Scalable Manufacturing of Fibrinogen Receptor Antagonist Intermediates Using Advanced Mitsunobu Chemistry
Scalable Manufacturing of Fibrinogen Receptor Antagonist Intermediates Using Advanced Mitsunobu Chemistry
The pharmaceutical industry constantly seeks robust synthetic pathways that balance efficiency, safety, and scalability, particularly for complex cardiovascular therapeutics like fibrinogen receptor antagonists. Patent CN1040534C introduces a transformative approach to synthesizing these critical bioactive molecules, specifically targeting the efficient construction of tyrosine-derived intermediates bearing heterocyclic side chains. This intellectual property outlines a sophisticated methodology that replaces legacy, hazard-prone alkylation techniques with a streamlined Mitsunobu etherification strategy. By leveraging the latent reactivity of 4-picoline and employing precise catalytic hydrogenation, this technology enables the production of high-purity intermediates essential for antithrombotic drug development. For global procurement teams and R&D directors, understanding this shift is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality while mitigating operational risks associated with dangerous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ether-linked tyrosine derivatives relied heavily on classical Williamson ether synthesis conditions, which often necessitated the use of strong bases like sodium hydride (NaH) in polar aprotic solvents such as dimethylformamide (DMF). As illustrated in the prior art reaction scheme, this traditional eleven-step process was not only lengthy but also fraught with significant safety hazards due to the pyrophoric nature of sodium hydride and the potential for exothermic runaway reactions. Furthermore, the crude products generated from these harsh conditions typically contained complex impurity profiles that could not be easily removed by simple crystallization. Consequently, manufacturers were forced to rely on resource-intensive chromatographic purification steps to achieve the necessary purity levels, drastically increasing production costs and extending lead times for high-purity pharmaceutical intermediates. These inefficiencies created substantial bottlenecks in the supply chain, making it difficult to scale production to meet commercial demand without compromising safety or economic viability.
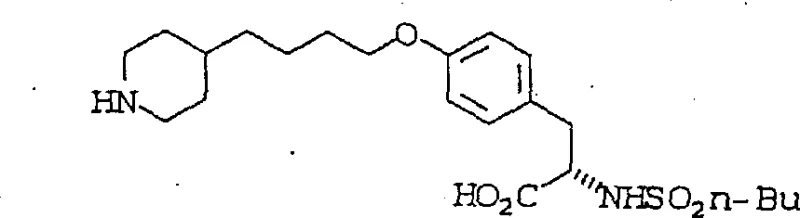
The Novel Approach
In stark contrast to the cumbersome legacy methods, the present invention delineated in the patent data offers a highly efficient six-step synthesis that fundamentally reimagines the construction of the ether linkage. By utilizing 4-picoline as a latent form of piperidine, the process cleverly bypasses the need for protecting group manipulations that typically add steps and reduce overall yield. The core innovation lies in the application of the Mitsunobu reaction for O-alkylation, which proceeds under mild, neutral conditions to couple the tyrosine derivative with the pyridyl-butanol side chain. This strategic shift allows for the removal of phosphine oxide byproducts through straightforward extraction and recrystallization techniques, completely eliminating the need for column chromatography. The result is a robust manufacturing route that delivers the coupled product in high yield and exceptional purity, setting a new standard for cost reduction in API manufacturing and providing a scalable solution for complex organic synthesis.
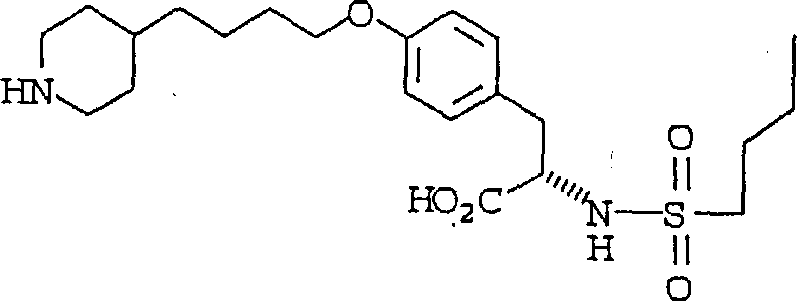
Mechanistic Insights into Mitsunobu Etherification and Selective Hydrogenation
The chemical elegance of this process is rooted in the precise control of reactivity during the etherification and reduction stages. The synthesis begins with the metalation of 4-picoline using n-butyllithium at cryogenic temperatures, generating a nucleophilic species that attacks a protected bromo-alcohol to establish the carbon chain. Following deprotection, the resulting 4-(4-pyridyl)butanol serves as the nucleophile in the Mitsunobu reaction. Mechanistically, this involves the activation of the alcohol by triphenylphosphine and diisopropyl azodicarboxylate (DIAD), creating an oxyphosphonium intermediate that is displaced by the phenolic oxygen of the tyrosine derivative with inversion of configuration, although stereochemistry is preserved at the tyrosine alpha-carbon. This mechanism is superior to base-mediated alkylation because it avoids the formation of reactive enolates that can lead to racemization or side reactions. Subsequently, the pyridine ring is selectively reduced to a piperidine ring using palladium on carbon (Pd/C) in acetic acid. This catalytic hydrogenation is carefully controlled at 70°C to ensure complete saturation of the heterocycle without reducing the carboxylic acid or sulfonamide moieties, demonstrating a high level of chemoselectivity crucial for maintaining the integrity of the final active pharmaceutical ingredient.
