Revolutionizing Benidipine Intermediate Production via Novel Nickel-Based Catalysis
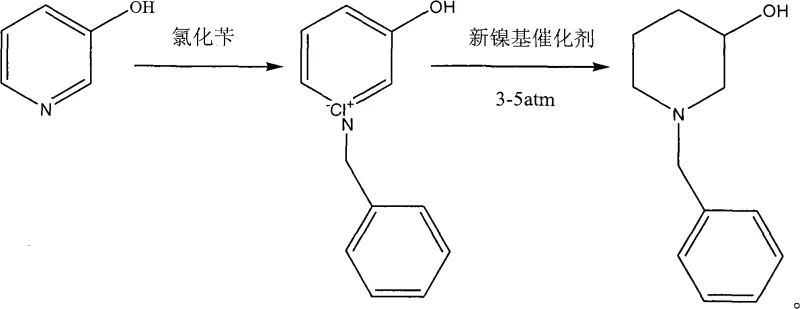
The pharmaceutical industry constantly seeks robust synthetic pathways that balance economic efficiency with operational safety, particularly for critical antihypertensive intermediates. Patent CN101817779A introduces a groundbreaking methodology for the production of N-benzyl-3-piperidinol, a pivotal building block in the synthesis of Benidipine, a third-generation calcium channel blocker. This innovation addresses the longstanding dichotomy between the high costs associated with noble metal catalysis and the severe safety hazards posed by traditional high-pressure hydrogenation methods. By utilizing a specifically engineered nickel-cobalt-boron catalyst supported on diatomite, the process achieves complete hydrogenation of the pyridine ring under remarkably mild conditions of 3 to 5 atmospheres. This technical breakthrough not only ensures high product purity exceeding 99 percent but also fundamentally alters the economic landscape for manufacturers seeking a reliable pharmaceutical intermediates supplier capable of delivering complex heterocyclic structures at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-benzyl-3-piperidinol has been plagued by significant technical and economic bottlenecks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. The traditional route involving Raney Nickel catalysts necessitates extreme hydrogen pressures approaching 100 atmospheres, requiring specialized, exorbitantly expensive autoclaves and posing substantial explosion risks due to the pyrophoric nature of the catalyst. Alternatively, routes employing noble metal catalysts such as platinum dioxide or rhodium on carbon, while operating at lower pressures, incur prohibitive raw material costs that erode profit margins. Furthermore, standard non-noble metal catalysts often suffer from poor selectivity, leading to the cleavage of the benzyl group (debenzylation) during the reduction phase, which drastically reduces yield and complicates downstream purification processes. These legacy methods represent a fragile supply chain vulnerable to safety incidents and volatile metal pricing, failing to meet modern standards for green and sustainable chemical production.
The Novel Approach
The patented methodology circumvents these obstacles by reversing the conventional synthetic logic, prioritizing the formation of the quaternary ammonium salt prior to the reduction step. Instead of reducing 3-hydroxypyridine first and then alkylating, this novel approach reacts 3-hydroxypyridine with benzyl chloride to generate N-benzyl-3-hydroxypyridinium chloride, which is subsequently hydrogenated. This strategic sequence, coupled with the unique composition of the new nickel-based catalyst, prevents the unwanted removal of the benzyl protecting group. The result is a streamlined process that operates at a mere 3 to 5 atmospheres of hydrogen pressure, dramatically lowering the capital expenditure required for reactor equipment. This shift enables the commercial scale-up of complex pharmaceutical intermediates with enhanced safety profiles and substantially reduced operational expenditures, making it an ideal solution for large-volume production facilities.
Mechanistic Insights into Ni-Co-B/Diatomite Catalytic Hydrogenation
The core of this technological advancement lies in the precise formulation of the heterogeneous catalyst, which consists of nickel, cobalt, and boron supported on a diatomite matrix. The inclusion of cobalt acts as a promoter that modifies the electronic state of the nickel active sites, enhancing the adsorption of the pyridine ring while simultaneously weakening the interaction that typically leads to C-N bond cleavage of the benzyl group. The boron component, introduced via sodium borohydride reduction during catalyst preparation, creates an amorphous alloy structure that offers a high density of active surface area. This specific metallurgical architecture allows for the effective saturation of the aromatic pyridine nucleus to the piperidine ring under moderate thermal and pressure conditions, specifically around 70 degrees Celsius. Such mechanistic control is critical for maintaining the structural integrity of the N-benzyl moiety, ensuring that the final product retains the necessary functionality for subsequent esterification into Benidipine.
