Advanced Neutral Hydrogenation Technology for Scalable TAHMS Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates like 2,4,5-triamino-6-hydroxypyrimidine sulfate (TAHMS), a pivotal building block for antiviral agents such as acyclovir and anti-anemia medications like folic acid. Patent CN102399194A introduces a transformative methodology that addresses longstanding inefficiencies in the hydrogenation step of TAHMS production. Traditionally, the reduction of the nitroso precursor has been plagued by environmental hazards and complex pH adjustments, but this invention leverages surfactant chemistry to enable direct catalytic hydrogenation under neutral conditions. By eliminating the need for excessive alkaline dissolution followed by acidic neutralization, the process significantly reduces wastewater generation and minimizes the formation of colored impurities caused by oxidative side reactions. This technical breakthrough not only enhances the economic viability of the synthesis but also aligns with modern green chemistry principles, offering a cleaner pathway for reliable pharmaceutical intermediate supplier networks to adopt. The strategic implementation of surfactants such as sodium dodecylbenzenesulfonate or polyvinylpyrrolidone allows for the effective dispersion of the hydrophobic substrate in aqueous media, fundamentally altering the mass transfer dynamics of the reaction.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of TAHMS has relied on reduction methods that are increasingly untenable due to environmental regulations and cost inefficiencies. Early techniques utilized sodium sulfide reduction, which generated toxic hydrogen sulfide gas and required rigorous pH control between 8 and 9, resulting in substantial wastewater treatment burdens. Subsequent iterations employed iron or zinc powder in acidic media, a method that produced massive quantities of hazardous metal sludge that was difficult to dispose of safely. Even the more advanced catalytic hydrogenation methods described in prior art, such as those utilizing alkaline conditions to dissolve the substrate, introduced significant operational complexities. Because the substrate 2,4-diamino-5-nitroso-6-hydroxypyrimidine is insoluble in neutral or acidic water, manufacturers were forced to dissolve it in alkali before hydrogenation, necessitating a subsequent neutralization step with hydrochloric acid before final salt formation with sulfuric acid. This alternating use of acids and bases not only inflated raw material costs but also promoted hydrolysis and oxidation side reactions, often yielding yellow-tinted products with compromised purity profiles that required additional refining steps.
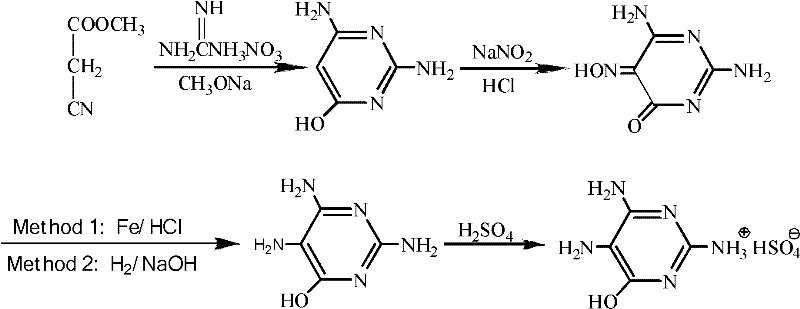
The Novel Approach
The innovative process detailed in the patent data circumvents these solubility and stability issues by introducing a surfactant-mediated dispersion system that operates entirely under neutral conditions. Instead of forcing the substrate into solution with harsh alkalis, the method utilizes specific surfactants to disrupt the strong intermolecular hydrogen bonding network of the nitroso compound, allowing it to form a stable suspension in water. This physical modification enables the hydrogen gas and solid catalyst to interact effectively with the substrate particles without the chemical degradation associated with high pH environments. Consequently, the reaction proceeds smoothly at moderate temperatures of 40-60°C and pressures of 1-5 MPa, yielding a white crystalline product with superior optical properties. By bypassing the alkaline dissolution and subsequent acid neutralization steps, the process drastically simplifies the workflow, reduces the consumption of auxiliary chemicals, and ensures a higher degree of cost reduction in API intermediate manufacturing through streamlined operations and reduced waste disposal fees.
Mechanistic Insights into Surfactant-Assisted Neutral Hydrogenation
The core scientific advancement of this technology lies in the manipulation of intermolecular forces to overcome solubility barriers without compromising chemical integrity. Molecular simulation analyses indicate that the nitroso pyrimidine derivative possesses a robust network of intermolecular hydrogen bonds that prevent water molecules from penetrating and solvating the structure under neutral conditions. The addition of amphiphilic surfactants acts to interfere with these cohesive forces, effectively lowering the energy barrier for dispersion and creating a micro-environment where the hydrophobic substrate is accessible to the catalyst surface. This mechanism ensures that the reduction of the nitroso group to the amino group occurs efficiently via heterogeneous catalysis using metals like palladium or nickel, without the need for the substrate to be fully dissolved in a reactive alkaline medium. The preservation of neutral pH throughout the hydrogenation phase is critical, as it suppresses the nucleophilic attack of hydroxide ions on the pyrimidine ring, thereby preventing the hydrolysis reactions that typically lead to ring-opening impurities and discoloration.
Furthermore, the control of impurity profiles is intrinsically linked to the avoidance of oxidative degradation pathways that are accelerated in alkaline solutions. In traditional alkaline hydrogenation processes, the presence of oxygen or oxidizing species can lead to the formation of complex byproducts that impart a yellow color to the final crystal lattice, necessitating costly recrystallization or charcoal treatment steps. The neutral surfactant system maintains a chemically inert environment regarding the pyrimidine core, ensuring that the only significant transformation is the desired reduction of the nitroso functionality. This selectivity results in a crude product that is already of high-purity pharmaceutical intermediates standard, often exceeding 98% purity directly after crystallization. The ability to produce white crystals consistently indicates a low level of conjugated impurities, which is a vital quality attribute for downstream coupling reactions in the synthesis of acyclovir and folic acid, where impurity carryover can affect the efficacy and safety of the final active pharmaceutical ingredient.
