Advanced Synthesis of 2,4,5-Triamino-6-Hydroxypyrimidine Sulfate for Commercial API Production
The global demand for high-purity antiviral and antianemia agents continues to drive innovation in the synthesis of critical heterocyclic intermediates. Patent CN115286584A introduces a transformative preparation method for 2,4,5-triamino-6-hydroxypyrimidine sulfate (TAHMS), a pivotal building block for renowned pharmaceuticals such as acyclovir and folic acid. This technical disclosure addresses long-standing inefficiencies in traditional manufacturing, specifically targeting the cumbersome handling of inorganic salts and excessive waste generation. By re-engineering the sequence of reagent addition during the cyclization phase, the inventors have achieved a process that not only simplifies downstream purification but also enables the high-value recovery of sodium nitrate byproducts. For R&D directors and procurement strategists, this represents a shift from linear production to a circular economy model within the reactor, where waste streams are converted into saleable commodities.
Furthermore, the implications of this technology extend beyond mere chemical elegance; they touch upon the core economic metrics of API manufacturing. The ability to recover sodium nitrate with a purity of 99.9% directly impacts the cost of goods sold (COGS) by offsetting raw material expenses and reducing hazardous waste disposal fees. As the pharmaceutical industry faces increasing regulatory pressure regarding environmental compliance, adopting such green chemistry principles becomes a strategic imperative rather than just an operational choice. This report analyzes the mechanistic advantages and commercial viability of this novel route, providing a roadmap for integrating this efficient synthesis into existing supply chains for reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4,5-triamino-6-hydroxypyrimidine derivatives has been plagued by the co-generation of substantial inorganic salts that are difficult to separate from the organic product. In standard protocols, guanidine nitrate, methyl cyanoacetate, and sodium methoxide are often reacted simultaneously or in a sequence that traps the resulting sodium nitrate within the reaction matrix. This entanglement creates a viscous, salt-laden slurry that complicates stirring and heat transfer, leading to inconsistent reaction kinetics and lower overall yields. Moreover, the presence of these salts necessitates extensive washing and filtration steps post-nitrosation, which not only consumes vast quantities of water but also generates significant volumes of saline wastewater that require expensive treatment before discharge. The traditional approach essentially treats the byproduct salt as a liability, incurring high energy costs for drying and disposal while offering no economic return.
The Novel Approach
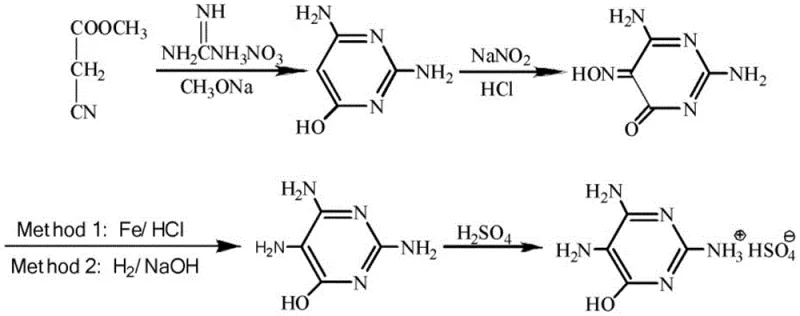
The methodology disclosed in CN115286584A fundamentally disrupts this paradigm by decoupling the salt formation from the organic cyclization. By initially reacting guanidine nitrate with sodium methoxide and immediately filter-pressing the mother liquor, the process isolates the sodium nitrate byproduct before the introduction of the carbon skeleton source, methyl cyanoacetate. This strategic sequencing ensures that the subsequent cyclization occurs in a salt-free environment, dramatically improving reaction homogeneity and thermal control. The result is a cleaner reaction profile that eliminates the need for difficult post-nitrosation filtration steps, as the bulk of the inorganic burden has already been removed. This streamlined workflow not only accelerates the production cycle but also facilitates the recovery of high-purity industrial-grade sodium nitrate, turning a previous waste stream into a revenue-generating asset while simultaneously reducing the environmental footprint of the manufacturing facility.
Mechanistic Insights into Sodium Methoxide-Catalyzed Cyclization
The core chemical transformation in this process is the base-catalyzed condensation between methyl cyanoacetate and guanidine nitrate to form the pyrimidine ring. Mechanistically, sodium methoxide acts as a strong base to deprotonate the active methylene group of methyl cyanoacetate, generating a nucleophilic enolate species. In the novel process, this enolate attacks the electrophilic carbon of the guanidine moiety. However, the critical innovation lies in the timing of this interaction relative to salt precipitation. By pre-forming the sodium guanidinate species and removing the sodium nitrate precipitate prior to adding the ester, the reaction medium is optimized for nucleophilic attack without the steric and solvation interference caused by high concentrations of dissolved salts. This leads to a more efficient ring closure, minimizing side reactions such as hydrolysis of the nitrile or ester groups which are common in salt-saturated environments.
