Advanced Catalytic Hydrogenation for N-Benzyl-3-Piperidinol Production
The pharmaceutical industry's relentless pursuit of cost-effective and safe manufacturing processes for antihypertensive agents has led to significant innovations in intermediate synthesis, specifically regarding the critical precursor N-benzyl-3-piperidinol. As detailed in patent CN101817779B, a groundbreaking methodology has been established that fundamentally alters the economic and safety landscape of producing this key building block for Benidipine. This novel approach leverages a specialized nickel-based catalyst system supported on diatomite, operating under remarkably moderate hydrogen pressures compared to historical standards. For R&D directors and procurement managers alike, this technology represents a pivotal shift away from hazardous high-pressure operations and expensive noble metal dependencies, offering a robust pathway for commercial scale-up of complex pharmaceutical intermediates. The integration of quaternary ammonium salt chemistry with heterogeneous catalysis ensures not only high yield but also exceptional purity profiles essential for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-benzyl-3-piperidinol has been plagued by severe engineering and economic bottlenecks associated with two primary legacy routes. The first conventional method relies on the direct hydrogenation of 3-hydroxypyridine using Raney Nickel, followed by alkylation with benzyl bromide. While the raw materials are inexpensive, this process necessitates extreme hydrogen pressures approaching 100 atm, imposing exorbitant capital expenditures on high-pressure hydrogenation reactors and creating significant safety liabilities due to the pyrophoric nature of Raney Nickel. 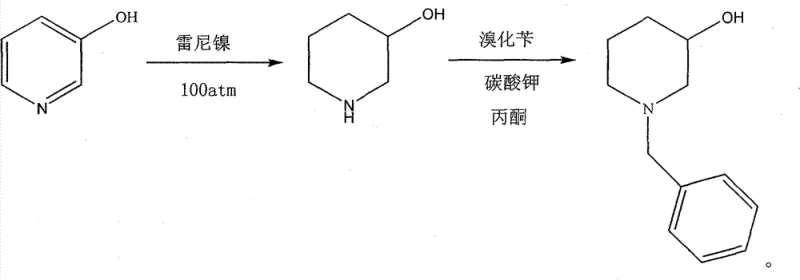 The second prevailing method utilizes noble metal catalysts such as platinum dioxide or platinum on carbon to lower the pressure requirements. Although this mitigates some safety concerns by allowing reactions below 5 atm, the prohibitive cost of precious metals like platinum and rhodium severely impacts the overall cost of goods sold (COGS), making it difficult to achieve competitive pricing in a generic drug market.
The second prevailing method utilizes noble metal catalysts such as platinum dioxide or platinum on carbon to lower the pressure requirements. Although this mitigates some safety concerns by allowing reactions below 5 atm, the prohibitive cost of precious metals like platinum and rhodium severely impacts the overall cost of goods sold (COGS), making it difficult to achieve competitive pricing in a generic drug market. 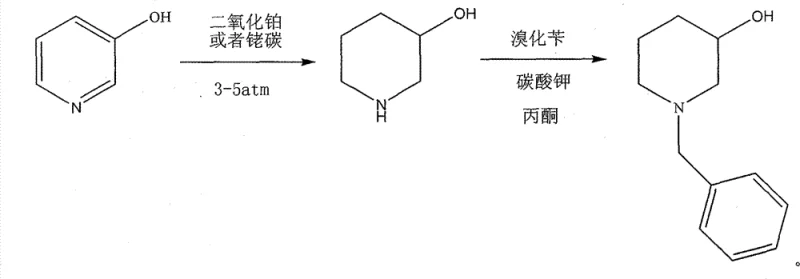 Both methods suffer from either unacceptable operational risks or unsustainable material costs, creating an urgent demand for a superior alternative.
Both methods suffer from either unacceptable operational risks or unsustainable material costs, creating an urgent demand for a superior alternative.
