Advanced Solid Phase Synthesis of Octreotide: Overcoming Racemization in Peptide Alcohol Manufacturing
Introduction to Next-Generation Octreotide Synthesis
The pharmaceutical landscape for peptide therapeutics is constantly evolving, driven by the need for higher purity and more efficient manufacturing processes. A pivotal advancement in this domain is documented in patent CN100335497C, which introduces a novel solid-phase synthesis technology for Octreotide, a potent somatostatin analogue widely used in treating acromegaly and neuroendocrine tumors. Traditional methods for synthesizing peptide alcohols like Octreotide often struggle with complex purification steps and the risk of racemization, particularly at the C-terminal end. This patent addresses these critical technical bottlenecks by employing a specialized 3,4-dihydropyran hydroxymethyl resin (DHP HM Resin) as the carrier. By shifting away from reduction excision methods, this technology ensures the stereochemical integrity of the molecule is maintained throughout the synthesis. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards more reliable pharmaceutical intermediates with superior quality profiles.
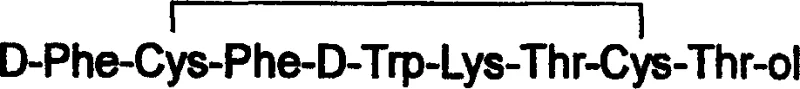
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Octreotide has relied heavily on liquid-phase segment condensation or solid-phase methods utilizing reduction excision strategies. Liquid-phase synthesis, while effective for small batches, is plagued by lengthy preparation times and the necessity for extensive purification after every single step, leading to significant solvent waste and environmental concerns. Furthermore, traditional solid-phase approaches often employ linking agents that require reduction for cleavage. This reduction step is notoriously problematic; it frequently induces racemization, compromising the optical purity of the final API. Documents cited in the background art indicate that isolated yields for these older methods were often suboptimal, ranging from as low as 3.1% to 14%, creating substantial inefficiencies in cost reduction in pharmaceutical manufacturing. The inability to consistently produce high-purity material without complex downstream processing has long been a pain point for supply chain managers seeking reliable sources.
The Novel Approach
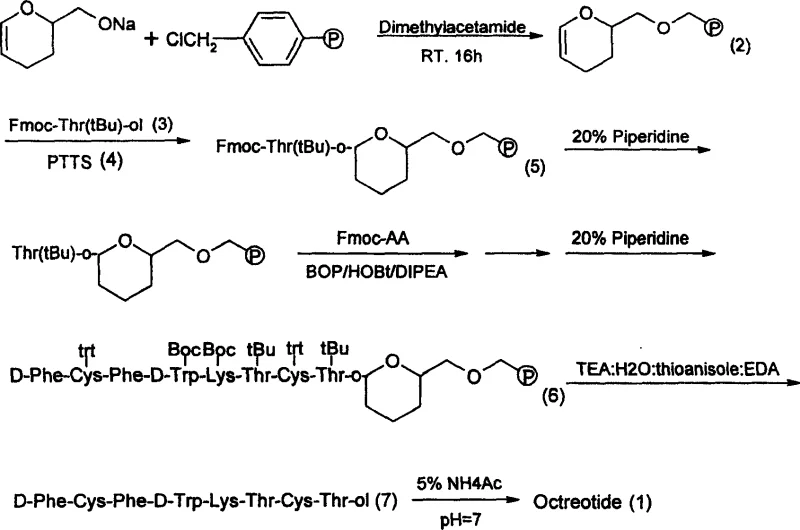
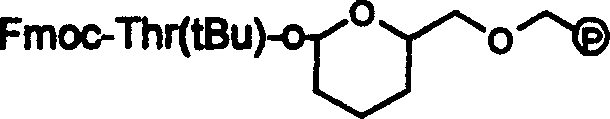
The methodology outlined in patent CN100335497C offers a transformative solution by utilizing a chloromethyl resin modified with 2-hydroxymethyl-3,4-dihydropyran to create the DHP HM Resin. This novel linker is stable under the alkaline conditions required for Fmoc/tBu solid-phase peptide synthesis but can be cleaved under relatively mild acidic conditions using a trifluoroacetic acid (TFA) cocktail. This eliminates the need for harsh reduction agents, thereby solving the technical problem of racemization entirely. The process involves coupling Fmoc-Thr(tBu)-ol to the resin, followed by the sequential addition of amino acids using standard activation reagents like BOP and HOBt. The result is a streamlined workflow that not only simplifies operations but also significantly enhances the crude purity of the peptide prior to final purification. This approach facilitates the commercial scale-up of complex peptide intermediates by providing a robust and reproducible pathway that minimizes side reactions.