Impurity control is another critical aspect where this mechanistic approach excels, particularly regarding the removal of triphenylphosphine oxide (Ph3PO), a stoichiometric byproduct of the Mitsunobu reaction. In many syntheses, Ph3PO is notoriously difficult to separate from the desired product, often requiring extensive chromatography. However, this patent describes a specialized workup procedure where the reaction mixture is treated with hexane and dichloromethane, causing the Ph3PO to precipitate or remain in specific solvent phases that can be easily filtered off using silica gel pads. Furthermore, the final product is purified through a series of acid-base extractions and recrystallizations from isopropanol, which effectively scrubs away any remaining organic impurities and residual metals from the hydrogenation catalyst. This rigorous purification protocol ensures that the final fibrinogen receptor antagonist intermediate meets stringent purity specifications, with HPLC assays confirming purity levels exceeding 99.5%, thereby minimizing the risk of toxicological issues in downstream drug formulation.
How to Synthesize N-n-butanesulfonyl-O-(4-(4-piperidinyl)butyl)-(L)-tyrosine Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters, particularly temperature control during the lithiation and hydrogenation steps to ensure safety and reproducibility. The process flow involves preparing the sulfonamide-protected tyrosine ester separately, synthesizing the pyridyl-butanol linker, coupling them via the Mitsunobu protocol, hydrolyzing the ester, and finally reducing the pyridine ring. Each step has been optimized to maximize yield while minimizing waste, making it an ideal candidate for technology transfer to commercial manufacturing sites. For technical teams looking to implement this route, the detailed standardized synthesis steps provided in the patent documentation serve as an essential guide for replicating these results at scale. The following section outlines the critical operational phases required to achieve the high-quality outcomes described in the intellectual property.
- Preparation of the alcohol side chain via lithiation of 4-picoline followed by quenching with a protected bromo-alcohol.
- Mitsunobu coupling of the resulting alcohol with N-sulfonyl tyrosine methyl ester using triphenylphosphine and DIAD.
- Hydrolysis of the methyl ester followed by selective catalytic hydrogenation of the pyridine ring to form the piperidine moiety.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring continuity of supply. The elimination of hazardous reagents like sodium hydride significantly reduces the safety infrastructure required at manufacturing plants, lowering insurance premiums and regulatory compliance costs associated with handling pyrophoric materials. Moreover, the replacement of chromatographic purification with crystallization and filtration represents a massive leap in processing efficiency, as chromatography is often the rate-limiting step in fine chemical production due to its low throughput and high solvent consumption. By simplifying the purification train, manufacturers can drastically increase batch sizes and reduce cycle times, leading to substantial cost savings in solvent procurement and waste disposal. This efficiency translates directly into a more competitive pricing structure for the final intermediate, allowing pharmaceutical companies to manage their raw material budgets more effectively while securing a reliable source of critical starting materials.
- Cost Reduction in Manufacturing: The transition to a Mitsunobu-based strategy eliminates the need for expensive and hazardous base-mediated alkylation conditions, which often require specialized equipment and rigorous safety protocols. By utilizing readily available reagents like triphenylphosphine and DIAD, and avoiding the high costs associated with disposing of sodium hydride residues, the overall cost of goods sold is significantly optimized. Additionally, the ability to purify the product through crystallization rather than chromatography reduces solvent usage and processing time, further driving down manufacturing expenses and enhancing the economic feasibility of large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 4-picoline and standard amino acid derivatives ensures that the raw material supply chain is robust and less susceptible to market volatility. Unlike specialized reagents that may have limited suppliers, the inputs for this process are widely available from multiple global vendors, reducing the risk of supply disruptions. Furthermore, the simplified process flow with fewer unit operations means that production can be scaled up more rapidly to meet surges in demand, ensuring that downstream drug manufacturers receive their intermediates on schedule without facing delays caused by complex purification bottlenecks.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with example batches demonstrated in multi-liter reactors that translate well to industrial tank sizes. The reduction in hazardous waste generation, particularly the avoidance of heavy metal contamination from alternative catalytic methods and the minimization of solvent waste from chromatography, aligns with modern green chemistry principles. This environmental stewardship not only simplifies regulatory approvals but also enhances the corporate sustainability profile of the supply chain, making it an attractive option for pharmaceutical companies committed to reducing their carbon footprint and adhering to strict environmental, social, and governance (ESG) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims within the patent, providing clarity on how this method compares to industry standards. Understanding these nuances is essential for technical procurement teams evaluating potential suppliers or considering in-house production of this key intermediate. The answers highlight the specific advantages in terms of safety, purity, and operational simplicity that define this innovative approach.
Q: How does this new method improve safety compared to traditional alkylation?
A: The novel process eliminates the use of hazardous sodium hydride (NaH) in DMF, replacing it with a Mitsunobu reaction that operates under milder conditions and avoids the generation of hydrogen gas.
Q: What purification methods are used to ensure high purity without chromatography?
A: The process utilizes crystallization and extraction techniques, specifically removing triphenylphosphine oxide byproducts through filtration and washing, thereby avoiding expensive and slow column chromatography.
Q: Is the pyridine to piperidine reduction selective?
A: Yes, the method employs Pd/C in acetic acid at controlled temperatures (70°C) to achieve selective hydrogenation of the pyridine ring without affecting other sensitive functional groups in the molecule.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fibrinogen Receptor Antagonist Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to drive innovation in cardiovascular therapeutics. Our team of expert chemists has extensively analyzed the technological breakthroughs presented in patent CN1040534C and possesses the capability to implement this efficient Mitsunobu-based route at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify that every batch meets stringent purity specifications, including chiral integrity and residual solvent limits, guaranteeing the safety and efficacy of your final drug product.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and safe manufacturing platform for your fibrinogen receptor antagonist programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable, high-performance chemical solutions.