The Novel Approach
The innovative strategy disclosed in the patent data reverses the traditional synthetic sequence to overcome these entrenched limitations effectively. Instead of reducing the pyridine ring first, the process begins by converting 3-hydroxypyridine into its corresponding N-benzyl-3-hydroxypyridine quaternary ammonium salt through reaction with benzyl chloride. This quaternization step activates the aromatic ring towards reduction, allowing for catalytic hydrogenation under much milder conditions. 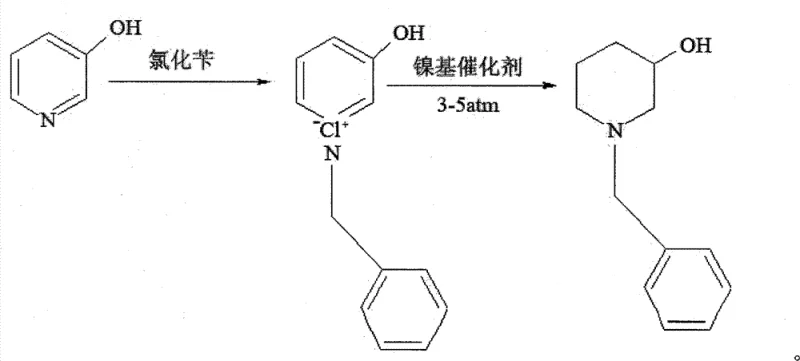 By employing a proprietary nickel-boron-cobalt catalyst supported on diatomite, the hydrogenation proceeds efficiently at merely 3 to 5 atm and 70°C. This inversion of the synthetic logic eliminates the need for dangerous high-pressure equipment while avoiding the financial burden of noble metals. Furthermore, the specific formulation of the catalyst prevents the common side reaction of debenzylation, ensuring that the benzyl group remains intact throughout the reduction, thereby maximizing yield and simplifying purification protocols significantly.
By employing a proprietary nickel-boron-cobalt catalyst supported on diatomite, the hydrogenation proceeds efficiently at merely 3 to 5 atm and 70°C. This inversion of the synthetic logic eliminates the need for dangerous high-pressure equipment while avoiding the financial burden of noble metals. Furthermore, the specific formulation of the catalyst prevents the common side reaction of debenzylation, ensuring that the benzyl group remains intact throughout the reduction, thereby maximizing yield and simplifying purification protocols significantly.
Mechanistic Insights into Ni-B-Co Catalytic Hydrogenation
The core of this technological breakthrough lies in the precise composition and structural characteristics of the non-noble metal catalyst. The active catalytic species consists of a ternary alloy system comprising approximately 40% to 70% nickel, 10% to 40% cobalt, and 3% to 20% boron, all dispersed upon a diatomite support matrix. The inclusion of cobalt acts as a promoter that enhances the electron density and stability of the nickel active sites, while boron modifies the surface acidity to favor the adsorption of the quaternary ammonium substrate. This synergistic effect allows the catalyst to facilitate the complete saturation of the pyridinium ring without cleaving the benzylic C-N bond, a selectivity challenge that often plagues standard nickel catalysts. The diatomite support provides a high surface area and mechanical strength, ensuring that the catalyst particles remain distinct and filterable, which is critical for preventing metal contamination in the final product.
From an impurity control perspective, the mechanism inherently suppresses the formation of 3-hydroxypiperidine, a major byproduct resulting from debenzylation. In conventional hydrogenation of N-benzyl pyridines, the benzyl group is susceptible to hydrogenolysis, especially under vigorous conditions or with overly active catalysts. However, the electron-deficient nature of the quaternary pyridinium ring makes it more susceptible to nucleophilic attack by hydride species generated on the catalyst surface than the benzylic bond is to cleavage. Consequently, the reduction occurs rapidly at the ring carbons before any significant degradation of the N-substituent can take place. This kinetic preference results in a crude product profile that is exceptionally clean, with gas chromatography analysis confirming purities greater than 99% prior to final distillation, drastically reducing the load on downstream purification units.
How to Synthesize N-Benzyl-3-Piperidinol Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding temperature, pressure, and stoichiometry to ensure reproducibility and safety. The process begins with the quaternization of 3-hydroxypyridine in toluene at elevated temperatures, followed by a carefully controlled hydrogenation step in an ethanol-triethylamine solvent system. The presence of triethylamine is crucial as it helps maintain the stability of the catalyst and neutralizes any acidic byproducts that might form during the reaction. Detailed standard operating procedures regarding catalyst activation, filtration techniques, and vacuum rectification parameters are essential for achieving the demonstrated laboratory yields of nearly 80% on a scalable basis.
