Scalable Synthesis of Key Aztreonam Intermediate Using Non-Toxic Sulfamic Acid Methodology
Scalable Synthesis of Key Aztreonam Intermediate Using Non-Toxic Sulfamic Acid Methodology
The pharmaceutical industry continuously seeks robust and safe pathways for the production of critical antibiotic intermediates, particularly for monobactams like Aztreonam. Patent CN100486964C, published on May 13, 2009, introduces a groundbreaking process for synthesizing (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid, a pivotal building block in the Aztreonam value chain. This innovation addresses long-standing safety and efficiency bottlenecks associated with traditional sulfonation methods by substituting hazardous reagents with safer, solid-state alternatives. For R&D directors and supply chain managers, this patent represents a significant opportunity to optimize the manufacturing of beta-lactam antibiotics while adhering to stricter environmental and safety regulations. The methodology leverages L-Threonine as a chiral pool starting material, ensuring stereochemical integrity throughout the five-step sequence, which includes amino protection, condensation with sulfamic acid, hydroxyl protection, cyclization, and final deprotection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key azetidinone sulfonic acid intermediate has relied heavily on aggressive and hazardous chemistry. As detailed in prior art such as US4775670, the conventional route typically involves an aminosulfonation step utilizing chlorosulfonic acid. This reagent is notoriously difficult to handle due to its extreme corrosivity and the evolution of toxic hydrogen chloride gas during the reaction. Furthermore, the operational complexity of managing liquid chlorosulfonic acid on an industrial scale requires specialized equipment and rigorous safety protocols, which inherently drives up capital expenditure and operational costs. Additionally, these older methods often suffer from lower yields in the critical sulfonation step and generate substantial acidic waste streams that require expensive neutralization and disposal procedures. The reliance on such dangerous reagents also complicates the supply chain, as the procurement and transport of chlorosulfonic acid are subject to stringent regulatory controls, potentially leading to delays and continuity risks for manufacturers.
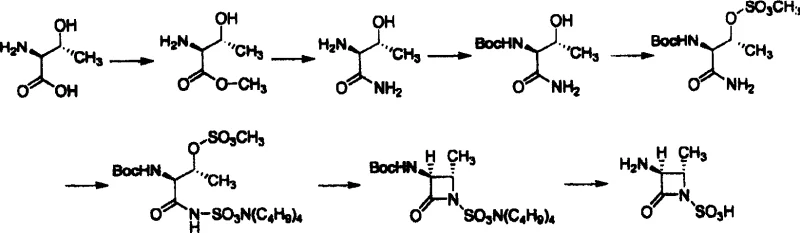
The Novel Approach
In stark contrast, the novel approach outlined in CN100486964C fundamentally reengineers the synthetic pathway to prioritize safety and operational simplicity without compromising yield or purity. Instead of chlorosulfonic acid, this method employs solid sulfamic acid for the initial introduction of the sulfonic acid moiety, followed by protection of the hydroxyl group using methylsulfonyl chloride. This strategic shift eliminates the need for handling free ammonia gas and highly corrosive liquid acids, thereby creating a much safer working environment for plant personnel. The process utilizes common organic solvents such as tetrahydrofuran and dichloromethane, allowing for seamless integration into existing fine chemical manufacturing facilities. By avoiding the harsh conditions of the past, this new route not only mitigates environmental impact but also streamlines the purification process, as evidenced by the high purity of the crude products obtained. This modernization of the synthetic logic demonstrates a clear path toward greener chemistry in the production of complex antibiotic intermediates.
