Advanced Manufacturing of Aztreonam Key Intermediate via Safe Aminosulfonic Acid Route
The pharmaceutical industry continuously seeks robust and scalable pathways for critical antibiotic intermediates, particularly for monobactams like Aztreonam. Patent CN1974551A introduces a transformative methodology for synthesizing (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid, a pivotal building block in the production of this potent Gram-negative antibiotic. Unlike legacy processes that rely on hazardous sulfonating agents, this innovation leverages a condensation strategy using solid aminosulfonic acid derivatives. This shift not only mitigates severe safety risks associated with corrosive liquids but also streamlines the purification workflow, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for beta-lactam antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the sulfonic acid moiety in azetidinone derivatives has been a significant bottleneck in process chemistry. Traditional routes, such as those disclosed in earlier patents like US4775670, frequently employ chlorosulfonic acid as the sulfonating agent. While chemically effective, chlorosulfonic acid presents formidable challenges for industrial implementation due to its extreme corrosivity and toxicity. Handling this reagent requires specialized glass-lined or Hastelloy reactors, rigorous safety protocols, and complex quenching procedures to manage the evolution of hydrogen chloride gas. Furthermore, the reaction conditions are often harsh, leading to potential racemization of the chiral center derived from L-threonine and generating substantial amounts of acidic waste that complicate environmental compliance and increase disposal costs significantly.
The Novel Approach
The methodology described in CN1974551A circumvents these hazards by utilizing aminosulfonic acid as a stable, solid alternative for introducing the sulfonamide functionality. This approach fundamentally changes the risk profile of the synthesis, replacing a dangerous liquid reagent with a manageable solid that can be weighed and transferred with standard equipment. The process initiates with the protection of L-threonine, followed by a peptide-like coupling with aminosulfonic acid activated by DCC and HOBt. This mild condensation preserves the stereochemical integrity of the starting material. Subsequent mesylation of the hydroxyl group and base-mediated cyclization efficiently construct the strained four-membered beta-lactam ring. By avoiding the direct use of chlorosulfonic acid, this route eliminates the need for expensive corrosion-resistant infrastructure and simplifies the downstream workup, resulting in a cleaner crude product profile.
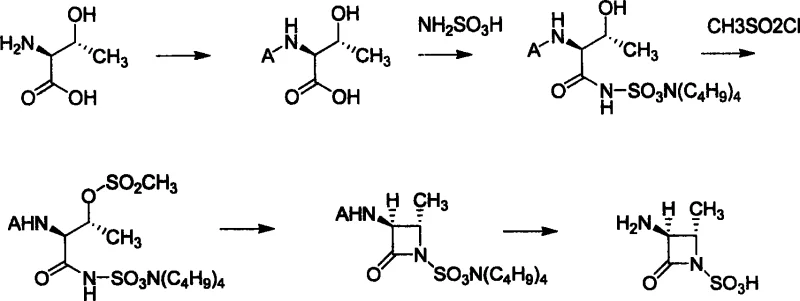
Mechanistic Insights into Peptide Coupling and Beta-Lactam Formation
The core of this synthetic strategy lies in the activation of the carboxylic acid of protected L-threonine to facilitate nucleophilic attack by the amine group of aminosulfonic acid. In the second step of the process, N,N'-dicyclohexylcarbodiimide (DCC) serves as the dehydrating agent, reacting with the carboxyl group to form an reactive O-acylisourea intermediate. The addition of 1-hydroxybenzotriazole (HOBt) is critical here; it converts the unstable O-acylisourea into a more stable and less racemization-prone active ester. This active species then undergoes nucleophilic substitution by the nitrogen of the aminosulfonic acid, forming the amide bond. The use of tetra-n-butylammonium hydrogen sulfate as a phase transfer catalyst during the extraction phase further enhances the recovery of the polar sulfonic acid derivative, ensuring high mass balance across this critical transformation step.
Following the formation of the linear precursor, the cyclization step is driven by the intramolecular nucleophilic attack of the sulfonamide nitrogen on the carbon bearing the mesylate leaving group. This displacement reaction closes the four-membered azetidinone ring. The reaction is typically conducted in a biphasic system using a mineral base such as potassium carbonate or sodium bicarbonate. The base serves a dual purpose: it neutralizes the methanesulfonic acid generated during the leaving group departure and maintains the nucleophilicity of the sulfonamide nitrogen by deprotonation. The stereochemistry is rigorously controlled throughout this sequence, as the reaction conditions are sufficiently mild to prevent epimerization at the C3 and C4 positions of the emerging beta-lactam ring, which is essential for the biological activity of the final Aztreonam API.
