Advanced Tridentate Phosphine Ligands for High-Efficiency Olefin Alkoxycarbonylation
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more atom-economical and selective catalytic processes. A significant breakthrough in this domain is documented in patent CN116003467A, which discloses a novel class of benzene-based triphosphine compounds. These ligands represent a paradigm shift in olefin alkoxycarbonylation, a reaction critical for producing high-value organic carboxylic acid esters. Unlike traditional bidentate systems, these tridentate architectures offer exceptional stability and activity when complexed with palladium precursors. The general reaction scheme involves the coupling of olefins, carbon monoxide, and alcohols to generate esters with one additional carbon atom, a transformation that is foundational for synthesizing key intermediates in the pharmaceutical and polymer industries.

For R&D directors focused on process optimization, the structural integrity of the catalyst is paramount. Conventional methods often rely on bidentate phosphine ligands, such as the widely known dtbpx (1,2-bis(di-tert-butylphosphinomethyl)benzene) developed by Lucite. While effective, these systems can sometimes suffer from limited turnover numbers or require harsh conditions to maintain activity. Furthermore, ferrocene-based ligands like butphos, while offering high TONs, introduce complexity in synthesis and potential stability issues under certain acidic conditions required for the reaction. The quest for a ligand that balances high activity, exceptional selectivity, and synthetic accessibility has led to the development of the novel structures described in this patent.
The novel approach introduced in CN116003467A centers on a unique tridentate phosphine architecture defined by Formula (I). This structure features a central benzene ring substituted with three phosphine groups linked via alkylene chains. This tridentate coordination mode creates a highly stable pocket around the palladium center, effectively preventing catalyst decomposition and suppressing the formation of branched byproducts. The steric bulk provided by substituents such as tert-butyl groups on the phosphorus atoms further enhances the linear selectivity of the insertion step. This structural innovation allows the catalytic system to operate under milder conditions while achieving conversion rates and selectivities that surpass many commercially available alternatives.
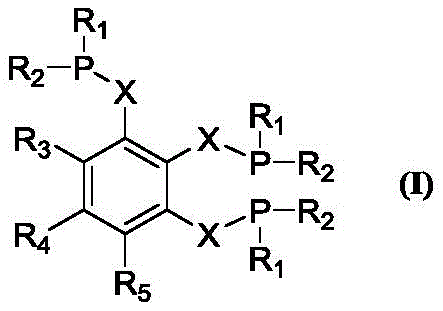
Mechanistically, the superiority of these triphosphine ligands lies in their ability to stabilize the active cationic palladium-hydride species throughout the catalytic cycle. In the alkoxycarbonylation of olefins, the rate-determining step often involves the migratory insertion of the olefin into the Pd-H bond. The rigid yet flexible backbone of the tridentate ligand ensures that the metal center remains coordinatively saturated enough to prevent aggregation into inactive palladium black, yet open enough to allow substrate binding. This delicate balance results in Turnover Numbers (TON) exceeding 60000 and Turnover Frequencies (TOF) approaching 20000 h⁻¹. Moreover, the electronic properties of the ligand can be finely tuned by varying the R groups on the phosphorus or the linker length, providing a versatile platform for optimizing specific substrate transformations.
Impurity control is another critical aspect where this technology excels. In the production of pharmaceutical intermediates, the presence of branched esters or oligomeric byproducts can complicate downstream purification significantly. The high linear-to-branched (l/b) ratio (>99%) achieved with these ligands minimizes the formation of iso-esters, thereby simplifying the distillation or crystallization steps required to isolate the target product. This high selectivity is attributed to the specific spatial arrangement of the three phosphine arms, which sterically hinders the formation of the branched alkyl-palladium intermediate. Consequently, manufacturers can achieve higher purity specifications with fewer processing units, directly impacting the overall cost of goods sold.