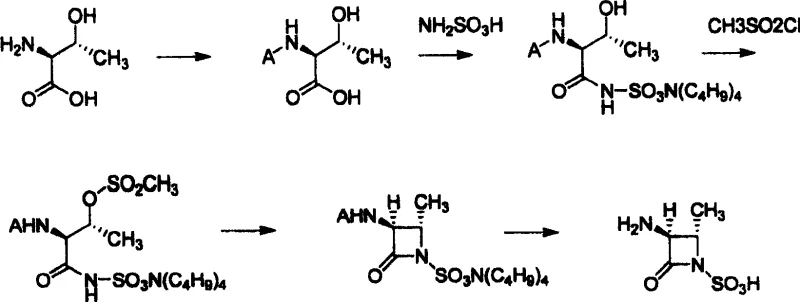
Mechanistic Insights into Stereoselective Beta-Lactam Formation
The core of this synthetic strategy lies in the precise manipulation of the L-Threonine backbone to construct the strained four-membered azetidinone ring while maintaining the critical (2S, trans) stereochemistry required for biological activity. The process begins with the protection of the alpha-amino group of L-Threonine, typically using Boc anhydride or benzyl chloroformate, which prevents unwanted side reactions during subsequent steps. The subsequent condensation with sulfamic acid is facilitated by coupling agents like DCC and HOBt in anhydrous THF, forming a stable threonyl aminosulfonic acid derivative. This step is crucial as it installs the nitrogen-sulfur bond that will eventually become part of the sulfonic acid side chain. The stereochemistry at the alpha and beta carbons of the threonine residue is preserved throughout these transformations, ensuring that the final product possesses the correct spatial configuration necessary for binding to penicillin-binding proteins in Gram-negative bacteria.
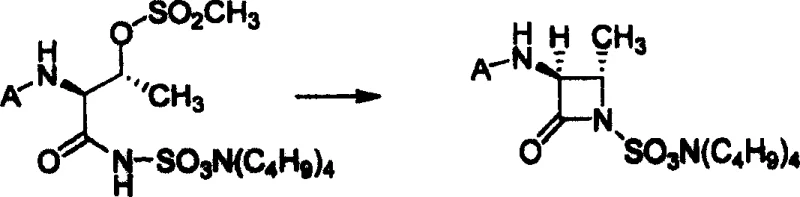
Following the installation of the sulfonic acid precursor, the hydroxyl group of the threonine side chain is activated via mesylation using methylsulfonyl chloride. This activation converts the poor leaving group (-OH) into an excellent leaving group (-OMs), setting the stage for the intramolecular nucleophilic attack that forms the beta-lactam ring. The cyclization step is promoted by a mineral base, such as potassium carbonate or sodium bicarbonate, in a biphasic system. This base deprotonates the sulfonamide nitrogen, generating a nucleophile that attacks the carbon bearing the mesylate group, displacing the mesylate and closing the four-membered ring. This intramolecular SN2-type reaction proceeds with inversion of configuration at the beta-carbon, resulting in the desired trans-relationship between the methyl group and the amino group in the final azetidinone structure. The final deprotection step removes the amino protecting group under mild acidic or hydrogenolytic conditions to reveal the free amine, yielding the target sulfonic acid intermediate.
How to Synthesize (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid Efficiently
The synthesis of this high-value intermediate requires careful control of reaction parameters, particularly temperature and stoichiometry, to maximize yield and minimize impurities. The patent provides a detailed roadmap that transitions from simple amino acid derivatives to the complex heterocyclic target through a series of well-defined unit operations. Operators must pay close attention to the anhydrous conditions required during the coupling and mesylation steps to prevent hydrolysis of activated intermediates. The use of phase transfer catalysts, such as tetrabutylammonium hydrogen sulfate, during the extraction phases significantly improves the recovery of the polar sulfonic acid derivatives from the aqueous layer. For a comprehensive understanding of the specific reagent quantities, reaction times, and workup procedures required to replicate this process in a GMP environment, please refer to the standardized technical guide below.
- Protect the amino group of L-Threonine using Boc anhydride or benzyl chloroformate in the presence of a base.
- Condense the protected threonine with sulfamic acid using DCC and HOBt in anhydrous THF to form the threonyl aminosulfonic acid derivative.
- Protect the hydroxyl group in the threonine segment by reacting with methylsulfonyl chloride.
- Perform a cyclization reaction using a mineral base to form the protected azacyclobutyl sulfonic acid quaternary ammonium salt.