How to Synthesize (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid Efficiently
Executing this synthesis requires precise control over temperature and stoichiometry, particularly during the activation and cyclization phases. The protocol begins with the protection of L-threonine using di-tert-butyl dicarbonate (Boc2O) or benzyl chloroformate in a water-dioxane mixture, establishing the necessary orthogonality for later deprotection. The subsequent coupling with aminosulfonic acid must be performed under anhydrous conditions in THF at low temperatures to minimize side reactions. After mesylation of the hydroxyl group with methanesulfonyl chloride, the cyclization is effected by refluxing in a dichloroethane-water system with a weak base. Finally, the protecting groups are removed—either via acidolysis for Boc groups or catalytic hydrogenation for Cbz groups—to yield the target zwitterionic sulfonic acid. For detailed operational parameters and specific molar ratios, please refer to the standardized guide below.
- Protect the amino group of L-Threonine using Boc anhydride or benzyl chloroformate in a biphasic system.
- Condense the protected threonine with aminosulfonic acid using DCC and HOBt in anhydrous THF at low temperature.
- Protect the hydroxyl group with methanesulfonyl chloride, followed by base-catalyzed cyclization to form the beta-lactam ring.
- Perform final deprotection using formic acid or catalytic hydrogenation to obtain the pure sulfonic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this aminosulfonic acid-based route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating chlorosulfonic acid, facilities can reduce their dependency on highly regulated hazardous chemicals, thereby lowering insurance premiums and reducing the administrative burden associated with transporting and storing corrosive substances. This shift also mitigates the risk of supply disruptions caused by strict environmental regulations on hazardous reagent manufacturers, ensuring a more resilient supply chain for this critical antibiotic intermediate.
- Cost Reduction in Manufacturing: The elimination of chlorosulfonic acid removes the necessity for specialized, high-cost corrosion-resistant reactors and piping systems, allowing production to occur in standard stainless steel or glass-lined vessels. Furthermore, the avoidance of aggressive acidic quenching steps reduces the consumption of neutralizing bases and lowers the volume of saline wastewater generated. The use of solid aminosulfonic acid also improves atom economy in the sulfonation step compared to traditional sulfonyl chlorides, leading to better overall material utilization and reduced raw material costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: Solid reagents like aminosulfonic acid and Boc-anhydride possess superior shelf stability compared to liquid sulfonyl chlorides or chlorosulfonic acid, which are prone to degradation upon exposure to moisture. This stability allows for larger batch purchasing and longer inventory holding periods without quality degradation, providing greater flexibility in production scheduling. Additionally, the simplified workup procedures, which avoid complex distillations of corrosive byproducts, shorten the overall cycle time per batch, enabling faster turnaround times and more responsive fulfillment of customer orders.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste, aligning with modern green chemistry principles and reducing the costs associated with waste treatment and disposal. The mild reaction conditions and the use of common organic solvents like THF and dichloromethane facilitate straightforward scale-up from pilot plant to commercial tonnage without encountering the heat transfer or mixing limitations often seen with highly exothermic sulfonation reactions. This scalability ensures that suppliers can reliably meet increasing market demand for Aztreonam without requiring massive capital expenditure on new specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a transparent view of the technology's capabilities and limitations for potential partners and licensees.
Q: Why is the aminosulfonic acid route preferred over chlorosulfonic acid for this intermediate?
A: The traditional chlorosulfonic acid route involves highly corrosive and toxic reagents that are difficult to handle on an industrial scale and generate significant hazardous waste. The novel aminosulfonic acid route utilizes solid, stable reagents, significantly improving operational safety and simplifying waste treatment protocols.
Q: What is the expected purity of the final product using this method?
A: According to the patent data, the liquid chromatography detection purity of the final product exceeds 98.5%, meeting the stringent requirements for downstream antibiotic synthesis such as Aztreonam.
Q: Can this process be scaled for commercial production?
A: Yes, the process is specifically designed for industrial suitability. It avoids extreme conditions and uses common solvents like THF and dichloromethane, making it adaptable for large-scale reactor systems without requiring specialized corrosion-resistant equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficient production of beta-lactam intermediates is foundational to the global supply of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of (2S-trans)-3-amino-2-methyl-4-oxo-1-azacyclobutyl sulfonic acid meets the exacting standards required for GMP API synthesis, providing our partners with absolute confidence in material quality.
We invite pharmaceutical manufacturers and procurement specialists to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By leveraging our optimized process knowledge, we can help you identify opportunities to reduce COGS and improve throughput. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can strengthen your supply chain resilience.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
