Advanced Manufacturing of Vonoprazan Intermediates via Optimized Catalytic Routes
Advanced Manufacturing of Vonoprazan Intermediates via Optimized Catalytic Routes
The pharmaceutical landscape for gastroesophageal reflux disease (GERD) treatment has been significantly reshaped by the introduction of potassium-competitive acid blockers (P-CABs), with Vonoprazan fumarate standing out as a premier therapeutic agent. The efficient production of its key building block, 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde, remains a critical bottleneck for generic manufacturers seeking market entry. Patent CN112194607A discloses a robust and innovative synthetic methodology that addresses these challenges through a streamlined five-step sequence. This technical insight report analyzes the proprietary process detailed in the patent, highlighting its potential to redefine supply chain reliability for this high-value pharmaceutical intermediate. By leveraging specific catalytic systems and optimized reaction conditions, this route offers a compelling alternative to traditional methods that often suffer from harsh conditions or poor impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole-based intermediates for acid-suppressing drugs has relied on methodologies that introduce significant operational hazards and purification complexities. Traditional routes often employ elemental bromine or other aggressive halogenating agents that require specialized corrosion-resistant equipment and rigorous safety protocols to manage toxic vapors. Furthermore, conventional cyclization strategies frequently result in the formation of stubborn chlorinated by-products that are difficult to separate, leading to reduced overall yields and compromised purity levels in the final active pharmaceutical ingredient. These legacy processes often necessitate multiple recrystallization steps or chromatographic purifications, which drastically increase manufacturing costs and extend production lead times. The accumulation of heavy metal residues from non-optimized catalysts also poses a risk for failing strict regulatory limits, creating a substantial burden for quality control departments in large-scale facilities.
The Novel Approach
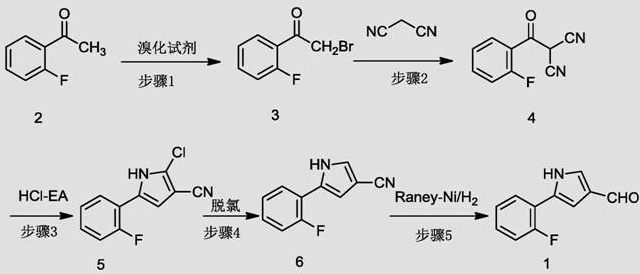
The methodology outlined in patent CN112194607A introduces a paradigm shift by utilizing tetrabutylammonium tribromide (TBATB) as a solid, safer brominating reagent, thereby eliminating the handling risks associated with liquid bromine. This innovative approach facilitates a smoother transition through the initial functionalization of 2-fluoroacetophenone, setting the stage for a highly efficient condensation with malononitrile. The subsequent cyclization in a hydrogen chloride-ethyl acetate system is carefully controlled at moderate temperatures, minimizing side reactions while maximizing the formation of the desired pyrrole core. Crucially, the process incorporates a dedicated catalytic dechlorination step using palladium on carbon, which effectively scrubs chlorine impurities before the final reduction. This strategic sequence ensures that the final aldehyde product is obtained with superior purity and in high yield, directly addressing the痛点 of previous synthetic limitations.
Mechanistic Insights into TBATB-Mediated Bromination and Catalytic Reduction
The cornerstone of this synthetic success lies in the precise control of electrophilic substitution and subsequent catalytic hydrogenation mechanisms. In the initial step, TBATB serves as a source of electrophilic bromine, reacting with the enolizable ketone 2-fluoroacetophenone in tetrahydrofuran (THF) to selectively form the alpha-bromo ketone. This solid reagent allows for a more controlled release of bromine species compared to molecular bromine, reducing the likelihood of poly-bromination and ensuring high regioselectivity. Following the condensation with malononitrile, the resulting dicyano intermediate undergoes an acid-catalyzed Paal-Knorr-type cyclization. The use of HCl in ethyl acetate provides the necessary acidic environment to promote ring closure while maintaining solubility, leading to the formation of the 2-chloro-pyrrole-3-carbonitrile scaffold with high efficiency.
