Advanced Enzymatic Route for High-Purity (S)-Indoline-2-Carboxylic Acid Production
The pharmaceutical industry continuously seeks robust methodologies for synthesizing chiral intermediates that balance high optical purity with economic feasibility. Patent CN100463903C introduces a transformative approach for preparing (S)-indoline-2-carboxylic acid and its methyl ester, pivotal building blocks for antihypertensive agents like Perindopril. This technology leverages industrially available hydrolases to resolve racemic mixtures, achieving optical purities exceeding 99% e.e. through a streamlined process that eliminates the need for expensive chiral auxiliaries or harsh chemical conditions. By shifting from traditional chemical resolution to biocatalysis, this method addresses critical bottlenecks in scalability and cost, positioning it as a superior choice for reliable pharmaceutical intermediate supplier networks aiming to optimize their API supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active (S)-indoline-2-carboxylic acid has relied on four primary strategies, each fraught with significant commercial and technical drawbacks that hinder large-scale adoption. Traditional recrystallization methods utilizing chiral auxiliaries like (+)-α-methylbenzylamine often struggle to exceed 96% e.e. and suffer from the high cost and difficult recovery of the resolving agents, severely impacting overall process economics. Furthermore, asymmetric hydrogenation techniques employing precious metal catalysts such as rhodium complexes require high-pressure equipment and sophisticated ligand systems, leading to substantial capital expenditure and complex purification protocols to remove trace metals. Chemical synthesis routes involving asymmetric reduction typically entail multi-step sequences with cumulative yields rarely exceeding 32%, while earlier enzymatic attempts using high molecular weight esters resulted in poor volumetric efficiency and complicated downstream processing due to emulsion formation.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a low molecular weight methyl ester substrate coupled with robust, commercially sourced hydrolases to achieve superior resolution efficiency. The process initiates with the straightforward esterification of racemic indoline-2-carboxylic acid using methanol and thionyl chloride, creating an ideal substrate for enzymatic attack.

By employing enzymes such as Savinase or Alcalase, the system selectively hydrolyzes the unwanted (R)-enantiomer, leaving the desired (S)-methyl ester intact with exceptional stereochemical fidelity. This kinetic resolution strategy operates under mild aqueous conditions, drastically simplifying the workflow compared to anhydrous organic synthesis and enabling cost reduction in API manufacturing by removing the dependency on rare earth metals and complex chiral ligands.
Mechanistic Insights into Hydrolase-Catalyzed Kinetic Resolution
The core of this technological advancement lies in the precise stereoselectivity of the chosen hydrolases, which function through a highly specific catalytic mechanism to differentiate between enantiomers. The enzyme, typically a serine protease like Savinase, possesses an active site geometry that accommodates the (R)-enantiomer of the indoline-2-carboxylic acid methyl ester far more readily than the (S)-form. Upon binding, the enzyme catalyzes the hydrolysis of the ester bond in the (R)-isomer, converting it into the corresponding carboxylic acid which becomes water-soluble in the buffered medium, while the (S)-ester remains unhydrolyzed and retains its lipophilic character.
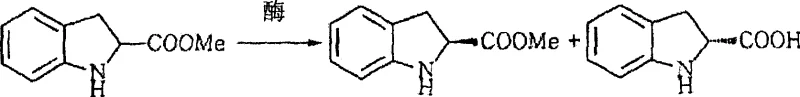
This differential reactivity allows for a clean physical separation where the desired product can be extracted into an organic phase, effectively decoupling the enantiomers without the need for diastereomeric salt formation. The patent highlights that maintaining the reaction pH between 7 and 9 and temperatures between 25°C and 50°C is critical for maximizing enzyme activity while preserving the structural integrity of the sensitive indoline ring. Impurity control is inherently managed by the enzyme's specificity, which minimizes side reactions common in chemical catalysis, thereby reducing the burden on downstream purification units and ensuring a cleaner impurity profile for the final active pharmaceutical ingredient.