How to Synthesize 2,4,5-Triamino-6-Hydroxypyrimidine Sulfate Efficiently
Implementing this surfactant-assisted hydrogenation protocol requires precise control over dispersion quality and reaction parameters to maximize the benefits of the neutral pH environment. The process begins with the preparation of a uniform suspension of the nitroso precursor in deionized water containing a carefully selected surfactant, such as Tween 80 or sodium dodecylbenzenesulfonate, at a concentration of 0.1-2% relative to the substrate mass. Once the suspension is stabilized, a solid catalyst like Pd/C or Raney Ni is introduced, and the system is purged with nitrogen to remove oxygen before pressurizing with hydrogen gas. The reaction is maintained at a temperature range of 40-60°C and a pressure of 1-5 MPa for a duration of 2-6 hours, ensuring complete conversion while minimizing thermal stress on the product. Following the hydrogenation, the catalyst is removed by filtration, and the filtrate undergoes a two-stage acidification process: first adjusted to pH 3-4 with dilute hydrochloric acid to remove residual basic impurities, and then to pH 1-2 with sulfuric acid to induce crystallization of the target sulfate salt. For a comprehensive guide on the specific reagent grades and equipment specifications required for this synthesis, please refer to the standardized protocol below.
- Disperse 2,4-diamino-5-nitroso-6-hydroxypyrimidine in water containing a surfactant (e.g., SDBS, PVP) to form a neutral suspension.
- Perform catalytic hydrogenation using Pd/C or Raney Ni at 40-60°C and 1-5 MPa pressure under nitrogen protection.
- Filter the catalyst, adjust pH to 3-4 with HCl, filter again, and finally acidify with sulfuric acid to pH 1-2 to crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this neutral hydrogenation technology offers substantial advantages in terms of operational expenditure and supply chain resilience. The elimination of the alkaline dissolution step removes the need for large quantities of caustic soda or other strong bases, which are not only costly but also require specialized handling and storage infrastructure. Similarly, the reduction in acid consumption for neutralization purposes directly lowers the recurring cost of raw materials, contributing to a more favorable cost structure for the final intermediate. The simplified workflow, which avoids the back-and-forth pH adjustments of traditional methods, reduces the total batch cycle time and increases the throughput capacity of existing hydrogenation reactors. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations, ensuring a steady supply of critical intermediates without the bottlenecks associated with complex waste treatment or extended purification cycles. The overall result is a more robust and economically sustainable production model that enhances competitiveness in the global marketplace.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in the consumption of auxiliary acids and bases. By operating under neutral conditions, the process avoids the stoichiometric consumption of alkali required to dissolve the substrate and the subsequent acid needed to neutralize it before salt formation. This chemical saving is compounded by the reduction in wastewater volume, which lowers the operational costs associated with effluent treatment plants and environmental compliance fees. Additionally, the high selectivity of the reaction minimizes product loss due to side reactions, effectively increasing the yield per unit of raw material input and further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: The use of common, commercially available surfactants and standard heterogeneous catalysts ensures that the supply chain for raw materials remains stable and resilient. Unlike processes that rely on specialized or scarce reagents, this method utilizes commodities like Pd/C or Raney Ni and widely produced surfactants such as PVP or Tween, which are readily sourced from multiple vendors globally. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the neutral condition process reduces the sensitivity to minor variations in water quality or reagent purity, making the manufacturing process more forgiving and consistent across different production sites.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure hydrogenation equipment that is common in fine chemical facilities, facilitating the commercial scale-up of complex heterocyclic compounds without the need for exotic reactor designs. The significant reduction in hazardous waste generation, specifically the absence of heavy metal sludge from iron/zinc methods and the reduction of saline wastewater from acid-base neutralization, simplifies regulatory compliance. This environmental advantage is increasingly critical as global regulations tighten around industrial effluent discharge, positioning manufacturers who adopt this technology as preferred partners for multinational corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this surfactant-assisted synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature, ensuring accuracy and relevance for process development scientists. These insights clarify the operational boundaries and quality expectations associated with the production of TAHMS using this advanced methodology.
Q: Why is surfactant addition critical in this hydrogenation process?
A: The substrate 2,4-diamino-5-nitroso-6-hydroxypyrimidine has strong intermolecular hydrogen bonds, making it insoluble in neutral water. Surfactants weaken these bonds, enabling dispersion without alkaline dissolution, which prevents hydrolysis side reactions.
Q: What are the typical purity levels achieved with this method?
A: According to patent data, this neutral hydrogenation method consistently yields white crystalline products with purity levels ≥98% and single-batch yields reaching up to 95%.
Q: Which catalysts are compatible with this surfactant system?
A: The process is versatile and supports conventional solid catalysts including skeleton nickel (Raney Ni), palladium on carbon (Pd/C), and platinum on carbon (Pt/C), allowing flexibility based on cost and availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Triamino-6-Hydroxypyrimidine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient production of life-saving medications like acyclovir and folic acid. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2,4,5-triamino-6-hydroxypyrimidine sulfate meets the exacting standards required by the global pharmaceutical industry. Our facility is equipped to handle the specific requirements of surfactant-assisted hydrogenation, providing a secure and compliant environment for the manufacture of this valuable intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics and our proven track record in delivering high-purity pharmaceutical intermediates reliably and efficiently.