Furthermore, the support material, diatomite, plays a crucial role in the physical handling and recovery of the catalyst, addressing one of the major pain points in heterogeneous catalysis. Unlike fine powder catalysts that are difficult to filter and prone to loss, the diatomite-supported particles are robust and easily separable from the reaction mixture via simple filtration. This physical stability facilitates catalyst recycling, allowing the active metal species to be reused across multiple batches without significant loss of activity or selectivity. From a quality control perspective, this minimizes the risk of metal leaching into the final product, ensuring that heavy metal residues remain well below the stringent thresholds required for API intermediates. The combination of electronic promotion by cobalt and physical stabilization by diatomite creates a synergistic effect that delivers consistent high-purity output batch after batch.
How to Synthesize N-benzyl-3-piperidinol Efficiently
The execution of this synthesis involves a two-stage process beginning with the quaternization of 3-hydroxypyridine in a toluene solvent system at elevated temperatures between 90 and 110 degrees Celsius. Following the isolation of the quaternary ammonium salt, the material is subjected to hydrogenation in an alcoholic medium containing triethylamine as a stabilizer. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and workup procedures for maximizing yield and purity, are outlined in the structured guide below.
- React 3-hydroxypyridine with benzyl chloride in toluene at 90-110°C to form the N-benzyl-3-hydroxypyridinium quaternary ammonium salt.
- Prepare the novel catalyst by loading nickel acetate and cobalt chloride onto diatomite, followed by reduction with sodium borohydride.
- Perform catalytic hydrogenation of the quaternary salt at 3-5 atm hydrogen pressure and 70°C using the new nickel-based catalyst to yield the final piperidinol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates directly into tangible operational improvements and risk mitigation strategies. The transition from high-pressure noble metal systems to a moderate-pressure nickel-based system eliminates the need for specialized high-specification reactors, thereby reducing both capital investment and maintenance overheads. Additionally, the reliance on abundant base metals like nickel and cobalt insulates the production cost from the extreme volatility often seen in the platinum and palladium markets. This stability allows for more accurate long-term budgeting and pricing contracts, fostering stronger relationships between suppliers and downstream pharmaceutical manufacturers who demand consistency.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals such as platinum and rhodium results in a drastic decrease in raw material costs per kilogram of finished product. Moreover, the ability to recycle the diatomite-supported catalyst multiple times further amortizes the catalyst cost over a larger production volume, leading to substantial overall savings. The moderate pressure requirements also reduce energy consumption associated with compression and heating, contributing to a leaner and more cost-effective manufacturing footprint that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: By removing the dependency on scarce noble metals and hazardous high-pressure operations, the supply chain becomes significantly more resilient to disruptions. The use of readily available base metals ensures that raw material sourcing is stable and less susceptible to geopolitical constraints or mining shortages. Furthermore, the improved safety profile reduces the likelihood of unplanned plant shutdowns due to safety incidents, guaranteeing a continuous and reliable flow of high-purity intermediates to meet tight production schedules for antihypertensive medications.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, with the solid-supported catalyst facilitating easy separation and minimizing waste generation. The absence of toxic heavy metal residues in the final product simplifies the purification workflow and reduces the burden on wastewater treatment facilities. This alignment with green chemistry principles not only lowers environmental compliance costs but also positions the manufacturer favorably in an increasingly regulated global marketplace that prioritizes sustainable and eco-friendly production methodologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on performance metrics and operational parameters.
Q: Why is the novel nickel-based catalyst superior to Raney Nickel for this synthesis?
A: Unlike Raney Nickel which requires dangerous high pressures (around 100 atm) and poses explosion risks, the novel nickel-cobalt-boron catalyst operates safely at moderate pressures (3-5 atm) while preventing the undesired debenzylation side reaction.
Q: What is the purity profile of N-benzyl-3-piperidinol produced via this method?
A: The process yields product with exceptional purity, typically exceeding 99% as determined by Gas Chromatography (GC), with heavy metal content controlled to extremely low levels (<0.1ppm).
Q: How does this route impact the cost of Benidipine manufacturing?
A: By replacing expensive noble metal catalysts (like Platinum or Rhodium) with a low-cost nickel-based alternative and reducing equipment pressure requirements, the overall manufacturing cost is significantly lowered without compromising yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-benzyl-3-piperidinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the nickel-catalyzed reduction of pyridinium salts are translated into reality with precision. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of N-benzyl-3-piperidinol meets the exacting standards required for Benidipine synthesis, guaranteeing consistency and reliability for our global partners.
We invite you to collaborate with us to leverage these technological advantages for your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
