Advanced Asymmetric Hydrogenation Route for Commercial Pramipexole Dihydrochloride Production
The pharmaceutical landscape for neurological disorders continues to demand robust, scalable, and cost-effective synthetic routes for critical active pharmaceutical ingredients (APIs). Patent CN108084115B, published in March 2020, introduces a transformative preparation method for Pramipexole Dihydrochloride Monohydrate, a potent non-ergot dopamine agonist widely prescribed for idiopathic Parkinson's disease and restless leg syndrome. This patent details a novel synthetic strategy that bypasses the inefficiencies of traditional chiral resolution, leveraging advanced asymmetric hydrogenation technology to achieve superior yields and purity. For global procurement leaders and R&D directors, this innovation represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates. The disclosed method not only streamlines the production of this vital neuroactive compound but also aligns with modern green chemistry principles by reducing waste and energy consumption associated with repetitive purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Pramipexole has been hindered by cumbersome multi-step sequences that rely heavily on chiral resolution to isolate the biologically active enantiomer. As illustrated in prior art documents such as US4731374, the conventional pathway typically initiates from 4-acetamidocyclohexanone and involves extensive protection and deprotection strategies. A critical bottleneck in these legacy processes is the reliance on chiral resolution, often performed multiple times, which inherently caps the maximum theoretical yield at 50% per resolution step. In practice, the cumulative yield of these older methods is dismally low, reported at approximately 5.27%, resulting in substantial material loss and inflated production costs. Furthermore, the use of stoichiometric resolving agents generates significant chemical waste, complicating downstream processing and environmental compliance. These factors collectively render traditional methods economically unviable for large-scale commercial manufacturing in a competitive generic drug market.
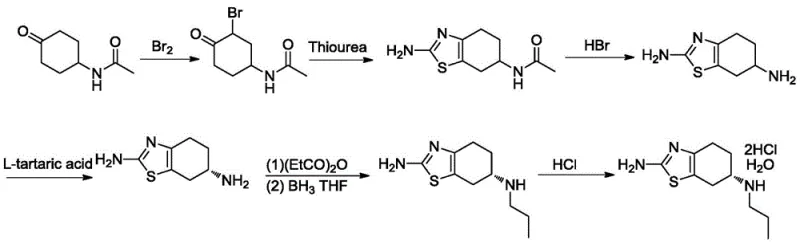
The Novel Approach
In stark contrast, the methodology disclosed in CN108084115B offers a paradigm shift by employing a direct, catalytic asymmetric synthesis. The core innovation lies in a one-pot condensation and reduction reaction where Pramipexole Intermediate III reacts with propylamine and hydrogen in the presence of a specialized chiral catalyst. This approach effectively installs the chiral center directly during the bond-forming event, obviating the need for inefficient resolution steps. The result is a dramatic improvement in process efficiency, with the total molar yield soaring to an impressive 40.5%, nearly eight times higher than conventional routes. The streamlined nature of this synthesis reduces the number of isolation steps, minimizes solvent usage, and shortens the overall production cycle time. By integrating the formation of the chiral amine directly into the main synthetic line, this novel approach provides a reliable pathway for producing high-purity Pramipexole Dihydrochloride that meets stringent raw material medicine standards.
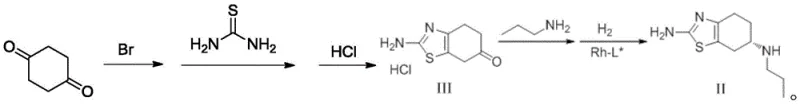
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
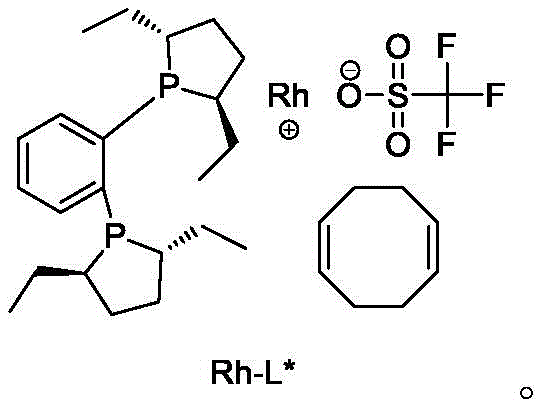
The heart of this technological breakthrough is the utilization of a sophisticated rhodium-based chiral catalyst system. Specifically, the patent identifies (+)-1,2-bis((2S,5S)-diethylphospholano)benzene(cyclooctadiene)rhodium trifluoromethanesulfonate as the preferred catalyst. This complex features a rigid chiral ligand framework that creates a highly defined steric environment around the rhodium center. During the reaction, the catalyst coordinates with the imine intermediate formed in situ from the ketone and propylamine, directing the addition of hydrogen to specifically favor the formation of the desired (S)-enantiomer. The precision of this catalytic cycle ensures that the enantiomeric excess (ee) is maintained at exceptionally high levels throughout the reaction, with final product specifications showing enantiomer content as low as 0.06%. This level of stereocontrol is critical for pharmaceutical applications, where the presence of the wrong enantiomer can lead to reduced efficacy or adverse side effects.