Impurity control is meticulously managed through the sequential application of heterogeneous catalysis in the later stages of the synthesis. The presence of a chlorine atom at the 2-position of the pyrrole ring, introduced during cyclization, is strategically removed via hydrogenolysis using 5% Pd-C under a hydrogen atmosphere. This dechlorination step is vital for preventing the carryover of genotoxic impurities into the final drug substance. Subsequently, the nitrile group is reduced to the corresponding aldehyde using Raney-Nickel, a cost-effective catalyst that offers excellent chemoselectivity. This reduction avoids the over-reduction to the alcohol or amine, which are common side reactions in less optimized protocols. The combination of these catalytic steps ensures a clean reaction profile, significantly simplifying downstream processing and isolation.
How to Synthesize 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to achieve the reported yields and purity levels. The process begins with the dissolution of 2-fluoroacetophenone in THF, followed by the controlled addition of TBATB at ambient temperatures to ensure complete conversion to the bromo-ketone. The subsequent condensation with malononitrile is performed in the presence of sodium carbonate, which acts as a mild base to facilitate the nucleophilic attack without degrading the sensitive intermediates. For the full standardized operating procedure, including exact stoichiometric ratios, work-up details, and crystallization protocols, please refer to the technical guide below.
- Brominate 2-fluoroacetophenone using tetrabutylammonium tribromide (TBATB) in THF to generate 2-fluoro-alpha-bromoacetophenone.
- Condense the brominated intermediate with malononitrile using sodium carbonate catalysis to form the dicarbonyl precursor.
- Perform acid-catalyzed cyclization in HCl-ethyl acetate, followed by Pd-C catalyzed dechlorination and Raney-Ni hydrogenation to yield the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic benefits for procurement managers and supply chain directors aiming to optimize their vendor portfolios. The shift towards safer, solid-state reagents like TBATB reduces the logistical complexity and insurance costs associated with transporting hazardous liquids, thereby enhancing overall supply chain resilience. Moreover, the high yields reported in each step, particularly the 91% yield in the dechlorination stage and 76.9% in the final reduction, translate directly into improved material efficiency and reduced waste generation. This efficiency is critical for maintaining competitive pricing structures in the generic pharmaceutical market, where margin compression is a constant challenge for manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the use of cost-effective catalysts like Raney-Nickel, drives down the raw material costs significantly. By avoiding complex purification steps such as column chromatography and relying instead on straightforward crystallization and filtration, the process reduces solvent consumption and labor hours. This streamlined workflow results in a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-fluoroacetophenone and malononitrile ensures a stable supply base that is less susceptible to market volatility. The robustness of the reaction conditions, which operate at mild temperatures between 20°C and 55°C, minimizes the risk of batch failures due to thermal runaway or equipment malfunction. This operational stability guarantees consistent delivery schedules, enabling pharmaceutical companies to plan their production cycles with greater confidence and reduce safety stock requirements.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common organic solvents like THF and ethyl acetate that are easily recovered and recycled in industrial settings. The reduction in hazardous waste generation, particularly through the avoidance of liquid bromine and the efficient removal of chlorine impurities, aligns with increasingly stringent environmental regulations. This green chemistry approach not only mitigates regulatory risks but also enhances the corporate sustainability profile of the manufacturing entity, a key factor for modern procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this specific pyrrole intermediate. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own supply chains.
Q: What are the key advantages of using TBATB over traditional brominating agents?
A: The use of tetrabutylammonium tribromide (TBATB) offers significant operational safety benefits compared to liquid bromine, while maintaining high regioselectivity and yield in the initial functionalization step.
Q: How does this process ensure low levels of chlorinated impurities?
A: The inclusion of a dedicated catalytic dechlorination step using 5% Pd-C effectively removes chlorine atoms introduced during cyclization, ensuring the final API intermediate meets stringent purity specifications.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes mild reaction temperatures (20-55°C) and common solvents like THF and ethyl acetate, making it highly suitable for large-scale commercial manufacturing without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications like Vonoprazan. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trials and full-scale market launch. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde meets the highest international standards for pharmaceutical applications.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your project timelines and quality objectives.
