Advanced Manufacturing of Tebipenem Pivoxil: A Technical Breakthrough in Carbapenem Prodrug Synthesis
Advanced Manufacturing of Tebipenem Pivoxil: A Technical Breakthrough in Carbapenem Prodrug Synthesis
The landscape of carbapenem antibiotic manufacturing is undergoing a significant transformation driven by the need for more efficient, scalable, and cost-effective synthetic routes. Patent CN102702204A introduces a pivotal advancement in the preparation of Tebipenem Pivoxil, a critical oral carbapenem prodrug known for its efficacy against pediatric respiratory infections. This intellectual property outlines a novel esterification strategy that replaces traditional, labor-intensive purification methods with a streamlined catalytic process. By leveraging a specific iodide salt catalyst system, the technology addresses long-standing challenges in yield optimization and impurity control. For R&D directors and procurement strategists, this represents a tangible opportunity to enhance the supply chain resilience of high-value beta-lactam intermediates. The following analysis dissects the technical merits of this approach, highlighting its potential to redefine production standards for reliable pharmaceutical intermediate suppliers globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tebipenem Pivoxil has been plagued by inefficiencies inherent in third-generation esterification protocols. Traditional methodologies typically rely on phase-transfer catalysts to drive the reaction between the tebibpenem acid precursor and the pivaloyloxymethyl halide. These legacy processes necessitate elevated reaction temperatures, often around 45°C, which inadvertently promote thermal degradation of the sensitive beta-lactam ring. Furthermore, the reaction kinetics are sluggish, requiring extended durations exceeding four hours to reach completion. Perhaps most critically from a manufacturing standpoint, the crude product obtained from these conventional routes is heavily contaminated with side products, mandating the use of column chromatography for purification. This reliance on chromatographic separation is a severe bottleneck for commercial scale-up, as it drastically limits throughput, increases solvent consumption, and results in suboptimal overall yields, historically hovering around a mere 30% for the final esterification step.
The Novel Approach
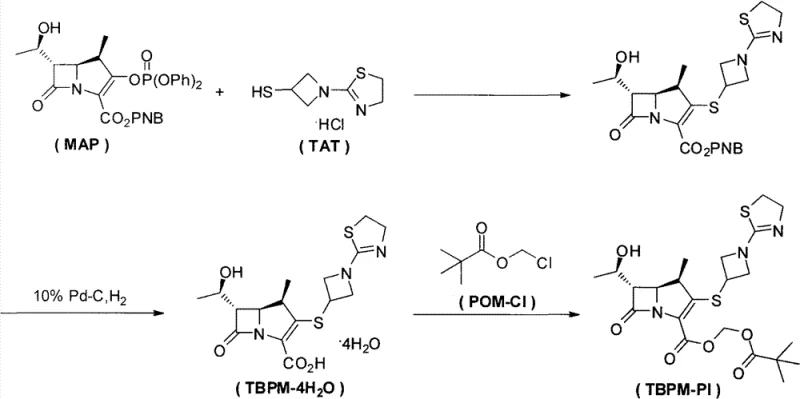
In stark contrast, the methodology disclosed in CN102702204A revolutionizes this transformation by introducing a catalytic amount of an iodide salt, such as potassium iodide or sodium iodide, into the reaction matrix. This innovation allows the esterification to proceed under mild cryogenic conditions, specifically maintained at 0°C using an ice-water bath. The introduction of the iodide species fundamentally alters the reaction pathway, likely through the in-situ generation of a more reactive iodomethyl pivalate intermediate, which undergoes rapid nucleophilic attack by the carboxylate anion. This mechanistic shift not only accelerates the reaction rate but also suppresses the formation of thermal impurities. Consequently, the post-reaction workup is dramatically simplified; the need for column chromatography is entirely eliminated in favor of a robust acid-base extraction followed by crystallization. This transition from chromatographic purification to crystallization is a hallmark of process intensification, enabling yields to surge to the 60%-65% range while achieving exceptional purity profiles of 99.5% to 99.9%.
Mechanistic Insights into Iodide-Catalyzed Esterification
The core of this technological leap lies in the nuanced role of the iodide ion as a nucleophilic catalyst within the esterification mechanism. In the absence of the iodide salt, the direct displacement of chloride from chloromethyl pivalate (POM-Cl) by the sterically hindered carboxylate of Tebipenem is kinetically unfavorable, necessitating harsh thermal conditions. However, the iodide ion, being a superior nucleophile and leaving group compared to chloride, rapidly attacks the chloromethyl pivalate to generate an iodomethyl pivalate species in situ. This transient intermediate is significantly more electrophilic, thereby lowering the activation energy for the subsequent attack by the Tebipenem carboxylate anion. This catalytic cycle ensures that the reaction proceeds efficiently even at 0°C, preserving the integrity of the fragile carbapenem core which is prone to hydrolysis and epimerization at higher temperatures. The precise control of stoichiometry, utilizing a mass ratio of Tebipenem tetrahydrate to iodide salt between 5:1 and 20:1, is critical to maintaining this catalytic efficiency without introducing excessive inorganic residues.
