Optimizing Ramelteon Intermediate Production: A Strategic Analysis of Patent CN103304524A for Global Supply Chains
Optimizing Ramelteon Intermediate Production: A Strategic Analysis of Patent CN103304524A for Global Supply Chains
The global demand for effective insomnia treatments continues to drive the market for melatonin receptor agonists, with Ramelteon standing out as a pivotal therapeutic agent. At the heart of its manufacturing lies the critical synthesis of its key precursor, 2-(1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-yl)ethylamine hydrochloride. Patent CN103304524A introduces a transformative methodology that addresses long-standing inefficiencies in producing this essential pharmaceutical intermediate. By shifting away from traditional phosphonate-based chemistry towards a more direct condensation strategy using cyanoacetic acid, this technology offers a robust pathway for enhancing both chemical efficiency and operational safety. For stakeholders in the fine chemical sector, understanding this shift is crucial for maintaining competitive advantage in the reliable pharmaceutical intermediate supplier landscape.
This technical insight delves deep into the mechanistic and commercial implications of the patented process. The innovation not only streamlines the synthetic route but also fundamentally alters the impurity profile, offering significant benefits for quality control teams. As regulatory scrutiny on residual solvents and heavy metals intensifies, a process that minimizes the use of exotic reagents and simplifies purification becomes invaluable. We will explore how this specific approach aligns with modern Green Chemistry principles while delivering the economic efficiencies required by procurement departments globally.
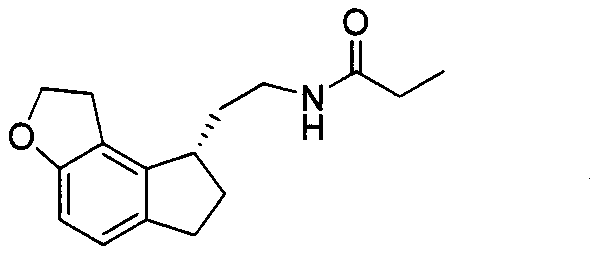
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Ramelteon amine intermediate has relied on a multi-step sequence that begins with the hydrodebromination of the dibromo-ketone starting material. As illustrated in the conventional pathway, this initial step converts the dibromo compound into a monoketone, which is subsequently reacted with diethyl cyanomethylphosphonate to extend the carbon chain. This traditional approach suffers from several critical drawbacks that hinder cost reduction in pharmaceutical intermediates manufacturing. Firstly, the hydrodebromination step is prone to over-reduction, leading to the formation of persistent byproducts such as compounds (VII) and (VIII), which are structurally similar to the desired product and difficult to separate. Secondly, the use of diethyl cyanomethylphosphonate introduces significant safety and environmental liabilities due to its toxicity and the generation of phosphorous-containing waste streams.
Furthermore, the conventional route necessitates harsh reaction conditions and expensive reagents that escalate the overall cost of goods sold (COGS). The requirement for distinct steps to install the ethylamine side chain after removing the bromine atoms adds unnecessary operational complexity, increasing the potential for yield loss at each stage. From a supply chain perspective, reliance on specialized phosphonate reagents creates vulnerability, as these materials often have longer lead times and stricter handling requirements compared to commodity chemicals. The accumulation of these inefficiencies results in a process that is chemically elegant but commercially suboptimal for large-scale production, prompting the industry to seek superior alternatives.
The Novel Approach
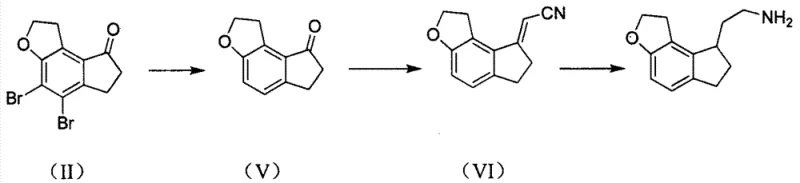
The methodology disclosed in CN103304524A represents a paradigm shift by utilizing 4,5-dibromo-1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-one directly in a condensation reaction with cyanoacetic acid. This innovative strategy bypasses the problematic hydrodebromination step entirely, thereby eliminating the root cause of the troublesome dehalogenated impurities. Instead of a stepwise installation of the side chain, the new process achieves chain extension and functional group transformation in a unified sequence. The reaction proceeds via a dehydration-condensation mechanism followed by thermal decarboxylation, yielding the nitrile intermediate (IV) with remarkable efficiency. This consolidation of steps not only reduces the total processing time but also significantly lowers the consumption of solvents and energy.
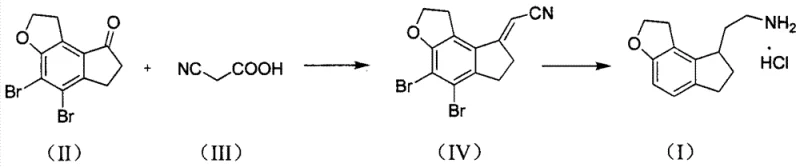
Subsequent conversion of the nitrile intermediate to the final amine hydrochloride is achieved through a sophisticated catalytic hydrogenation protocol. By employing a combination of Palladium on Carbon and Raney Nickel under controlled basic and ammoniacal conditions, the process simultaneously accomplishes debromination, double bond saturation, and nitrile reduction. This telescoped transformation is a masterclass in atom economy and process intensification. The result is a streamlined workflow that delivers the target intermediate in high yield and purity, ready for the final chiral resolution and acylation steps to produce Ramelteon. For manufacturers, this translates to a more resilient and cost-effective supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
Mechanistic Insights into Cyanoacetic Acid Condensation and Catalytic Hydrogenation
The core of this technological advancement lies in the unique reactivity of cyanoacetic acid when paired with the specific dibromo-ketone substrate. In the presence of organic catalysts such as benzylamine or carboxylic acids, cyanoacetic acid undergoes a Knoevenagel-type condensation with the carbonyl group of the ketone. This reaction is driven forward by the removal of water, often facilitated by azeotropic distillation using solvents like toluene. The resulting alpha,beta-unsaturated nitrile intermediate is inherently unstable under the reaction conditions and spontaneously undergoes decarboxylation. This thermal decarboxylation is a critical driving force that pushes the equilibrium towards the formation of the desired exocyclic double bond product, effectively installing the two-carbon nitrile fragment while retaining the bromine atoms necessary for the subsequent reduction phase.