Beyond stereocontrol, the mechanistic design of this process also facilitates superior impurity management. The one-pot nature of the condensation and reduction minimizes the exposure of reactive intermediates to harsh conditions that could generate degradation byproducts. Additionally, the subsequent workup procedures, involving pH adjustment and extraction with dichloromethane, are optimized to remove residual catalyst metals and organic impurities effectively. The patent data indicates that the crude product obtained after hydrogenation already possesses high purity (over 96%), which is further enhanced to 99.28% through a single recrystallization step using L-di-p-toluoyl tartaric acid if necessary. This robust impurity profile simplifies the quality control burden and ensures that the final API intermediate consistently meets the rigorous specifications required for regulatory submission and commercial release.
How to Synthesize Pramipexole Dihydrochloride Efficiently
Implementing this advanced synthesis requires careful attention to reaction parameters, particularly regarding the catalyst loading and hydrogen pressure. The process begins with the preparation of Intermediate III from 1,4-cyclohexanedione, followed by the critical asymmetric hydrogenation step. Operators must maintain strict control over temperature (preferably 35-65°C) and hydrogen pressure (4-10 atmospheres) to ensure optimal conversion rates and selectivity. The detailed procedural nuances, including solvent ratios and workup protocols, are essential for reproducing the high yields reported in the patent examples. For technical teams looking to adopt this methodology, the following guide outlines the standardized operational steps derived directly from the patent disclosure.
- Synthesize Pramipexole Intermediate III by reacting 1,4-cyclohexanedione with bromine and thiourea in methanol, followed by acidification.
- Perform one-pot condensation and asymmetric hydrogenation of Intermediate III with propylamine using a Rh-L* chiral catalyst to obtain Pramipexole II.
- Convert Pramipexole II to the final dihydrochloride monohydrate salt using concentrated hydrochloric acid in isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of chiral resolution steps fundamentally alters the cost structure of Pramipexole manufacturing by removing the need for expensive resolving agents and the associated volume losses. This structural change translates directly into a more favorable cost of goods sold (COGS), allowing for more competitive pricing in the generic pharmaceutical market. Furthermore, the reduction in unit operations decreases the demand for reactor time and labor, enhancing overall plant throughput and asset utilization. These efficiencies make the supply of this critical neurological agent more resilient against market fluctuations and raw material price volatility.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic increase in overall molar yield, which rises from roughly 5% in legacy methods to over 40% in the new route. This nearly eight-fold improvement means that significantly less starting material is required to produce the same amount of final API, leading to substantial savings on raw material procurement. Additionally, by avoiding the multiple crystallization and filtration steps associated with chiral resolution, the process consumes less solvent and energy, further driving down utility costs. The simplified workflow also reduces the generation of hazardous waste, lowering disposal fees and environmental compliance costs, thereby creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of readily available, commodity-grade starting materials such as 1,4-cyclohexanedione and propylamine, which are not subject to the same supply constraints as specialized chiral pool reagents. The robustness of the catalytic hydrogenation step ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. Moreover, the shorter synthetic sequence reduces the lead time for production cycles, enabling manufacturers to respond more agilely to sudden spikes in demand for Parkinson's disease medications. This reliability is crucial for maintaining uninterrupted supply to downstream formulators and ensuring patient access to essential therapies.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard unit operations like hydrogenation and liquid-liquid extraction that are well-understood in industrial chemical engineering. The ability to scale from kilogram to multi-ton quantities without significant process re-engineering facilitates rapid capacity expansion to meet global market needs. From an environmental perspective, the atom economy of the catalytic route is superior to stoichiometric resolution methods, resulting in a lower E-factor (mass of waste per mass of product). This alignment with green chemistry principles supports corporate sustainability goals and simplifies the regulatory approval process for new manufacturing sites, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived from the experimental data and process descriptions found within CN108084115B, providing clarity on the feasibility and advantages of this technology for potential partners and licensees. Understanding these details is essential for evaluating the strategic fit of this route within your existing manufacturing portfolio.
Q: What is the primary advantage of the new Rh-catalyzed route over traditional methods?
A: The new route eliminates the need for multiple chiral resolution steps, significantly improving the total molar yield from roughly 5% to over 40% while simplifying the operational workflow.
Q: How is high enantiomeric purity achieved in this process?
A: High purity is achieved through the use of a specialized chiral rhodium catalyst ((+)-1,2-bis((2S,5S)-diethylphospholano)benzene(cyclooctadiene)rhodium trifluoromethanesulfonate) during the hydrogenation step, ensuring precise stereocontrol.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available starting materials and standard unit operations like hydrogenation and crystallization, making it highly scalable for commercial manufacturing up to 100 MT annually.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pramipexole Dihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting next-generation synthetic technologies to maintain a competitive edge in the pharmaceutical intermediate market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity Pramipexole Dihydrochloride that adheres to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement complex chiral catalysis protocols allows us to offer a superior product profile that meets the exacting standards of global regulatory bodies.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced manufacturing route for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project benefits from the highest levels of quality, efficiency, and reliability available in the industry.
