Advanced Palladium-Catalyzed Synthesis of Trifluoromethyl Indene-Indole Hybrids for Pharma
Advanced Palladium-Catalyzed Synthesis of Trifluoromethyl Indene-Indole Hybrids for Pharma
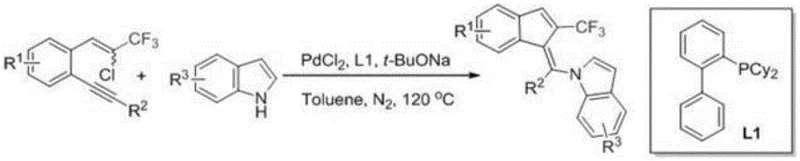
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex polycyclic scaffolds that incorporate fluorine atoms, given their profound impact on metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN109320446B, which discloses a highly efficient synthetic route for 2-trifluoromethylindenylmethylene indole derivatives. This technology addresses a critical gap in organic synthesis, as prior art lacked methods to seamlessly integrate both indene and indole moieties within a single molecular framework while simultaneously introducing a trifluoromethyl group. The disclosed method utilizes a palladium-catalyzed tandem cyclization strategy, transforming readily available 1-(2-chloro-3,3,3-trifluoroprop-1-en-1-yl)-2-(phenylethynyl)benzene derivatives and indoles into high-value intermediates. This innovation is particularly relevant for the development of new drug candidates, drawing inspiration from bioactive molecules like sulindac and ellipticine, where such fused ring systems play pivotal roles in therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of polycyclic compounds containing both indene and indole cores has been a formidable challenge for synthetic chemists, often requiring multi-step sequences that suffer from low overall yields and poor atom economy. Conventional approaches typically involve the separate synthesis of the indene skeleton followed by coupling reactions to attach the indole unit, or vice versa, which necessitates the isolation and purification of unstable intermediates. These stepwise processes not only increase the consumption of solvents and reagents but also generate substantial amounts of chemical waste, conflicting with modern green chemistry principles. Furthermore, introducing a trifluoromethyl group at a specific position on the indene ring usually demands harsh fluorinating agents or specialized precursors that are expensive and hazardous to handle. The lack of a unified strategy to assemble these fragments in a single operation has historically hindered the rapid exploration of structure-activity relationships for potential drug candidates containing this specific pharmacophore.
The Novel Approach
In stark contrast to these laborious traditional pathways, the novel approach described in the patent data leverages a sophisticated palladium-catalyzed cascade reaction to achieve the target architecture in a single pot. By employing 1-(2-chloro-3,3,3-trifluoroprop-1-en-1-yl)-2-(phenylethynyl)benzene as a multifunctional substrate, the method triggers an intramolecular cyclization followed by an intermolecular coupling with indole, effectively building two rings and forming a carbon-carbon double bond simultaneously. This tandem process eliminates the need for intermediate isolation, drastically simplifying the workflow and reducing the time required for synthesis. The use of standard palladium chloride coupled with bulky phosphine ligands allows the reaction to proceed under relatively mild thermal conditions (120°C) in common solvents like toluene. This streamlined methodology not only enhances the overall yield, reaching up to 76% in optimized examples, but also significantly improves the stereoselectivity, predominantly favoring the Z-isomer, which is crucial for the biological activity of the resulting derivatives.
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
Understanding the catalytic cycle is essential for R&D directors aiming to optimize this process for specific analogues. The reaction initiates with the in situ generation of the active Pd(0) species from the Pd(II) precursor under the influence of the phosphine ligand. This active catalyst undergoes oxidative addition with the vinyl chloride moiety of the substrate, forming a key organopalladium intermediate (Intermediate A). Subsequently, an intramolecular carbopalladation occurs where the palladium center adds across the triple bond of the phenylethynyl group, leading to the formation of the indene ring system (Intermediate B). This step is critical as it establishes the core carbocyclic framework. Following this cyclization, a ligand exchange takes place where the indole nitrogen coordinates to the palladium center, facilitated by the base sodium tert-butoxide, generating complex C. The cycle concludes with a reductive elimination step that forms the final exocyclic double bond connecting the indene and indole units, releasing the product and regenerating the Pd(0) catalyst for the next turnover.
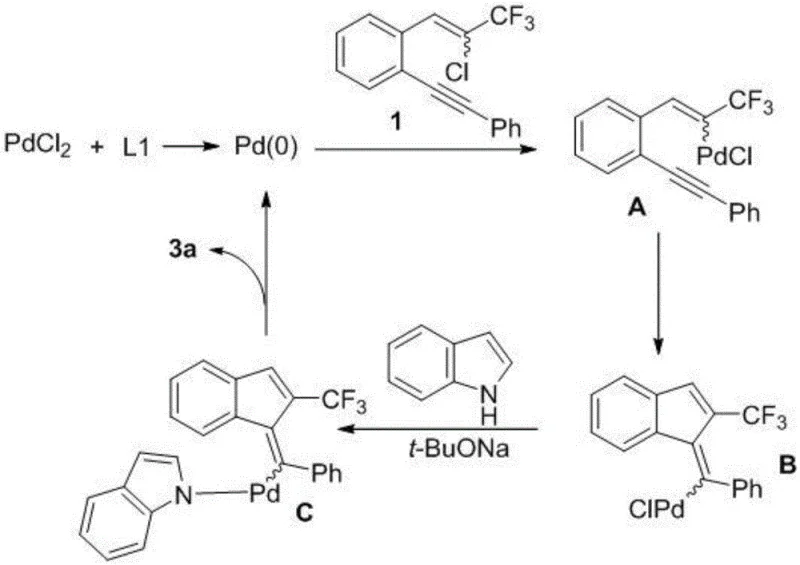
From an impurity control perspective, the mechanism offers inherent advantages that simplify downstream processing. The high regioselectivity of the intramolecular carbopalladation ensures that the trifluoromethyl group is positioned precisely at the 2-position of the indene ring, minimizing the formation of regioisomers that are difficult to separate. Additionally, the reaction exhibits excellent stereoselectivity, with Z/E ratios often exceeding 98:2, as evidenced by NMR analysis in the patent examples. This high level of stereocontrol reduces the burden on purification teams who would otherwise need to employ chiral chromatography or recrystallization to isolate the active isomer. The robustness of the catalytic system also means that side reactions such as homocoupling of the alkyne or dehalogenation are minimized, leading to a cleaner crude reaction profile. For process chemists, this translates to higher throughput and reduced solvent usage during the workup phase, directly impacting the cost of goods sold for the final active pharmaceutical ingredient.
