Advanced Chiral Synthesis of Cinacalcet Hydrochloride for Commercial API Manufacturing
Advanced Chiral Synthesis of Cinacalcet Hydrochloride for Commercial API Manufacturing
The pharmaceutical landscape for treating secondary hyperparathyroidism and parathyroid carcinoma has been significantly shaped by the introduction of Cinacalcet Hydrochloride, a potent calcimimetic agent. As global demand for this critical medication rises, the imperative for robust, scalable, and environmentally sustainable manufacturing processes becomes paramount for supply chain stability. Patent CN101993379B discloses a groundbreaking preparation method that fundamentally reimagines the synthetic architecture of this molecule. By shifting away from traditional reductive amination and racemic resolution strategies, this technology introduces a sophisticated chiral auxiliary approach starting from the commodity chemical 1-acetonaphthone. This report provides a deep technical and commercial analysis of this novel pathway, highlighting its potential to redefine cost structures and supply reliability for reliable cinacalcet hydrochloride supplier networks globally.
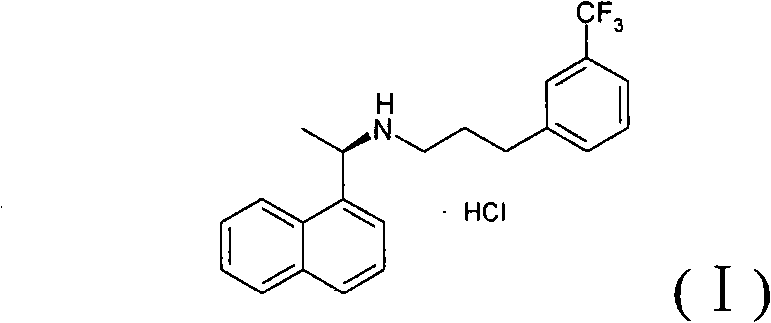
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cinacalcet has been plagued by significant chemical and operational inefficiencies that hinder cost reduction in API manufacturing. Early methodologies, such as those referenced in USP 6,211,244, relied heavily on reductive amination between naphthylethylamine and trifluoromethyl-phenyl-propionaldehyde. A critical flaw in this legacy approach is the dependence on sodium cyanoborohydride as the reducing agent. This reagent is not only notoriously toxic, generating hazardous cyanide byproducts that complicate waste stream management, but it also suffers from poor selectivity, leading to serious side reactions and diminished overall yields. Furthermore, alternative pathways described in literature often involve the resolution of racemic mixtures, a process inherently limited by a maximum theoretical yield of 50% for the desired enantiomer. These resolution techniques frequently necessitate the use of expensive chiral acids and multiple recrystallization steps, inflating both raw material consumption and processing time, thereby creating bottlenecks for commercial scale-up of complex pharmaceutical intermediates.
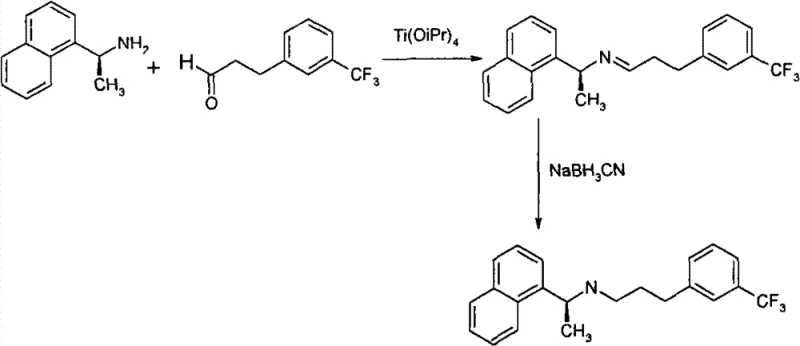
The Novel Approach
In stark contrast to these legacy constraints, the methodology outlined in Patent CN101993379B presents a streamlined, four-step sequence that prioritizes atom economy and stereochemical control from the outset. The innovation lies in the early introduction of chirality using a chiral sulfonamide auxiliary rather than attempting to separate enantiomers at the end of the synthesis. This route begins with the condensation of 1-acetonaphthone with a chiral reagent, mediated by titanium tetraisopropoxide, followed by a highly selective reduction. This strategic pivot eliminates the need for toxic cyanoborohydrides entirely, replacing them with the safer and more manageable sodium borohydride. Subsequent steps involve a precise propargylation and a palladium-catalyzed cross-coupling reaction to install the trifluoromethylphenyl group, followed by a final hydrogenation. This logical progression not only simplifies the purification profile but also ensures that the chiral integrity is maintained throughout the synthesis, offering a superior alternative for producing high-purity pharmaceutical intermediates with minimal environmental footprint.
