical intermediates that must meet strict ICH guidelines for elemental impurities. The primary byproducts in this reaction are typically unreacted starting materials or simple sulfur-containing species that are easily removed during the aqueous workup and recrystallization steps described in the patent examples. The high yield of 89% reported in the optimal examples using isopropanol indicates that the reaction pathway is highly favored thermodynamically and kinetically under the specified conditions. Furthermore, the use of a nitrogen atmosphere prevents oxidative degradation of the sensitive aldehyde moiety and the pyrrole ring, ensuring that the final product maintains its structural integrity. This mechanistic robustness translates directly into a more consistent quality profile across different batches, providing supply chain managers with the confidence needed for long-term procurement planning.
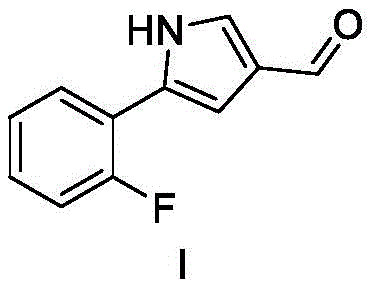
How to Synthesize 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde Efficiently
Implementing this synthesis route in a production facility requires careful attention to the specific operational parameters outlined in the patent to maximize efficiency and safety. The process begins with the preparation of the thioamide intermediate, where precise control of the molar ratio between o-fluorobenzonitrile and the sulfur reagent is essential to drive the reaction to completion while minimizing excess reagent waste. Following the isolation of the thioamide, the photochemical step demands specialized reactor equipment capable of uniform light distribution to ensure consistent reaction rates throughout the batch volume. The selection of isopropanol as the solvent for the cyclization step is critical, as experimental data within the patent demonstrates it outperforms methanol and ethanol in terms of yield and product quality. Operators must maintain the reaction temperature strictly within the 30-40°C window during irradiation to balance reaction speed with thermal stability. Detailed standardized synthesis steps see the guide below.
- React o-fluorobenzonitrile with a sulfur reagent like ammonium sulfide in 1,4-dioxane at 55°C to form the thioamide intermediate.
- Mix the thioamide intermediate with furan in isopropanol under a nitrogen atmosphere.
- Irradiate the mixture with a mercury lamp at 30-40°C for 5 hours to yield the target pyrrole aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative advantages that extend far beyond simple chemical yield improvements. The primary value proposition lies in the drastic simplification of the raw material portfolio, shifting from expensive, specialized reagents to commodity chemicals that are readily available in the global market. By eliminating the dependency on noble metals like palladium, gold, and silver, manufacturers can insulate their production costs from the volatile fluctuations of the precious metals market, ensuring more stable pricing for the final intermediate. Furthermore, the removal of high-pressure hydrogenation steps significantly reduces the capital expenditure required for plant infrastructure, as standard glass-lined or stainless steel reactors can be utilized instead of specialized high-pressure autoclaves. This reduction in equipment complexity also lowers maintenance costs and decreases the downtime associated with safety inspections and certification of pressure vessels. The overall process intensification, achieved by reducing the step count from over five steps in prior art to just two steps, leads to a substantial reduction in solvent usage and waste generation, aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the complete elimination of expensive transition metal catalysts and the reduction of unit operations. Traditional routes require costly reagents such as DIBAL-H and TPAP, which not only have high purchase prices but also require careful handling and disposal, adding to the operational overhead. By replacing these with inexpensive sulfur reagents and furan, the direct material cost is significantly lowered. Additionally, the high yield of the thioamide formation step (up to 99%) ensures that the expensive starting material, o-fluorobenzonitrile, is utilized with maximum efficiency, minimizing material loss. The simplified workup procedure, which involves standard extraction and recrystallization, reduces labor hours and utility consumption compared to the complex chromatographic purifications often needed for metal-catalyzed reactions. These factors combine to create a manufacturing process with a markedly lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of widely available starting materials and the removal of bottleneck reagents. Reagents like ammonium sulfide and furan are produced on a massive industrial scale for various chemical applications, ensuring a stable and continuous supply even during market disruptions. In contrast, specialized catalysts used in prior art routes often have limited suppliers and long lead times, creating single points of failure in the supply chain. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites without requiring extensive requalification of specialized equipment. This flexibility allows for a more diversified manufacturing network, reducing the risk of supply interruptions due to site-specific issues. The robustness of the chemistry ensures consistent batch-to-batch quality, reducing the likelihood of production failures or out-of-specification results that could delay shipments to downstream API manufacturers.
- Scalability and Environmental Compliance: The scalability of this process is supported by its inherent safety profile and reduced environmental footprint. The absence of high-pressure hydrogenation eliminates the risk of catastrophic pressure vessel failures, making the process safer to scale from pilot plant to commercial tonnage. The photochemical step, while requiring specific lighting equipment, operates at ambient pressure and moderate temperatures, simplifying the engineering controls needed for scale-up. From an environmental perspective, the reduction in step count and the use of less toxic reagents lead to a lower E-factor (mass of waste per mass of product). The avoidance of heavy metals simplifies wastewater treatment, as there is no need for complex metal scavenging or specialized disposal protocols for metal-contaminated waste streams. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental compliance costs associated with manufacturing permits and waste disposal fees.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, based on the detailed data provided in the patent documentation. These answers are designed to clarify the feasibility of the process for industrial application and to highlight the specific advantages over legacy methods. Understanding these details is crucial for technical teams evaluating the potential for technology transfer and for procurement teams assessing the long-term viability of the supply source. The information provided here reflects the specific experimental conditions and results reported in the patent, ensuring accuracy and reliability for decision-making purposes.
Q: What are the primary cost drivers eliminated in this new synthesis route?
A: This route eliminates the need for expensive noble metal catalysts such as silver trifluoromethanesulfonate or gold trichloride, and avoids high-pressure hydrogenation equipment, drastically reducing capital and operational expenditures.
Q: How does the photochemical step impact industrial scalability?
A: The photochemical cyclization operates at mild temperatures (30-40°C) and ambient pressure, removing thermal safety risks associated with high-temperature cyclization and simplifying reactor requirements for large-scale production.
Q: What is the purity profile of the intermediate produced via this method?
A: The method utilizes high-selectivity reagents and mild conditions which minimize side reactions, resulting in a crude product with high purity that simplifies downstream recrystallization and quality control processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel pharmaceutical intermediates requires more than just a patent; it demands a partner with the technical expertise to translate laboratory innovation into industrial reality. Our CDMO division possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results of CN112961092B can be realized on a global scale. We understand the critical nature of this intermediate in the Vonoprazan supply chain and are committed to maintaining stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. Our team of process chemists is adept at optimizing photochemical reactions and handling sulfur-containing intermediates safely, mitigating the technical risks associated with technology transfer. By leveraging our infrastructure, clients can accelerate their time-to-market for P-CAB inhibitors while ensuring a secure and compliant supply of high-quality intermediates.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this novel synthesis can optimize your supply chain economics. Whether you require development support to finalize the process parameters or immediate commercial supply to meet market demand, NINGBO INNO PHARMCHEM is equipped to deliver solutions that align with your strategic goals. Contact us today to discuss how we can support your Vonoprazan projects with reliability and excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
