Advanced Chiral Synthesis of L-Carnitine: Overcoming Racemic Limitations for Commercial Scale
The pharmaceutical and nutraceutical industries have long sought a robust, economically viable pathway for the production of L-carnitine, a critical molecule involved in fatty acid metabolism and energy production. Patent CN1158244C presents a significant technological breakthrough by detailing a method for preparing R-(-)-carnitine (L-carnitine) starting from S-(-)-chlorosuccinic acid or its derivatives. This approach fundamentally shifts the paradigm from inefficient racemic resolutions or costly enzymatic processes to a direct, stereospecific chemical synthesis. By leveraging the chiral pool of naturally occurring amino acids, specifically L-aspartic acid, this methodology ensures high optical purity while mitigating the substantial material losses inherent in traditional resolution techniques. For a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is essential for delivering high-quality active ingredients that meet stringent regulatory standards.
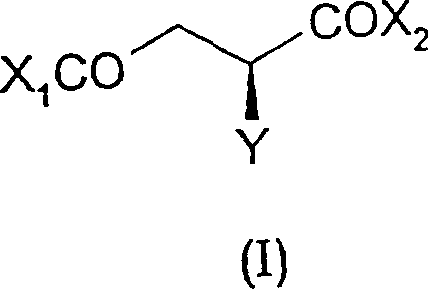
The core innovation described in the patent revolves around the reduction of a specific chiral intermediate, represented generally by Formula (I), followed by quaternization with trimethylamine. This route bypasses the need for separating enantiomers from a racemic mixture, a step that theoretically discards half of the produced material. Instead, the stereochemical integrity of the starting S-(-)-chlorosuccinic acid is preserved throughout the synthetic sequence, resulting in a final product with an enantiomeric excess (ee) of ≥99.6%. This level of purity is paramount for applications in human health, where the presence of the D-isomer can be detrimental or inactive. The process demonstrates exceptional control over impurity profiles, ensuring that the final L-carnitine inner salt contains less than 0.2% of the unwanted D-carnitine isomer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-carnitine has been plagued by significant inefficiencies, primarily stemming from the reliance on achiral or racemic starting materials. Traditional methods often begin with compounds such as racemic carnitine amide, epichlorohydrin, or 3-butenoic acid, necessitating a resolution step to isolate the biologically active L-enantiomer. This resolution process, whether achieved through crystallization with chiral acids or enzymatic kinetic resolution, imposes a hard ceiling on theoretical yield, capping it at 50% unless complex racemization and recycling loops are implemented. Furthermore, enzymatic approaches, while selective, introduce challenges related to catalyst cost, stability, and the removal of biological impurities which can act as potential allergens. The separation of enantiomers with identical physicochemical properties often requires sophisticated and expensive chromatographic techniques, driving up the cost of goods sold and complicating the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN1158244C utilizes a chiral building block, S-(-)-chlorosuccinic acid, which dictates the stereochemistry of the final product from the outset. This strategy eliminates the need for resolution entirely, effectively doubling the potential yield relative to racemic starting materials. The process is designed for industrial robustness, employing standard chemical reagents such as hydrides for reduction and trimethylamine for quaternization, rather than specialized biocatalysts. By converting the dicarboxylic acid into reactive derivatives like anhydrides, acid chlorides, or esters, the synthesis facilitates a smooth reduction of the carboxyl group to the corresponding alcohol without affecting the chiral center. This direct transformation not only simplifies the operational workflow but also significantly reduces the environmental footprint by minimizing solvent usage and waste generation associated with multiple purification cycles.
Mechanistic Insights into Hydride-Mediated Stereospecific Reduction
The heart of this synthetic route lies in the chemoselective and stereospecific reduction of the S-(-)-chlorosuccinic acid derivative. The patent describes the use of various hydride reducing agents, including diborane, lithium borohydride, and sodium borohydride, depending on the specific nature of the starting derivative (e.g., free acid, anhydride, or ester). When S-(-)-chlorosuccinic anhydride is treated with a mixed hydride in an aprotic solvent like tetrahydrofuran (THF) or dimethoxyethane (DME), the carbonyl group is reduced to a hydroxyl group. Crucially, this reduction proceeds with retention of configuration at the chiral carbon bearing the chlorine atom. The reaction conditions are meticulously controlled, often requiring low temperatures around -15°C to prevent side reactions such as dehalogenation or racemization, ensuring that the spatial arrangement of the atoms remains intact.
Following the reduction, the intermediate undergoes a nucleophilic substitution reaction where the chlorine atom is displaced by a trimethylamine group. This quaternization step is typically performed in the presence of a base, such as aqueous sodium hydroxide, which neutralizes the acidic byproducts and facilitates the formation of the zwitterionic L-carnitine inner salt. The mechanism ensures that the newly formed quaternary ammonium center is positioned correctly relative to the hydroxyl and carboxyl groups, replicating the natural structure of L-carnitine found in biological systems. The ability to perform these steps in a telescoped manner, without isolating the unstable reduced intermediate, further enhances the efficiency of the process and minimizes exposure to potentially hazardous reagents.