Mechanistic Insights into Titanium-Mediated Asymmetric Reduction and Pd-Coupling
The cornerstone of this synthetic breakthrough is the first step, which establishes the critical stereocenter adjacent to the naphthalene ring. The mechanism involves the formation of a chiral titanium-imine complex. When 1-acetonaphthone reacts with a chiral sulfonamide (such as tert-butyl sulfonamide) in the presence of titanium tetraisopropoxide, a rigid cyclic transition state is formed. This coordination effectively shields one face of the imine double bond, directing the subsequent hydride attack from sodium borohydride to occur exclusively from the less hindered face. This asymmetric induction is crucial, as it generates the (R)-configured amine intermediate with high enantiomeric excess, bypassing the need for downstream resolution. The use of titanium as a Lewis acid catalyst is particularly advantageous because it is inexpensive, readily available, and facilitates the reaction under mild thermal conditions (60~80 ℃), ensuring energy efficiency and operational safety during the most critical stereochemistry-defining step of the entire process.
Following the establishment of chirality, the synthesis proceeds through a robust carbon-carbon bond-forming sequence. The intermediate chiral amine is alkylated with a propargyl halide to introduce an alkyne handle, which serves as a versatile synthon for the subsequent coupling reaction. The third step utilizes a palladium-catalyzed cross-coupling, likely a Sonogashira-type reaction, between the terminal alkyne and a meta-halogenated benzotrifluoride (such as m-iodobenzotrifluoride). This transition metal-catalyzed step is highly efficient, tolerating the trifluoromethyl group and the bulky naphthalene moiety without significant degradation. The final step involves the catalytic hydrogenation of the triple bond to a saturated propyl chain using Pd/C under hydrogen pressure. This reduction is chemoselective, leaving the aromatic rings and the chiral amine center intact while fully saturating the linker chain. The entire mechanistic pathway is designed to minimize side products, resulting in a cleaner reaction profile that simplifies isolation and enhances the overall purity of the final Cinacalcet Hydrochloride product.
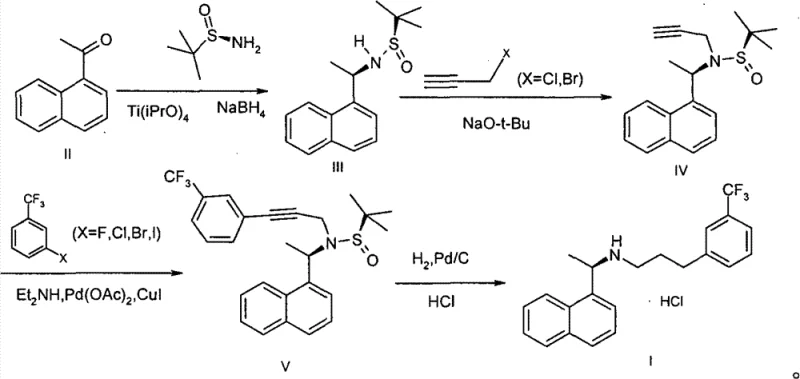
How to Synthesize Cinacalcet Hydrochloride Efficiently
The execution of this patented process requires precise control over reaction parameters to maximize the benefits of the chiral auxiliary strategy. The synthesis is divided into four distinct operational units: asymmetric reduction, N-alkylation, cross-coupling, and final hydrogenation. Each step has been optimized to balance reaction kinetics with product stability, ensuring that the chiral information installed in the first step is preserved through to the final salt formation. The use of common solvents like THF, MTBE, and ethanol further enhances the practicality of this route, allowing for straightforward solvent recovery and recycling protocols. For detailed operational parameters including specific molar ratios, temperature ramps, and workup procedures, please refer to the standardized synthesis guide below.
- Condense 1-acetonaphthone with a chiral sulfonamide using Ti(OiPr)4, followed by stereoselective reduction with NaBH4 to form the chiral amine intermediate.