Furthermore, the impurity profile of the final product is intrinsically linked to the mildness of the reaction conditions facilitated by this catalytic system. Beta-lactam antibiotics are notoriously sensitive to thermal stress and pH fluctuations. By conducting the reaction at 0°C and utilizing diisopropylethylamine (DIPEA) as a non-nucleophilic base, the process minimizes the risk of beta-lactam ring opening and racemization at the C-1 position. The workup procedure further refines the quality by employing a strategic pH swing; initial acidification to pH 4.0 removes basic impurities and unreacted amines into the aqueous phase, while subsequent basification to pH 7.6 with potassium bicarbonate ensures the selective extraction of the product into the organic phase. This rigorous control over the chemical environment ensures that the final crystalline solid meets the stringent specifications required for oral administration, effectively mitigating the risks associated with genotoxic impurities or residual solvents often found in less optimized processes.
How to Synthesize Tebipenem Pivoxil Efficiently
The practical implementation of this synthesis route requires strict adherence to temperature control and reagent addition sequences to maximize the benefits of the iodide catalysis. The process begins with the dissolution of the starting material, Tebipenem tetrahydrate, and the catalytic iodide salt in a polar aprotic solvent such as THF, followed by cooling to 0°C. The subsequent addition of the base and the acylating agent must be performed slowly to manage the exotherm and maintain the low-temperature regime essential for high selectivity. Following the reaction period of 2 to 6 hours, the solvent is removed, and the residue is subjected to a liquid-liquid extraction protocol involving ethyl acetate, citric acid, and potassium bicarbonate solutions.
- Dissolve Tebipenem tetrahydrate and a catalytic amount of iodide salt (e.g., Potassium Iodide) in THF and cool the mixture to 0°C.
- Slowly add Diisopropylethylamine and Chloromethyl pivalate while maintaining the temperature below 0°C, stirring for 2-6 hours.
- Remove solvent, dissolve residue in Ethyl Acetate, perform acid-base extraction workup, and crystallize the product from concentrated solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodide-catalyzed process translates directly into substantial operational efficiencies and cost optimizations. The elimination of column chromatography is perhaps the most significant economic driver, as chromatographic steps are notoriously expensive due to high solvent usage, silica gel costs, and low throughput. By replacing this with a crystallization-based purification, the manufacturing cycle time is drastically reduced, allowing for faster batch turnover and improved asset utilization. This simplification also reduces the environmental footprint of the process by minimizing hazardous waste generation, aligning with increasingly strict global environmental regulations. Furthermore, the doubling of the reaction yield from historical averages of 30% to over 60% implies a massive reduction in the cost of goods sold (COGS), as the consumption of the valuable Tebipenem acid precursor is effectively halved per kilogram of final API produced.
- Cost Reduction in Manufacturing: The primary economic advantage stems from the dramatic improvement in reaction yield and the removal of expensive purification steps. By avoiding column chromatography, manufacturers save significantly on consumables like silica gel and the large volumes of organic solvents required for elution. Additionally, the higher yield means that less raw material is wasted, directly lowering the material cost per unit of output. The use of inexpensive, commodity-grade catalysts like potassium iodide further contributes to a leaner cost structure compared to proprietary phase-transfer catalysts.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain stability by reducing the complexity of the manufacturing process. Simpler processes with fewer unit operations are less prone to operational failures and batch-to-batch variability. The ability to produce high-purity material through crystallization rather than chromatography ensures a more consistent supply of quality product, reducing the risk of stockouts caused by failed purification runs. Moreover, the reagents involved, such as THF, DIPEA, and POM-Cl, are widely available commodity chemicals, mitigating the risk of raw material shortages.
- Scalability and Environmental Compliance: From a scale-up perspective, the transition to a crystallization-based workup is a critical enabler for commercial production. Chromatography is difficult to scale beyond pilot plant levels, whereas crystallization is a standard, easily scalable unit operation in the fine chemical industry. This scalability ensures that the process can meet growing market demand for pediatric antibiotics without requiring disproportionate capital investment. Additionally, the reduced solvent consumption and waste generation simplify wastewater treatment and disposal, ensuring compliance with environmental standards and reducing the overall ecological impact of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the operational parameters and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: Why is an iodide salt used in the synthesis of Tebipenem Pivoxil?
A: The iodide salt acts as a nucleophilic catalyst, facilitating the substitution reaction between the carboxyl group of Tebipenem and Chloromethyl pivalate. This catalytic effect significantly accelerates the reaction rate at lower temperatures, reducing the formation of thermal degradation impurities.
Q: How does this method improve upon conventional phase-transfer catalysis?
A: Conventional methods often require higher temperatures (around 45°C) and phase-transfer catalysts, leading to longer reaction times and complex purification via column chromatography. The iodide-catalyzed method operates at 0°C, eliminates the need for chromatography, and doubles the yield from approximately 30% to over 60%.
Q: What is the expected purity of the final Tebipenem Pivoxil product?
A: By utilizing a simplified crystallization workup instead of column chromatography, this process consistently achieves high purity levels ranging from 99.5% to 99.9%, meeting stringent pharmaceutical standards for oral carbapenem antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tebipenem Pivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the iodide-catalyzed synthesis of Tebipenem Pivoxil can be seamlessly translated from the laboratory to the manufacturing floor. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of carbapenem intermediate meets the highest global regulatory standards. We are committed to delivering high-purity pharmaceutical intermediates that empower our clients to bring life-saving antibiotics to market faster and more cost-effectively.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments, ensuring that your production of Tebipenem Pivoxil is built on a foundation of scientific excellence and commercial viability.