Mechanistic Insights into DHP Linker Stability and Acidolysis
The core innovation of this synthesis lies in the chemical behavior of the tetrahydropyranyl methyl (THP) linkage formed between the resin and the C-terminal threoninol. Mechanistically, the formation of this acetal-like linkage is catalyzed by pyridinium p-toluenesulfonate (PPTS), ensuring a secure attachment that withstands the repetitive basic deprotection cycles (using 20% piperidine) inherent to Fmoc chemistry. Unlike ester linkers that might be prone to premature cleavage or base-catalyzed racemization, the THP ether linkage remains inert until the final cleavage step. Upon exposure to the TFA:water:thioanisole:ethanedithiol cocktail, the linker undergoes acidolysis. This reaction mechanism is gentle enough to preserve the chiral center of the threoninol residue, which is critical for the biological activity of Octreotide. The avoidance of nucleophilic attack or redox conditions during cleavage is the key differentiator that sets this method apart from prior art, ensuring that the final product retains its specific conformational requirements for receptor binding.
Furthermore, the impurity profile generated by this method is significantly cleaner compared to reduction-based excision. In traditional methods, side products arising from incomplete reduction or over-reduction can be difficult to separate from the target peptide. In contrast, the acidolytic cleavage of the DHP linker produces well-defined byproducts that do not interfere with the subsequent air oxidation step required to form the intramolecular disulfide bond between the two cysteine residues. The patent data indicates that the crude product obtained after cleavage possesses an HPLC purity of 57.1%, which is remarkably high for a crude peptide of this complexity. Following air oxidation in ammonium acetate buffer, the purity further improves to 87.5%. This high level of crude purity reduces the burden on preparative HPLC, lowering solvent consumption and increasing overall throughput, a crucial factor for reducing lead time for high-purity peptide APIs.
How to Synthesize Octreotide Efficiently
The synthesis of Octreotide via this solid-phase route is designed for operational simplicity and scalability. The process begins with the preparation of the functionalized resin, followed by the stepwise assembly of the octapeptide chain using protected amino acids. The final stages involve cleavage, oxidation, and purification. While the general workflow is straightforward, precise control over reaction conditions—such as temperature, reagent equivalents, and washing protocols—is essential to maximize yield and purity. The patent provides detailed embodiments that serve as a blueprint for manufacturing, demonstrating that the method is viable not just on a gram scale but also in larger batch sizes. For technical teams looking to implement this, the standardized use of Fmoc chemistry means that existing peptide synthesis infrastructure can be readily adapted. Detailed standardized synthesis steps see the guide below.
- Preparation of DHP HM Resin by reacting chloromethyl resin with sodium hydride and 2-hydroxymethyl-3,4-dihydropyran in dimethylacetamide.
- Coupling of Fmoc-Thr(tBu)-ol to the resin using PPTS catalyst, followed by sequential Fmoc/tBu solid-phase peptide synthesis.
- Cleavage of the peptide from the resin using a TFA-based cocktail, followed by air oxidation to form the disulfide bond and HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solid-phase synthesis technology translates into tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the robustness of the supply chain for raw materials and the efficiency of the production process. By utilizing a resin system that avoids exotic or hazardous reduction reagents, the process relies on widely available commodity chemicals like TFA, thioanisole, and standard Fmoc-amino acids. This availability mitigates the risk of supply disruptions caused by the scarcity of specialized reagents. Moreover, the elimination of the reduction step simplifies the workflow, reducing the number of unit operations required. Fewer processing steps inherently mean lower labor costs, reduced equipment occupancy time, and decreased energy consumption, all of which contribute to a more favorable cost structure for the final API.