How to Synthesize 1,2,3-Tri(di-tert-butylphosphinomethyl)benzene Efficiently
The synthesis of these high-performance ligands is designed for scalability and operational simplicity, addressing a common bottleneck in ligand manufacturing. The patent outlines a robust two-step strategy that avoids the use of exotic reagents or cryogenic conditions typically associated with sensitive organophosphorus chemistry. The process begins with the activation of an aromatic core, such as 1,2,3-trimethylbenzene, using a strong base or metal reagent to generate a nucleophilic intermediate. This intermediate is then trapped with a chlorophosphine electrophile to forge the crucial carbon-phosphorus bonds. The detailed standardized synthesis steps for producing these ligands are provided in the guide below.
- Prepare the organometallic intermediate by reacting a substituted aromatic compound (such as 1,2,3-trimethylbenzene) with a strong base or metal reagent like n-butyllithium or magnesium powder in an inert solvent.
- React the generated organometallic intermediate with a chlorophosphine compound (e.g., di-tert-butylchlorophosphine) at controlled temperatures to form the phosphorus-carbon bonds.
- Quench the reaction mixture, separate the organic phase, and purify the crude product through recrystallization using methanol or similar solvents to obtain high-purity white crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel catalytic technology translates into tangible strategic advantages beyond mere technical performance. The primary value driver is the drastic reduction in catalyst loading enabled by the high TON values. Since palladium is a precious metal with volatile pricing, minimizing the amount of metal required per kilogram of product significantly lowers the raw material cost base. Furthermore, the high selectivity reduces the burden on waste treatment facilities, as fewer byproducts mean less solvent consumption for purification and lower disposal costs for hazardous waste streams.
- Cost Reduction in Manufacturing: The implementation of this triphosphine ligand system allows for a substantial decrease in operating expenses through multiple mechanisms. By achieving TONs greater than 60000, the consumption of the expensive palladium precursor is minimized, directly lowering the variable cost per unit of production. Additionally, the simplified downstream processing resulting from high selectivity reduces energy consumption associated with distillation and separation columns. The elimination of complex purification steps also decreases the requirement for specialized solvents and adsorbents, contributing to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The synthetic route for these ligands relies on commodity chemicals such as trimethylbenzene and di-tert-butylchlorophosphine, which are readily available from multiple global suppliers. This diversification of raw material sources mitigates the risk of supply disruptions that often plague specialty chemical manufacturing. The robustness of the synthesis, which tolerates a range of temperatures and solvents, ensures consistent batch-to-batch quality, allowing production planners to maintain reliable inventory levels without the need for excessive safety stock.
- Scalability and Environmental Compliance: The process described in the patent is inherently scalable, having been demonstrated in multi-gram to kilogram scales without loss of efficiency. The use of standard organic solvents and the absence of highly toxic reagents simplify the environmental permitting process for new production lines. Moreover, the high atom economy of the alkoxycarbonylation reaction itself, combined with the efficient catalyst, aligns with modern green chemistry principles, helping companies meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system. These answers are derived directly from the experimental data and embodiments disclosed in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their specific applications.
Q: How does this novel triphosphine ligand improve upon existing bidentate ligands like dtbpx?
A: According to patent CN116003467A, the novel benzene-based triphosphine ligands demonstrate significantly higher Turnover Numbers (TON > 60000) and superior linear-to-branched (l/b) selectivity (>99%) compared to traditional bidentate systems, leading to reduced catalyst loading and fewer byproducts.
Q: Is the synthesis of these ligands scalable for industrial production?
A: Yes, the patent outlines a robust two-step synthesis method using readily available starting materials like 1,2,3-trimethylbenzene and chlorophosphines. The process utilizes standard solvents and manageable temperature ranges (-30°C to 180°C), facilitating easy commercial scale-up.
Q: What specific applications benefit most from this catalytic technology?
A: This technology is particularly valuable for the alkoxycarbonylation of olefins to produce organic carboxylic acid esters, such as methyl propionate, which serves as a critical intermediate for polymethyl methacrylate (PMMA) and various pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphosphine Ligand Supplier
As the demand for high-efficiency catalytic processes grows, partnering with an experienced CDMO is essential for successful technology transfer. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of ligand or intermediate meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to leverage our technical expertise to optimize your production costs and supply chain resilience. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific process needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced catalytic solutions can drive value for your organization.