Following cyclization, the nitrosation step introduces the amino group at the 5-position via a diazonium intermediate mechanism, which is subsequently reduced to the amine. The patent specifies tight pH control between 1.5 and 2.0 during nitrosation, which is crucial for stabilizing the nitroso intermediate and preventing over-oxidation or decomposition. The subsequent catalytic hydrogenation, utilizing catalysts such as Raney nickel or palladium on carbon, reduces the nitroso group to an amino group with high selectivity. Because the earlier steps have removed the bulk of inorganic salts, the hydrogenation catalyst is less prone to poisoning or fouling, ensuring consistent activity over multiple batches. This robustness in the catalytic cycle is essential for maintaining high purity specifications and reducing the frequency of catalyst replacement, thereby enhancing the overall process economics and reliability for commercial scale-up of complex heterocycles.
How to Synthesize 2,4,5-Triamino-6-Hydroxypyrimidine Sulfate Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable pathway that balances reaction efficiency with operational simplicity. The process begins with the preparation of a salt-free cyclization solution, followed by a controlled nitrosation under acidic conditions, and concludes with a catalytic reduction and acid salification. Each step is designed to maximize yield while minimizing the generation of hazardous waste, making it an ideal candidate for modern green manufacturing facilities. The detailed standardized synthetic steps see the guide below, which breaks down the specific temperature profiles, stoichiometric ratios, and safety precautions required for successful implementation.
- Cyclization: React guanidine nitrate with sodium methoxide, filter to recover sodium nitrate, then add methyl cyanoacetate for ring closure.
- Nitrosation: Treat the cyclization product with sodium nitrite and dilute sulfuric acid under strict pH control (1.5-2.0).
- Reduction & Salifying: Perform catalytic hydrogenation to reduce the nitroso group, followed by crystallization with sulfuric acid to obtain the final sulfate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible benefits that extend well beyond the laboratory bench. The most immediate impact is seen in the drastic reduction of raw material consumption, particularly sodium methoxide, which is a costly reagent in fine chemical synthesis. By optimizing the reaction stoichiometry and eliminating salt interference, the process achieves the same conversion with significantly less base, directly lowering the variable cost per kilogram of the final API intermediate. Furthermore, the ability to recover and sell high-purity sodium nitrate and sodium sulfate byproducts creates a new revenue stream that offsets production costs, effectively subsidizing the manufacturing of the primary product and improving the overall margin structure.
- Cost Reduction in Manufacturing: The elimination of excess sodium methoxide usage translates to substantial direct savings on raw material procurement. Additionally, the process simplifies the workflow by removing the post-nitrosation filtration and alkali dissolution steps, which reduces labor hours, energy consumption for heating and cooling, and the wear and tear on processing equipment. The recovery of valuable byproducts further enhances the economic profile, turning waste disposal costs into potential income and significantly lowering the net production cost compared to legacy methods.
- Enhanced Supply Chain Reliability: By simplifying the unit operations and reducing the dependency on complex separation techniques, the process becomes more robust and less prone to batch failures or delays. The reduced water consumption—reported to be around 15 tons less per ton of product—alleviates pressure on facility utilities, ensuring that production schedules are not impacted by water availability or wastewater treatment bottlenecks. This operational stability ensures a consistent supply of high-purity intermediates, reducing lead time for high-purity intermediates and securing the continuity of the downstream API supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been demonstrated in multi-thousand-liter reactors with excellent reproducibility. The significant reduction in three wastes (wastewater, waste gas, and solid waste) aligns with stringent environmental regulations, minimizing the risk of regulatory shutdowns or fines. The ability to treat process wastewater biologically due to the removal of high salt content simplifies environmental compliance, making the facility more sustainable and attractive to eco-conscious partners and investors looking for responsible chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves specific pain points in traditional manufacturing. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this supply partner.
Q: How does the new cyclization method improve sodium nitrate recovery?
A: By reacting guanidine nitrate and sodium methoxide first and filtering the mother liquor before adding methyl cyanoacetate, the process isolates sodium nitrate in a high-purity state (99.9%) without interference from the organic product.
Q: What are the primary cost drivers reduced in this patented process?
A: The process significantly reduces the consumption of sodium methoxide by approximately 400-450kg per ton of product and eliminates the need for extensive wastewater treatment associated with traditional salt-heavy methods.
Q: Is this method suitable for large-scale API intermediate manufacturing?
A: Yes, the patent demonstrates scalability in 2000L to 5000L reactors, featuring simplified unit operations such as eliminating post-nitrosation filtration, which enhances throughput and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Triamino-6-Hydroxypyrimidine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires not just chemical expertise but deep engineering capability. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised in the lab are fully realized on the plant floor. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 2,4,5-triamino-6-hydroxypyrimidine sulfate meets the exacting standards required for antiviral and vitamin synthesis, providing our clients with the confidence needed to accelerate their own drug development timelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener, more efficient route can improve your bottom line. Contact us today to discuss specific COA data and route feasibility assessments, and let us help you secure a sustainable, cost-effective supply of this critical pharmaceutical intermediate.