How to Synthesize (S)-Indoline-2-Carboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for generating high-value chiral intermediates suitable for clinical and commercial applications. The process begins with the activation of the racemic acid followed by a carefully controlled enzymatic resolution step where parameters such as enzyme loading and substrate concentration are optimized to balance reaction rate and optical purity. Once the resolution is complete, the unreacted (S)-ester is isolated and subsequently hydrolyzed under basic conditions to yield the final free acid, with the entire sequence designed to minimize material loss and maximize throughput.
- React racemic indoline-2-carboxylic acid with methanol and thionyl chloride to form the racemic methyl ester.
- Perform enantioselective hydrolysis using a hydrolase (e.g., Savinase) in a buffered solution to isolate the (S)-ester.
- Hydrolyze the isolated (S)-ester in aqueous alkaline solution followed by acidification to recover the final (S)-acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this enzymatic platform offers profound strategic benefits that extend beyond simple yield improvements. The substitution of expensive chiral catalysts and auxiliaries with bulk industrial enzymes fundamentally alters the cost structure of the synthesis, driving down raw material expenses and reducing the complexity of waste management. Since the enzymes used, such as Savinase, are produced via fermentation on a massive scale for the detergent industry, they are available in large quantities at stable prices, insulating the supply chain from the volatility often associated with precious metal markets.
- Cost Reduction in Manufacturing: The elimination of costly chiral ligands and precious metal catalysts removes a significant portion of the bill of materials, while the simplified workup procedures reduce solvent consumption and energy usage. By avoiding high-pressure hydrogenation steps, the process lowers capital requirements for specialized reactor equipment, allowing for production in standard stainless steel vessels. The high selectivity of the enzyme minimizes the formation of byproducts, which translates to higher overall yields and less material wasted in purification columns, directly enhancing the gross margin of the final product.
- Enhanced Supply Chain Reliability: Utilizing commercially available enzymes ensures a consistent and reliable source of catalytic activity, mitigating the risks associated with custom-synthesized chiral reagents that may have long lead times. The robustness of the enzymatic process allows for flexible manufacturing schedules, as the reaction conditions are mild and do not require extreme temperatures or pressures that could cause unplanned downtime. Furthermore, the ability to source key reagents like methanol and thionyl chloride from multiple global suppliers reduces single-source dependency, strengthening the resilience of the supply network against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The aqueous nature of the enzymatic step aligns perfectly with green chemistry principles, significantly reducing the generation of hazardous organic waste compared to traditional chemical resolution methods. Scaling this process from pilot to commercial tonnage is straightforward because the kinetics are well-understood and the heat load is manageable, avoiding the thermal runaway risks associated with exothermic chemical reductions. This environmental compatibility simplifies regulatory approvals and waste disposal logistics, facilitating faster time-to-market for new drug formulations relying on this critical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this enzymatic resolution technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific production needs, as it clarifies the operational boundaries and performance expectations defined by the patent data.
Q: What optical purity can be achieved with this enzymatic method?
A: The patented process utilizing industrial hydrolases such as Savinase consistently achieves an optical purity of at least 99% e.e., surpassing traditional recrystallization methods.
Q: Which enzymes are suitable for this kinetic resolution?
A: The patent identifies a specific group of effective hydrolases including Savinase, Alcalase, Lipase 243, Everlase, Esperase, Protease 7, and Acylase, with Savinase being particularly preferred for its speed and cost-effectiveness.
Q: Why is the low molecular weight ester substrate advantageous?
A: Using the methyl ester rather than high molecular weight esters increases the number of moles per unit volume, significantly improving reaction efficiency and economic viability for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Indoline-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality chiral intermediates for the development of next-generation cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-indoline-2-carboxylic acid meets the exacting standards required by global regulatory bodies, providing our partners with the confidence needed to advance their clinical programs.
We invite you to engage with our technical procurement team to discuss how this advanced enzymatic technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient synthesis route. Contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, and let us demonstrate our commitment to being your trusted partner in pharmaceutical innovation.