- Cost Reduction in Manufacturing: The economic impact of this synthesis route is driven by the significant improvement in crude purity and the elimination of complex purification burdens. Because the acidolytic cleavage avoids racemization, the amount of diastereomeric impurities is drastically reduced. In peptide manufacturing, separating diastereomers is often the most expensive and yield-limiting step in downstream processing. By preventing their formation in the first place, the process achieves substantial cost savings in chromatography media and solvent usage. Additionally, the high yield of the crude peptide (reported as 91.7% in the patent examples) ensures that the input cost of expensive protected amino acids is maximized, minimizing waste and optimizing the overall material cost per kilogram of finished Octreotide.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical oncology and endocrine drugs is paramount. This synthesis method enhances reliability by decoupling production from the limitations of liquid-phase synthesis, which is difficult to scale. Solid-phase synthesis is inherently modular and easier to parallelize or scale up using larger reactors. The patent explicitly confirms that large-scale compound experiments yielded good results, validating the scalability of the process. This means that suppliers utilizing this technology can respond more flexibly to surges in demand without compromising quality. The use of stable intermediates and a robust resin linker also extends the shelf-life of key pre-clinical materials, allowing for better inventory management and reducing the risk of batch failures due to reagent degradation.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a cleaner alternative to traditional methods. Liquid-phase synthesis generates vast quantities of organic solvent waste due to the need for extraction and crystallization at every step. In contrast, solid-phase synthesis confines the reaction to the resin bead, allowing for simple filtration and washing, which significantly reduces solvent volume. Furthermore, avoiding heavy metal catalysts or toxic reducing agents simplifies the waste treatment process and ensures compliance with stringent environmental regulations regarding heavy metal residuals in pharmaceuticals. This 'green' aspect is increasingly important for multinational corporations aiming to meet sustainability goals while maintaining a steady supply of compliant active ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the solid-phase synthesis of Octreotide. These insights are derived directly from the experimental data and technical specifications provided in the patent literature. Understanding these nuances is essential for stakeholders evaluating the feasibility of this manufacturing route for commercial production. The answers reflect the consensus on the superiority of the DHP linker strategy over conventional reduction-based methods.
Q: How does the DHP HM Resin linker prevent racemization during Octreotide synthesis?
A: The novel 3,4-dihydropyran hydroxymethyl (DHP HM) linker allows for cleavage under mild acidic conditions (TFA cocktail) rather than harsh reduction methods. This preserves the stereochemical integrity of the C-terminal threoninol, effectively eliminating the racemization issues common in traditional reduction excision methods.
Q: What are the purity levels achievable with this solid-phase synthesis method?
A: According to the patent data, the crude peptide obtained after cleavage demonstrates an HPLC purity of approximately 57.1%. Following air oxidation to form the critical disulfide bond, the purity increases significantly to around 87.5% before final preparative HPLC purification, indicating a highly efficient process with manageable impurity profiles.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the patent explicitly details successful large-scale compound experiments alongside lab-scale synthesis. The use of standard Fmoc/tBu chemistry and robust resin linkers ensures that the process is amenable to scale-up, offering consistent yields and quality suitable for industrial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octreotide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with state-of-the-art peptide synthesis reactors and rigorous QC labs capable of handling the stringent purity specifications required for oncology and endocrine APIs. We understand that the synthesis of Octreotide demands meticulous control over stereochemistry and impurity profiles, and our team is dedicated to delivering products that meet the highest global regulatory standards.
We invite you to collaborate with us to leverage this advanced solid-phase synthesis technology for your Octreotide requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our optimized processes can enhance your bottom line. We encourage potential partners to contact us to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to driving efficiency and quality in your pharmaceutical supply chain.
