Advanced Fluralaner Intermediate Synthesis: A Scalable, Catalyst-Free Commercial Route
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective synthetic routes for high-value active ingredients. A significant breakthrough in this domain is detailed in patent CN113461653B, which discloses a novel method for preparing Fluralaner, a potent veterinary insecticide, and its key intermediates. This technology addresses critical bottlenecks in traditional manufacturing by replacing cryogenic conditions and expensive catalytic systems with a streamlined, mild-temperature process. For R&D directors and supply chain managers, this represents a pivotal shift towards more reliable pharmaceutical intermediates production. The core innovation lies in the strategic use of a Grignard reagent for nucleophilic addition, bypassing the need for complex chiral catalysts and ultra-low temperature reactors that have historically plagued the commercial scale-up of complex fluorinated compounds.
Furthermore, the patent outlines a comprehensive pathway that not only enhances chemical efficiency but also aligns with modern green chemistry principles. By eliminating hazardous oxidants like ozone and toxic sulfur compounds, the process mitigates environmental risks while maintaining high purity standards. This dual focus on operational simplicity and environmental compliance makes the technology particularly attractive for large-scale veterinary drugs manufacturing. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a superior alternative for securing the supply chain of this critical animal health product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Fluralaner relied heavily on academically elegant but industrially cumbersome routes. One prominent method, reported in literature such as Nat. Chem. 2016, utilized a nucleophilic addition reaction requiring a specialized, hard-to-synthesize catalyst and dimethoxy zinc to achieve stereocontrol. While effective in a laboratory setting, this approach presents severe challenges for commercial production. The requirement for ultra-low temperature reaction conditions, specifically maintaining temperatures around -78°C, demands significant energy investment and specialized cryogenic equipment that is often impractical for multi-ton manufacturing. Additionally, the reliance on ozone and dimethyl sulfide for oxidation steps introduces significant safety hazards and environmental disposal issues.
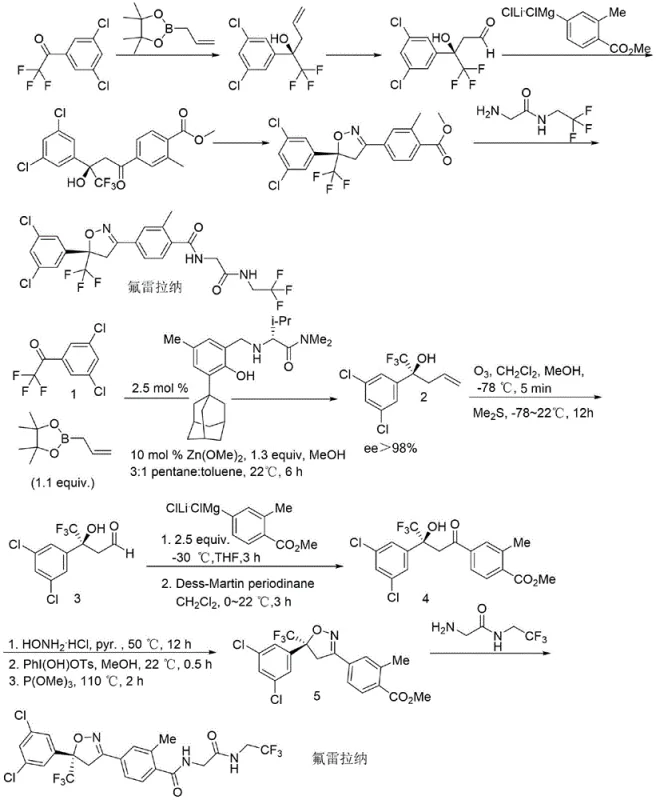
Moreover, the starting materials for these conventional routes, such as specific allyl boronic acid pinacol esters, are not readily available off-the-shelf and require separate synthesis, adding layers of complexity and cost to the supply chain. The low yields observed in key steps, such as the 1,3-dipolar addition cyclization in other reported routes, further exacerbate the economic inefficiency. These factors combined create a fragile production model where minor deviations in temperature or reagent quality can lead to batch failures, posing a substantial risk to supply continuity for global pharmaceutical partners.