- Preparation of N-Benzyl-3-Hydroxypyridine Quaternary Ammonium Salt by reacting 3-hydroxypyridine with benzyl chloride in toluene at 90-110°C.
- Catalytic hydrogenation of the quaternary salt using a diatomite-supported Ni-B-Co catalyst in ethanol/triethylamine at 3-5 atm hydrogen pressure and 70°C.
- Workup involving filtration, solvent recovery, extraction with ethyl acetate, and high-vacuum rectification to obtain the final product with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates directly into tangible strategic advantages regarding cost structure and operational reliability. The elimination of noble metals removes a significant variable cost component that is subject to volatile global market pricing, stabilizing the long-term cost projection for this intermediate. Additionally, the shift from 100 atm to 3-5 atm operating pressure allows manufacturers to utilize standard, off-the-shelf hydrogenation equipment rather than custom-engineered high-pressure vessels, significantly lowering capital expenditure barriers for new production lines. This accessibility enhances supply chain resilience by enabling a broader base of qualified contract manufacturing organizations (CMOs) to produce the material without requiring specialized high-risk infrastructure.
- Cost Reduction in Manufacturing: The replacement of expensive platinum or palladium catalysts with a low-cost nickel-cobalt-boron system results in substantial raw material savings per kilogram of product. Since the catalyst is supported on diatomite, it can be filtered, washed, and potentially recycled multiple times, further amortizing the catalyst cost over larger production batches. The moderate pressure requirements also lead to significantly reduced energy consumption for compression and heating, contributing to a lower overall utility cost profile for the manufacturing facility.
- Enhanced Supply Chain Reliability: By utilizing readily available commodity chemicals such as benzyl chloride and 3-hydroxypyridine, the process avoids reliance on scarce or geopolitically sensitive specialty reagents. The robustness of the catalyst against deactivation ensures consistent batch-to-batch performance, minimizing the risk of production delays caused by failed runs or extended reaction times. This reliability is critical for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time inventory schedules.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst simplifies the separation process, eliminating the need for complex aqueous workups often required for homogeneous catalysts. This leads to a significant reduction in wastewater generation and solvent usage, aligning the process with increasingly stringent environmental regulations. The low heavy metal residue in the final product (<0.1 ppm) reduces the burden on quality control labs and ensures compliance with ICH guidelines for elemental impurities, facilitating faster regulatory approval for the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on safety, purity, and scalability concerns. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer or process optimization projects.
Q: How does the new nickel-based catalyst compare to traditional Raney Nickel in terms of safety?
A: Unlike traditional Raney Nickel which requires extremely high hydrogen pressures around 100 atm and poses significant explosion risks, the novel Ni-B-Co catalyst operates safely at moderate pressures of 3-5 atm, drastically reducing equipment costs and operational hazards.
Q: What is the expected purity and heavy metal residue of the final product?
A: The patented process demonstrates the ability to achieve product purity exceeding 99% (GC) with minimal side reactions such as debenzylation. Furthermore, the heavy metal residue is controlled to extremely low levels, typically less than 0.1 ppm, meeting stringent pharmaceutical standards.
Q: Is the catalyst reusable and how does it impact production costs?
A: Yes, the catalyst is supported on diatomite, which facilitates easy filtration and recovery. This reusability, combined with the elimination of expensive noble metals like platinum or palladium, leads to substantial reductions in raw material costs and waste disposal expenses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Benzyl-3-Piperidinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient production of life-saving antihypertensive medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and secure. We are committed to delivering N-Benzyl-3-Piperidinol with stringent purity specifications and rigorous QC labs testing every batch to guarantee consistency. Our facility is equipped to handle the specific requirements of this nickel-catalyzed process, providing a stable and compliant source for your global supply chain needs.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your current sourcing strategy. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to be your long-term strategic partner in pharmaceutical intermediate manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