- Execute a final deprotection step using acid (for Boc) or hydrogenation (for Cbz) to obtain the final sulfonic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical advantages over legacy methods. The primary driver for cost optimization is the substitution of chlorosulfonic acid with solid sulfamic acid and methylsulfonyl chloride. This change eliminates the need for specialized corrosion-resistant reactors and extensive scrubbing systems required to handle toxic gases, thereby significantly reducing capital investment and maintenance costs for manufacturing facilities. Furthermore, the avoidance of hazardous reagents simplifies the procurement process, as sulfamic acid and mesyl chloride are commodity chemicals with stable supply chains and lower regulatory burdens compared to controlled corrosives. This stability ensures a more reliable supply of raw materials, reducing the risk of production stoppages due to raw material shortages or transportation restrictions.
- Cost Reduction in Manufacturing: The elimination of toxic chlorosulfonic acid and gaseous ammonia leads to substantial savings in waste treatment and environmental compliance costs. Traditional processes generate large volumes of acidic wastewater that require neutralization before discharge, whereas the new method produces less hazardous waste streams that are easier and cheaper to treat. Additionally, the simplified operational procedure reduces the labor hours required for reactor charging and monitoring, contributing to lower overall conversion costs. The higher yields reported in the patent embodiments further enhance cost efficiency by maximizing the output per kilogram of starting L-Threonine, effectively lowering the cost of goods sold for the final intermediate.
- Enhanced Supply Chain Reliability: By relying on widely available solid reagents and standard organic solvents, manufacturers can diversify their supplier base and reduce dependency on single-source vendors of hazardous chemicals. The robustness of the process against minor variations in reaction conditions also improves batch-to-b consistency, ensuring a steady flow of high-quality intermediate to downstream customers. This reliability is critical for maintaining uninterrupted production schedules for finished antibiotic formulations, especially in times of global supply chain volatility. The ability to store stable intermediates and perform reactions at moderate temperatures further adds to the flexibility of the supply chain, allowing for better inventory management.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind from the outset. The use of biphasic systems and common extraction solvents facilitates easy scale-up from pilot plant to commercial tonnage without the need for exotic equipment. From an environmental perspective, the reduction in toxic waste generation aligns with green chemistry principles and increasingly stringent global environmental regulations. This compliance not only avoids potential fines but also enhances the corporate sustainability profile of the manufacturer, making the product more attractive to environmentally conscious pharmaceutical partners who prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for adopting this newer methodology in an existing production portfolio.
Q: Why is the new sulfamic acid method preferred over the traditional chlorosulfonic acid route?
A: The traditional method utilizes highly toxic and corrosive chlorosulfonic acid for the aminosulfonation step, which poses significant safety hazards and environmental disposal challenges. The novel process described in CN100486964C replaces this with solid sulfamic acid and methylsulfonyl chloride, drastically reducing toxicity, simplifying operational handling, and improving overall process safety for industrial scale-up.
Q: What is the expected purity of the final intermediate using this protocol?
A: According to the experimental embodiments provided in the patent, the final product, (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid, achieves a liquid chromatography detection purity of greater than 98.5%. This high level of purity is critical for downstream coupling reactions in the synthesis of the final antibiotic, Aztreonam.
Q: Can this synthesis route be adapted for large-scale commercial production?
A: Yes, the process is explicitly designed for industrial suitability. By avoiding difficult-to-handle gaseous ammonia and liquid chlorosulfonic acid, and utilizing standard organic solvents like dichloromethane and ethyl acetate, the route offers simplified unit operations. The use of solid reagents and phase transfer catalysts further enhances its feasibility for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the global supply of life-saving antibiotics. Our technical team has extensively analyzed the pathway described in CN100486964C and possesses the expertise to execute this non-toxic, high-yield process with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your demand for Aztreonam precursors is met with consistency and quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the >98.5% purity benchmark highlighted in the patent, guaranteeing that every batch meets the exacting standards required for pharmaceutical synthesis.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security for your antibiotic production programs.