The Novel Approach
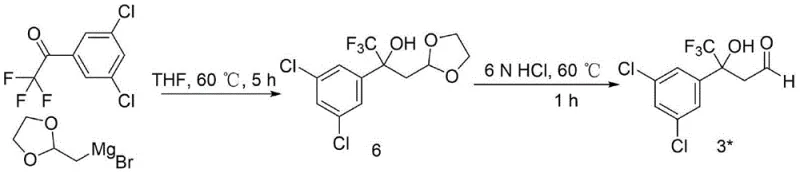
The method disclosed in patent CN113461653B fundamentally reengineers the synthesis to overcome these industrial barriers. The new route initiates with a nucleophilic addition reaction between 1-(3,5-dichlorophenyl)-2,2,2-trifluoroacetone and (1,3-dioxolan-2-ylmethyl)magnesium bromide. Crucially, this transformation proceeds efficiently in tetrahydrofuran at a moderate temperature of 60°C, completely eliminating the need for cryogenic cooling. This shift from -78°C to 60°C is not merely a numerical change; it represents a transition from a high-risk, energy-intensive operation to a standard, easily controllable chemical process suitable for any general-purpose reactor.
Furthermore, the novel approach dispenses with the expensive and difficult-to-source dimethoxy zinc and custom catalysts entirely. Instead, it leverages commercially available Grignard reagents, ensuring a stable and cost-effective raw material supply. The subsequent steps, including acidolysis, oxidation, and cyclization, are optimized to avoid hazardous reagents like ozone. By replacing these with safer alternatives such as Dess-Martin periodinane and hydroxyl(tosyloxy)iodobenzene, the process significantly reduces the environmental footprint. This streamlined workflow ensures that the production of high-purity Fluralaner intermediates is both economically viable and operationally robust.
Mechanistic Insights into Grignard-Mediated Nucleophilic Addition
The cornerstone of this improved synthesis is the initial nucleophilic addition step, which constructs the carbon skeleton necessary for the final isoxazoline ring. In this mechanism, the carbonyl group of 1-(3,5-dichlorophenyl)-2,2,2-trifluoroacetone acts as the electrophile, attacked by the nucleophilic carbon of the (1,3-dioxolan-2-ylmethyl)magnesium bromide Grignard reagent. The presence of the trifluoromethyl group adjacent to the carbonyl enhances the electrophilicity of the ketone, facilitating the addition even under mild thermal conditions. The dioxolane moiety serves as a protected form of the aldehyde, preventing unwanted side reactions during the Grignard addition and allowing for controlled deprotection in the subsequent acidolysis step.
Following the formation of the tertiary alcohol intermediate (Intermediate 6), the process employs a strategic acid-catalyzed hydrolysis to reveal the aldehyde functionality. This aldehyde is then subjected to a second Grignard addition with an aryl magnesium species, followed by oxidation using Dess-Martin periodinane to generate the key ketone precursor. The final cyclization to form the isoxazoline ring involves the formation of an oxime, followed by an oxidative coupling mediated by hydroxyl(tosyloxy)iodobenzene. This hypervalent iodine reagent facilitates the intramolecular N-O bond formation under mild conditions, avoiding the harsh oxidants typically required for such transformations. The final deoxygenation using trimethoxy phosphorus completes the heterocyclic core, setting the stage for the final amidation.
From an impurity control perspective, this mechanistic pathway offers distinct advantages. The use of protected intermediates minimizes the formation of polymerization byproducts often seen with free aldehydes. Furthermore, the specificity of the Dess-Martin oxidation and the hypervalent iodine cyclization ensures high regioselectivity, reducing the burden on downstream purification processes. For R&D teams, understanding these mechanistic nuances is vital for troubleshooting and optimizing the process further, ensuring that the final high-purity Fluralaner meets stringent regulatory specifications for veterinary use.