- Alkylate the chiral amine intermediate with 3-propargyl halide under basic conditions to introduce the alkyne functionality.
- Perform a palladium-catalyzed cross-coupling reaction between the alkyne intermediate and meta-halogenated benzotrifluoride to construct the carbon skeleton.
- Execute catalytic hydrogenation to reduce the triple bond to a single bond, followed by acidification with hydrochloric acid to yield the final salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates directly into tangible strategic advantages regarding cost, risk, and continuity. The elimination of sodium cyanoborohydride removes a major regulatory and safety hurdle, significantly reducing the costs associated with hazardous waste disposal and specialized handling equipment. Furthermore, by avoiding racemic resolution, the process theoretically doubles the yield of the chiral intermediate compared to resolution-based methods, effectively halving the raw material burden for the naphthalene precursor. This improvement in atom economy drives down the Cost of Goods Sold (COGS) and insulates the supply chain from volatility in raw material pricing. The reliance on commodity chemicals like 1-acetonaphthone and standard catalysts ensures that the supply base is broad and resilient, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of single-source supplier bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and toxic reagents. Traditional routes often require stoichiometric amounts of costly chiral resolving agents or hazardous reducing agents that demand expensive quenching and disposal protocols. By utilizing a catalytic amount of titanium and a recyclable chiral auxiliary strategy, the variable costs per kilogram are drastically simplified. Additionally, the high selectivity of the palladium coupling step minimizes the formation of difficult-to-remove impurities, reducing the number of purification cycles and solvent consumption required to meet pharmacopeial standards. This leaner manufacturing approach allows for significant margin improvement without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-proprietary starting materials. 1-acetonaphthone is a widely produced industrial chemical with a stable global supply, unlike specialized chiral pool materials that may suffer from agricultural or fermentation-based supply shocks. The process conditions are also moderate, avoiding extreme cryogenic temperatures or high-pressure regimes that require specialized reactor infrastructure. This accessibility means that multiple qualified manufacturing sites can potentially adopt this technology, creating a redundant supply network that ensures continuous availability of Cinacalcet HCl for downstream formulation partners, even in the face of regional disruptions.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route represents a significant advancement towards green chemistry principles. The replacement of cyanide-based reductants with borohydrides eliminates the generation of cyanide-containing wastewater, a major liability in modern chemical manufacturing. The process generates fewer byproducts, leading to a lower E-factor (mass of waste per mass of product). This reduced environmental burden simplifies the permitting process for new manufacturing lines and aligns with the increasingly stringent sustainability mandates of global pharmaceutical buyers. The scalability is proven by the straightforward nature of the unit operations—filtration, extraction, and crystallization—which are easily transferred from pilot plant to multi-ton commercial production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. These insights are derived directly from the experimental data and comparative analysis provided in Patent CN101993379B, offering clarity on how this method outperforms legacy technologies in terms of safety, yield, and operational simplicity. Understanding these nuances is essential for technical teams evaluating technology transfer opportunities.
Q: How does this novel route improve safety compared to traditional reductive amination?
A: Traditional methods often utilize sodium cyanoborohydride, a highly toxic reagent posing significant EHS risks. This patented process replaces it with sodium borohydride in a titanium-mediated asymmetric reduction, drastically reducing toxicity and waste treatment costs while maintaining high stereoselectivity.
Q: What is the optical purity advantage of this chiral auxiliary method?
A: Unlike resolution methods which theoretically cap yield at 50% and require multiple crystallizations, this route employs a chiral sulfonamide auxiliary to induce asymmetry directly. This ensures high enantiomeric excess from the initial steps, avoiding the material loss associated with racemic splitting.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available commodity chemicals like 1-acetonaphthone and avoids expensive or hazardous coupling reagents like HOBt and DCC. The operational simplicity and robust yields across the four steps make it highly amenable to commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereochemical controls of this chiral synthesis are maintained at every batch size. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every gram of Cinacalcet Hydrochloride meets the highest international regulatory standards. Our facility is designed to handle complex organic synthesis safely, with dedicated containment systems for handling reactive intermediates and efficient waste treatment protocols that align with the green chemistry benefits of this novel route.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your procurement budget. We encourage interested parties to contact our technical procurement team to request specific COA data, route feasibility assessments, and sample availability. Let us help you secure a sustainable, high-quality supply of this critical API intermediate, driving value and reliability for your end-product formulations.
