Industrial Scale Production of High-Purity L-Carnitine via Continuous Aqueous Synthesis
Industrial Scale Production of High-Purity L-Carnitine via Continuous Aqueous Synthesis
The pharmaceutical and nutritional supplement industries have long sought efficient, scalable routes for the production of L-carnitine, a vital compound for lipid metabolism. Patent CN1265092A introduces a groundbreaking methodology that addresses the historical bottlenecks of yield, purity, and cost associated with traditional synthesis routes. This technology leverages a continuous aqueous phase reaction system, utilizing (S)-3-activated hydroxybutyrolactone as a chiral starting material to achieve high-purity L-carnitine without the need for complex intermediate purification steps. By integrating ring-opening, stereoselective epoxidation, and nucleophilic substitution into a streamlined workflow, this process represents a significant leap forward for manufacturers aiming to secure a reliable pharma intermediate supplier status while minimizing environmental impact and operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
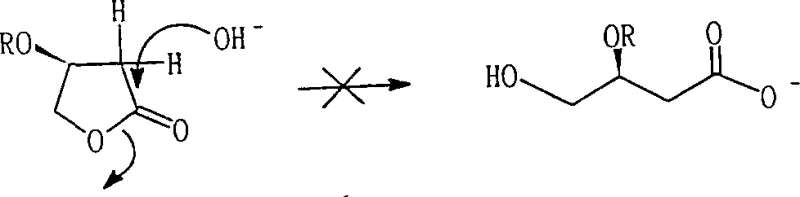
Historically, the production of optically pure L-carnitine has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional chemical optical resolution methods require the use of expensive chiral resolving agents, such as D-dextrocamphoric acid or dibenzoyl-tartrate, which not only inflate raw material costs but also limit the theoretical maximum yield to 50% for the desired enantiomer. Furthermore, biological methods, while stereoselective, suffer from extremely low reaction densities and prolonged reaction times ranging from 24 hours to several days, making them economically unviable for high-volume manufacturing. Previous chemical attempts using base-catalyzed ring-opening of activated lactones often failed due to competitive elimination reactions, where the base abstracts an acidic alpha-proton instead of attacking the carbonyl, leading to the formation of useless unsaturated byproducts rather than the desired open-chain precursor.
The Novel Approach
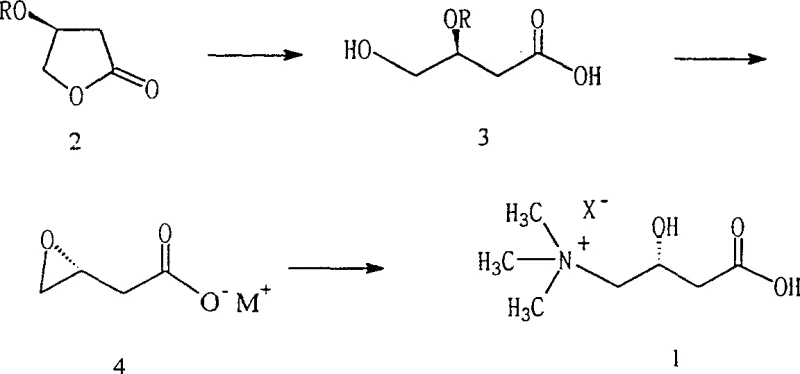
The innovative process described in the patent circumvents these issues by employing a specific acid-catalyzed ring-opening strategy followed by a controlled stereoinversion. Unlike prior art that struggled with side reactions, this method utilizes dilute mineral acids, such as sulfuric acid, to facilitate the hydrolysis of the lactone ring in an aqueous environment, effectively suppressing the elimination pathway. This allows for the quantitative formation of 4-hydroxy-3-activated hydroxybutyric acid, which then undergoes a rapid epoxidation with stereochemical inversion upon treatment with alkali. The entire sequence can be performed continuously in a single reactor system, drastically simplifying the workflow and eliminating the need for isolating unstable intermediates, thereby enhancing overall process robustness and throughput.
Mechanistic Insights into Acid-Catalyzed Ring Opening and Stereoinversion
A critical understanding of the reaction mechanism reveals why this specific pathway succeeds where others have failed, particularly regarding the stability of the chiral center and the suppression of side products. In conventional base-catalyzed attempts, the hydrogen atom at the alpha-position of the carbonyl group in the activated lactone exhibits significant acidity. When a base is introduced, it preferentially abstracts this proton, triggering an E2 elimination reaction that expels the activated hydroxyl group and forms an unsaturated furanone derivative, completely destroying the chiral integrity required for L-carnitine synthesis. The patent data explicitly demonstrates through NMR analysis that under basic conditions, the desired ring-opened product is barely detectable, with the elimination product dominating the reaction mixture.