Following the isolation of the nitrile intermediate, the catalytic hydrogenation step requires precise control to manage multiple concurrent reductions. The process utilizes a dual-catalyst system where Palladium on Carbon initially facilitates the hydrodebromination and alkene reduction under mild basic conditions. The addition of Raney Nickel in an ammonia-saturated medium is then employed to reduce the nitrile group to the primary amine without causing excessive hydrogenolysis of the benzylic position or ring saturation. The presence of ammonia is crucial as it suppresses the formation of secondary and tertiary amine byproducts, ensuring high selectivity for the primary amine. This nuanced control over the catalytic environment allows for the direct conversion of the complex brominated nitrile into the clean amine hydrochloride salt, demonstrating a high level of chemical sophistication.
How to Synthesize Ramelteon Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly regarding the molar ratios of cyanoacetic acid and the choice of catalyst system. The patent specifies that a molar excess of cyanoacetic acid (1:1.05 to 1:5 relative to the ketone) ensures complete conversion, while the use of solvents capable of forming azeotropes with water, such as toluene or xylene, is preferred to drive the condensation to completion. The subsequent hydrogenation must be conducted in a pressure reactor capable of handling hydrogen gas safely, with temperature control maintained between 20°C and 65°C to optimize reaction kinetics without compromising selectivity. Detailed standard operating procedures for scaling this route are essential for maintaining batch-to-batch consistency.
- Condense 4,5-dibromo-1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-one with cyanoacetic acid using an organic catalyst in a refluxing solvent like toluene to form the nitrile intermediate.
- Perform catalytic hydrogenation using Palladium on Carbon under basic conditions, followed by Raney Nickel reduction in an ammonia-containing medium.
- Isolate the final product by salification with hydrochloric acid gas to obtain the hydrochloride salt with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple yield improvements. The substitution of expensive and hazardous phosphonate reagents with commodity-grade cyanoacetic acid represents a fundamental shift in the cost structure of the intermediate. Cyanoacetic acid is widely available from multiple global suppliers, reducing dependency on single-source vendors and mitigating supply risk. Furthermore, the elimination of the hydrodebromination step removes an entire unit operation from the manufacturing schedule, effectively shortening the production cycle time and increasing facility throughput without the need for capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the raw material portfolio. By removing the need for diethyl cyanomethylphosphonate, manufacturers avoid the high costs associated with purchasing, storing, and disposing of toxic phosphorus compounds. Additionally, the higher overall yield resulting from the avoidance of side reactions means that less starting material is required to produce the same amount of final product. This improvement in mass balance directly lowers the variable cost per kilogram, providing a competitive edge in pricing negotiations with downstream API manufacturers while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of robust, non-proprietary reagents. Unlike specialized phosphonates which may face regulatory shipping restrictions or availability fluctuations, cyanoacetic acid and standard hydrogenation catalysts are staple chemicals in the fine chemical industry. This accessibility ensures that production schedules are less likely to be disrupted by raw material shortages. Moreover, the simplified process flow reduces the number of intermediate isolations and transfers, minimizing the logistical complexity and the potential for human error or contamination during manufacturing operations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The absence of phosphorous waste simplifies effluent treatment, reducing the burden on wastewater processing facilities and lowering compliance costs. The ability to isolate the intermediate via simple crystallization from the reaction mixture, rather than complex chromatographic separations, makes the process inherently scalable from pilot plant to multi-ton commercial production. This scalability ensures that the supply of the Ramelteon intermediate can be rapidly ramped up to meet market surges, securing the continuity of supply for the finished dosage form.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of the cyanoacetic acid route over the phosphonate method?
A: The cyanoacetic acid route eliminates the need for toxic and expensive diethyl cyanomethylphosphonate, reduces reaction steps by combining debromination and reduction, and avoids the formation of difficult-to-remove byproducts associated with hydrodebromination.
Q: How does this process improve impurity control for GMP manufacturing?
A: By avoiding the initial hydrodebromination step, the process prevents the generation of dehalogenated side products (compounds VII and VIII), resulting in a cleaner crude profile that simplifies downstream purification and ensures higher final purity.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method utilizes common solvents like toluene and ethanol, employs standard heterogeneous catalysts (Pd/C, Raney Ni), and relies on crystallization for isolation, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ramelteon Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Ramelteon depends on the reliability and quality of the supply chain. Our technical team has extensively analyzed advanced synthetic routes, including the innovations described in CN103304524A, to ensure our manufacturing capabilities are aligned with the highest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity intermediates. Our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting requirements necessary for GMP API synthesis, minimizing the risk of downstream processing failures.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized processes and robust supply network, we can help you achieve significant efficiencies in your procurement strategy. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to technological excellence and operational integrity can support your goals in the competitive global pharmaceutical market.