How to Synthesize 2-Trifluoromethylindenylmethylene Indole Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The standard protocol involves charging a reaction vessel with the chloro-trifluoropropenyl benzene substrate and the chosen indole derivative in anhydrous toluene. To this mixture, palladium chloride (10 mol%) and the ligand 2-dicyclohexylphosphino biphenyl (20 mol%) are added, followed by sodium tert-butoxide as the base. The system is then heated to 120°C under a nitrogen atmosphere to prevent catalyst oxidation and stirred for approximately 24 hours. Upon completion, the reaction mixture is cooled, diluted with ethyl acetate, and washed with saturated brine to remove inorganic salts. The organic layer is dried over anhydrous sodium sulfate and concentrated under vacuum, with the final product purified via flash column chromatography. This procedure has been validated across a wide range of substrates, demonstrating its versatility for generating diverse libraries of compounds.
- Mix 1-(2-chloro-3,3,3-trifluoroprop-1-en-1-yl)-2-(phenylethynyl)benzene substrate with indole or pyrrole derivative in toluene solvent.
- Add palladium chloride catalyst (10 mol%), 2-dicyclohexylphosphino biphenyl ligand, and sodium tert-butoxide base (2 equivalents).
- Stir the mixture at 120°C under nitrogen atmosphere for 24 hours, then purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology presents compelling economic and logistical benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By consolidating what would traditionally be a multi-step synthesis into a single tandem reaction, manufacturers can significantly lower labor costs, energy consumption, and equipment occupancy time. The elimination of intermediate isolation steps means fewer unit operations are required, reducing the risk of material loss and cross-contamination. Furthermore, the use of commodity chemicals such as palladium chloride, toluene, and sodium tert-butoxide ensures that raw material sourcing is stable and cost-effective, mitigating the risks associated with supply chain disruptions for exotic reagents. This reliability is crucial for maintaining continuous production schedules in a high-demand pharmaceutical environment.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis inherently drives down manufacturing costs by minimizing solvent usage and waste generation. Since the reaction proceeds with high atom economy and does not require expensive fluorinating agents or specialized catalysts beyond standard palladium sources, the direct material costs are kept low. The high yields reported, reaching up to 76%, ensure that raw material input is efficiently converted into valuable product, reducing the cost per kilogram of the intermediate. Additionally, the simplified workup procedure reduces the volume of wastewater and organic waste that requires treatment, leading to substantial savings in environmental compliance and disposal fees. These cumulative efficiencies make the process highly competitive for large-scale commercial production.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials enhances the resilience of the supply chain against market volatility. Unlike processes that depend on custom-synthesized building blocks with long lead times, the substrates for this reaction, such as substituted phenylacetylenes and indoles, are commercially accessible from multiple global suppliers. This diversity in sourcing options allows procurement teams to negotiate better pricing and secure backup supplies easily. Moreover, the robustness of the reaction conditions, which tolerate a variety of functional groups without needing protection-deprotection strategies, means that the process is less sensitive to minor variations in raw material quality. This tolerance reduces the frequency of batch failures and ensures a consistent output of high-purity intermediates, supporting reliable delivery commitments to downstream clients.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is exceptionally well-suited for transition from laboratory to pilot and commercial plant scales. The reaction operates at atmospheric pressure and moderate temperatures, removing the need for high-pressure reactors or cryogenic cooling systems, which simplifies engineering requirements and capital investment. The low toxicity of the reagents and the minimal generation of hazardous byproducts align perfectly with increasingly stringent environmental regulations. The high selectivity of the reaction reduces the need for extensive chromatographic purification, which is often a bottleneck in scaling up fine chemical processes. Consequently, manufacturers can achieve faster time-to-market for new drug candidates while maintaining a smaller environmental footprint, a key metric for modern sustainable chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived from the detailed experimental data and background provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for pharmaceutical intermediate production. Understanding these nuances helps stakeholders make informed decisions about process integration and resource allocation.
Q: What are the key advantages of this Pd-catalyzed synthesis method?
A: The method offers a novel one-pot tandem cyclization that constructs both indene and indole skeletons simultaneously. It features high atom utilization, simple operation without special instruments, and achieves yields up to 76% with excellent Z/E stereoselectivity.
Q: What substrates are compatible with this trifluoromethyl indene synthesis?
A: The process demonstrates broad substrate universality. It tolerates various substituents on the benzene ring (methyl, methoxy, fluoro, chloro) and works with diverse indole derivatives (3-methyl, 5-methoxy, 5-fluoro, etc.) as well as pyrrole substrates.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the protocol uses commercially available reagents like PdCl2 and standard solvents like toluene. The reaction proceeds under normal pressure without specialized equipment, making it highly scalable for industrial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethylindenylmethylene Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN109320446B for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop concept to industrial reality. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. We are committed to delivering high-purity 2-trifluoromethylindenylmethylene indole derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your supply chain and reduce overall project costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in developing novel fluorinated therapeutics.