Conversely, the introduction of an acid catalyst fundamentally shifts the reaction landscape by protonating the carbonyl oxygen or stabilizing the transition state, which renders the alpha-proton less susceptible to abstraction and directs the nucleophilic attack of water towards the carbonyl carbon. Following the successful ring opening, the subsequent epoxidation step is engineered to induce a precise stereochemical inversion at the chiral center. By treating the open-chain acid with a stoichiometric amount of alkali, an intramolecular nucleophilic substitution occurs where the deprotonated hydroxyl group displaces the activated leaving group. This SN2-type displacement ensures that the (S)-configuration of the starting lactone is inverted to the (R)-configuration in the epoxy intermediate, which is the necessary stereochemical precursor for the final nucleophilic attack by trimethylamine to yield biologically active L-carnitine.
How to Synthesize L-Carnitine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production, emphasizing the importance of maintaining specific pH conditions and solvent ratios to maximize yield. The process begins with the preparation of the activated lactone, followed by the critical acid-catalyzed hydrolysis in a water-organic cosolvent system to ensure solubility while maintaining reaction kinetics. Detailed standard operating procedures for temperature control, reagent addition rates, and workup strategies are essential for replicating the high purity levels reported in the intellectual property documentation.
- Perform acid-catalyzed ring-opening of (S)-3-activated hydroxybutyrolactone in an aqueous solvent to form 4-hydroxy-3-activated hydroxybutyric acid.
- Treat the open-chain acid with an alkali to induce epoxidation with stereochemical inversion, yielding 3,4-epoxy butyrate.
- React the epoxy intermediate with trimethylamine in aqueous solution to complete the nucleophilic substitution and form L-carnitine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous aqueous synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. By eliminating the need for expensive chiral resolving agents and complex extraction protocols associated with resolution-based methods, the raw material costs are significantly reduced, allowing for more competitive pricing structures in the global market. Furthermore, the ability to conduct multiple reaction steps in a single reactor without intermediate isolation minimizes solvent consumption and waste generation, aligning with modern green chemistry principles and reducing the burden on environmental compliance teams.
- Cost Reduction in Manufacturing: The elimination of costly optical resolution reagents and the reduction of unit operations lead to substantial savings in both material and labor costs. Since the process avoids the use of precious metal catalysts or specialized enzymes, the dependency on volatile commodity markets for these inputs is removed, stabilizing the cost of goods sold. Additionally, the high yield and purity achieved without chromatographic purification mean that downstream processing costs are drastically minimized, enhancing overall margin potential for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing readily available bulk chemicals such as sulfuric acid, sodium hydroxide, and trimethylamine ensures that the supply chain is not vulnerable to the shortages often associated with specialized biochemical reagents. The robustness of the aqueous phase chemistry allows for flexible manufacturing schedules, as the reaction is less sensitive to minor fluctuations in temperature or moisture compared to sensitive organometallic processes. This reliability translates to consistent lead times and the ability to fulfill large-volume orders without the risk of batch failures due to reagent instability.
- Scalability and Environmental Compliance: The continuous nature of the process facilitates seamless scale-up from pilot plants to multi-ton production facilities without the need for extensive re-engineering of the reaction infrastructure. Operating primarily in water reduces the volume of hazardous organic solvents required, simplifying waste treatment protocols and lowering the environmental footprint of the manufacturing site. This alignment with sustainability goals not only mitigates regulatory risks but also enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, drawing directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances is crucial for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing control strategies for the final active ingredient.
Q: Why is acid catalysis preferred over base catalysis for the ring-opening step?
A: Base catalysis leads to the deprotonation of the acidic alpha-hydrogen adjacent to the carbonyl, causing an elimination reaction that forms an unwanted unsaturated lactone. Acid catalysis suppresses this side reaction, allowing water to attack the carbonyl for successful ring opening.
Q: How is optical purity maintained during the synthesis?
A: The process starts with optically pure (S)-3-activated hydroxybutyrolactone. The subsequent epoxidation step involves a stereoselective inversion at the chiral center, ensuring the final L-carnitine product retains high optical purity (>95%) without the need for resolution.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly highlights that all reactions occur in an aqueous phase within a single reactor system without intermediate purification steps, making it highly suitable for continuous industrial mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnitine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying robust, scalable chemistries to meet the growing global demand for high-quality nutritional and pharmaceutical ingredients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of L-carnitine meets the highest international standards for optical purity and chemical identity, providing our partners with absolute confidence in their supply continuity.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency in your supply chain.