How to Synthesize Fluralaner Intermediate Efficiently
The synthesis of Fluralaner via this patented route involves a sequence of five distinct chemical transformations, each optimized for yield and operational simplicity. The process begins with the preparation of the protected alcohol intermediate, followed by deprotection, chain extension, cyclization, and final amide coupling. Detailed protocols for reaction times, temperatures, and workup procedures are critical for reproducibility. For a comprehensive guide on executing this synthesis in a pilot or production plant, please refer to the standardized operating procedures outlined below.
- Perform nucleophilic addition of 1-(3,5-dichlorophenyl)-2,2,2-trifluoroacetone with (1,3-dioxolan-2-ylmethyl)magnesium bromide in THF at 60°C to form Intermediate 6.
- Hydrolyze Intermediate 6 using 6N HCl at 60°C for 1 hour to generate the aldehyde Intermediate 3*.
- React Intermediate 3* with a Grignard reagent followed by Dess-Martin oxidation to yield ketone Intermediate 4*.
- Convert Intermediate 4* to isoxazoline Intermediate 5* via oxime formation, oxidative cyclization with PhI(OH)OTs, and deoxygenation.
- Complete the synthesis by amidating Intermediate 5* with 2-amino-N-(2,2,2-trifluoroethyl)acetamide to obtain Fluralaner.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into tangible business value. The primary advantage lies in the drastic simplification of the manufacturing infrastructure. By removing the requirement for ultra-low temperature reactors capable of sustaining -78°C, facilities can utilize standard glass-lined or stainless steel reactors equipped with conventional heating and cooling systems. This reduction in specialized equipment requirements lowers the barrier to entry for contract manufacturing organizations and reduces the capital expenditure needed for production lines.
- Cost Reduction in Manufacturing: The elimination of expensive and proprietary catalysts, such as the complex chiral ligands and dimethoxy zinc used in prior art, results in significant raw material cost savings. Additionally, the replacement of hazardous oxidants like ozone with safer, solid-state reagents reduces the costs associated with specialized gas handling systems and waste treatment. The overall simplification of the process flow leads to a more economical production cost structure without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available, commodity-grade reagents like Grignard reagents and common solvents ensures a stable supply chain. Unlike custom-synthesized catalysts that may have long lead times and single-source risks, the materials required for this process are readily accessible from multiple global suppliers. This diversification of the supply base mitigates the risk of production delays caused by raw material shortages, ensuring consistent delivery of veterinary drug intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gases like ozone make this process inherently safer and easier to scale from kilogram to metric ton quantities. The reduced environmental hazard profile simplifies regulatory compliance and waste disposal, lowering the operational overhead associated with environmental health and safety (EHS) management. This sustainability aspect is increasingly important for multinational corporations aiming to meet their green chemistry goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational benefits and chemical feasibility of the new route.
Q: How does this new synthesis route improve upon previous methods for Fluralaner?
A: Unlike prior art which required ultra-low temperatures of -78°C and expensive catalysts like dimethoxy zinc, this patented method operates at mild temperatures (60°C) without specialized catalysts, significantly simplifying industrial operations and reducing equipment costs.
Q: What are the environmental benefits of this manufacturing process?
A: The process eliminates the use of hazardous reagents such as ozone and dimethyl sulfide, which are common in older synthetic routes. This substitution results in a greener manufacturing profile with reduced toxic waste generation and improved operator safety.
Q: Is the raw material supply chain stable for this synthesis?
A: Yes, the route utilizes commercially available and inexpensive reagents like (1,3-dioxolan-2-ylmethyl)magnesium bromide, avoiding the reliance on difficult-to-source custom catalysts or unstable intermediates found in alternative pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluralaner Supplier
The technological advancements presented in patent CN113461653B highlight the potential for a more efficient and sustainable supply of Fluralaner intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes are optimized for maximum yield and purity. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the high standards required for veterinary pharmaceutical applications.
We invite potential partners to collaborate with us to leverage this cost-effective synthesis technology. By engaging with our technical team, you can receive a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects, ensuring a secure and competitive supply chain for your veterinary product portfolio.