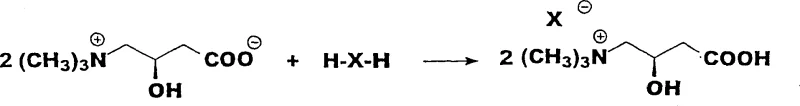
How to Synthesize L-Carnitine Inner Salt Efficiently
The synthesis of L-carnitine via this chiral route involves a sequence of activation, reduction, and amination steps that are optimized for high throughput and purity. The process begins with the preparation of S-(-)-chlorosuccinic acid from L-aspartic acid, followed by its conversion to a reactive anhydride. This anhydride is then subjected to reduction and subsequent treatment with trimethylamine to yield the final product. The detailed standardized synthesis steps for implementing this protocol in a GMP-compliant environment are provided in the guide below.
- Convert S-(-)-chlorosuccinic acid to its corresponding anhydride using acetic anhydride at elevated temperatures (50-90°C) to activate the carboxyl groups.
- Perform stereospecific reduction of the anhydride using mixed hydrides (e.g., NaBH4 or LiBH4) in aprotic solvents like THF or DME at controlled low temperatures (-15°C).
- Treat the crude reduction intermediate directly with aqueous sodium hydroxide and trimethylamine at 60-100°C to effect quaternization and ring closure, yielding L-carnitine inner salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain for raw materials. By relying on L-aspartic acid, a commodity fermentation product available in bulk quantities globally, manufacturers can decouple their production from the volatile markets of specialized chiral catalysts or resolution agents. This stability in raw material sourcing translates directly into enhanced supply chain reliability and predictable lead times, crucial for maintaining continuous production schedules in the competitive nutraceutical and pharmaceutical sectors. Moreover, the elimination of resolution steps means that the effective capacity of existing manufacturing infrastructure is nearly doubled, as every kilogram of starting material contributes to the final product rather than being discarded as the unwanted enantiomer.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven principally by the avoidance of the 50% theoretical yield loss associated with racemic resolution. By utilizing a chiral pool starting material, the process maximizes atom economy, ensuring that the cost of goods sold is significantly lower compared to traditional methods. Additionally, the ability to recycle mother liquors from the S-chlorosuccinic acid preparation step further reduces waste disposal costs and raw material consumption. The use of standard chemical reagents instead of expensive enzymes or transition metal catalysts also lowers the operational expenditure, making cost reduction in API manufacturing a reality without compromising on quality.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for proprietary enzymes or chiral ligands poses a significant risk to supply continuity. This synthetic route mitigates such risks by utilizing widely available commodity chemicals. The robustness of the chemical steps, which do not require sterile conditions or sensitive biological environments, allows for flexible manufacturing across different facilities. This flexibility ensures that production can be scaled or shifted rapidly in response to market demand fluctuations, thereby reducing lead time for high-purity pharmaceutical intermediates and securing the supply chain against external disruptions.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner profile. The reduction in solvent usage, achieved through higher reaction concentrations and the elimination of multiple crystallization steps for resolution, aligns with green chemistry principles. The waste stream is simpler to manage, consisting primarily of inorganic salts that are easier to treat than complex organic mixtures containing chiral auxiliaries. This simplification facilitates easier regulatory approval and supports the commercial scale-up of complex pharmaceutical intermediates, ensuring that production growth does not come at the expense of environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: How does this method improve optical purity compared to racemic resolution?
A: Unlike traditional methods starting from achiral materials which suffer a theoretical 50% yield loss during resolution, this process utilizes S-(-)-chlorosuccinic acid, a chiral pool starting material. This ensures the stereochemistry is maintained throughout the reduction, achieving an enantiomeric excess (ee) of ≥99.6% with D-carnitine impurities ≤0.2%.
Q: What are the key cost drivers in this synthesis route?
A: The primary cost advantage lies in the availability of the starting material. S-(-)-chlorosuccinic acid can be derived efficiently from L-aspartic acid with yields exceeding 80%. Furthermore, the process allows for the recycling of mother liquors and avoids expensive enzymatic catalysts or complex chiral chromatography steps required in other routes.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly addresses industrial scalability by optimizing reaction concentrations (e.g., >15% w/v for aspartic acid conversion) and utilizing standard unit operations like precipitation and filtration. The ability to use crude intermediates without extensive purification between steps significantly enhances throughput and reduces solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnitine Supplier
The technological advancements detailed in CN1158244C underscore the potential for producing L-carnitine with unprecedented efficiency and purity. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of L-carnitine meets the highest international pharmacopoeia standards, providing our partners with a consistent and reliable supply of this critical nutrient.
We invite global pharmaceutical and nutraceutical companies to collaborate with us to leverage this advanced synthesis route for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and security for your business operations.
