Advanced Octreotide Purification Technology for Commercial Scale API Production
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and yield of complex polypeptide therapeutics while minimizing environmental footprints. As detailed in patent CN109942678B, a significant technological breakthrough has been achieved in the refining of octreotide, a critical synthetic polypeptide used in the treatment of acromegaly and gastrointestinal disorders. This innovative approach leverages a one-step reversed-phase chromatography system that integrates enrichment, salt conversion, and purification into a single continuous workflow. By utilizing super water-resistant fillers and optimizing mobile phase compositions, this method addresses the longstanding challenges of impurity removal and solvent waste management inherent in traditional polypeptide processing. For R&D directors and procurement specialists, understanding this shift is vital for securing a reliable octreotide supplier capable of meeting stringent regulatory and sustainability standards. The transition from multi-step batch processing to this streamlined continuous flow represents a paradigm shift in how high-value peptide APIs are manufactured at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for polypeptide drugs often rely on a fragmented sequence of medium-low pressure chromatography followed by high-pressure refinement, a process fraught with inefficiencies and high operational costs. Conventional methods typically necessitate the use of molecular sieve gel columns or ultrafiltration membranes, which are often ill-suited for target polypeptides with molecular weights around 1 kDa, such as octreotide, leading to low sample capacity and sluggish flow rates. Furthermore, the reliance on organic solvents for sample loading and elution in standard reversed-phase chromatography generates substantial volumes of hazardous waste liquid, imposing heavy burdens on waste treatment facilities and escalating overall production expenses. The inability to effectively control pH during salt conversion steps in older methodologies can also compromise product stability, resulting in lower yields and inconsistent quality profiles that fail to meet the rigorous demands of modern pharmaceutical supply chains. These structural inefficiencies create bottlenecks that hinder the commercial scale-up of complex pharmaceutical intermediates and increase the lead time for high-purity polypeptides.
The Novel Approach
In stark contrast, the novel refining method introduces a cohesive one-step protocol that seamlessly combines reversed-phase enrichment, salt conversion, and purification within a single elution process. This approach utilizes a specialized super water-resistant filler that allows the mobile phases during the critical balance, loading, and salt conversion stages to be predominantly aqueous solutions, drastically reducing the reliance on volatile organic compounds. By integrating these steps, the process eliminates the need for intermediate isolation and multiple column transfers, thereby minimizing product loss and enhancing the overall removal rate of impurities from the crude octreotide solution. The strategic use of specific mobile phase gradients ensures that the target polypeptide is retained and purified with exceptional precision, while the aqueous nature of the initial effluents allows for direct recycling or simplified sewage treatment. This innovation not only optimizes the production process for cost reduction in pharmaceutical manufacturing but also aligns with global environmental compliance standards, offering a sustainable pathway for the commercial production of vital peptide therapeutics.
Mechanistic Insights into Reversed-Phase Chromatography Refining
The core of this technological advancement lies in the sophisticated interaction between the octreotide molecule and the super water-resistant stationary phase, which facilitates high-resolution separation under mild conditions. The process begins with the oxidation of reduced octreotide crude product, where precise pH control between 7.0 and 9.0 is maintained using alkaline substances to ensure the formation of the correct disulfide bonds without degrading the sensitive polypeptide structure. The crude solution is then introduced to the chromatographic column packed with UniSil ODS-AQ filler, where hydrophobic interactions drive the adsorption of the polypeptide onto the stationary phase during the enrichment stage. This mechanism allows for the concentration of dilute crude solutions without the need for prior evaporation or precipitation, preserving the integrity of the bioactive molecule. The subsequent gradient elution carefully modulates the ratio of acetic acid and acetonitrile to selectively desorb impurities before collecting the target fraction, ensuring that the final product meets the stringent purity specifications required for clinical applications.
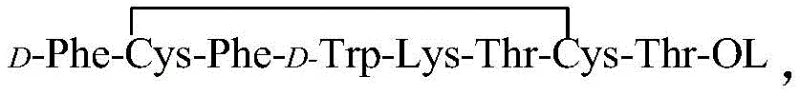
Impurity control is further enhanced by the specific design of the elution gradient, which separates the salt conversion and purification phases to prevent the co-elution of unwanted byproducts. During the salt conversion phase, a weak base mobile phase effectively removes trifluoroacetate ions and other acidic impurities that may have accumulated during the solid-phase synthesis and cleavage steps. The use of a super water-resistant filler with a defined pore diameter of 7-10 nm and particle size of 10 μm ensures optimal flow dynamics and mass transfer, allowing for high throughput without compromising resolution. This precise engineering of the chromatographic environment minimizes the risk of polypeptide degradation, which is particularly critical given octreotide's instability under high pH or prolonged exposure to harsh solvents. The result is a highly pure product with a significantly reduced impurity profile, demonstrating the method's capability to deliver high-purity pharmaceutical intermediates consistently.
How to Synthesize Octreotide Efficiently
The implementation of this refining protocol requires a systematic approach to column preparation and mobile phase management to ensure reproducibility and efficiency at scale. The process begins with the dynamic axial compression of the super water-resistant filler into the preparative column, establishing a stable bed height that can withstand the pressures of continuous operation. Operators must carefully prepare the crude octreotide solution by dissolving the reduced crude product in acetic acid and water, followed by oxidation with hydrogen peroxide under controlled pH conditions to generate the feed solution. The chromatographic system is then equilibrated, and the enrichment, salt conversion, and purification steps are executed sequentially using the predefined gradient profile, with fractions collected based on UV detection at 220 nm. Detailed standardized synthesis steps see the guide below.
- Prepare the crude octreotide solution by oxidizing the reduced octreotide crude product synthesized via solid phase.
- Load the solution onto a preparative column packed with super water-resistant filler (UniSil ODS-AQ).
- Execute sequential reversed-phase enrichment, salt conversion, and purification using aqueous mobile phases.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refining technology translates into tangible operational benefits that extend beyond mere technical specifications. The elimination of extensive organic solvent usage during the enrichment and salt conversion stages leads to substantial cost savings by reducing the volume of hazardous waste that requires specialized disposal. This shift towards aqueous-based processing not only lowers the environmental compliance burden but also mitigates the risks associated with the storage and handling of large quantities of flammable organic solvents in a manufacturing facility. Furthermore, the continuous nature of the one-step process enhances supply chain reliability by reducing the overall cycle time and minimizing the potential for human error during intermediate handling steps. These factors collectively contribute to a more resilient and cost-effective supply chain for critical peptide APIs.
- Cost Reduction in Manufacturing: The process achieves significant economic optimization by removing the need for expensive transition metal catalysts and reducing the consumption of high-grade organic solvents. By utilizing aqueous mobile phases for the initial stages of purification, the facility can drastically cut down on the costs associated with solvent procurement, recovery, and waste treatment. The high removal rate of impurities in a single pass also reduces the need for reprocessing batches, thereby improving overall yield and resource utilization. This streamlined approach ensures that the cost of goods sold is minimized without compromising the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the super water-resistant filler and the simplicity of the one-step protocol contribute to a more stable and predictable production schedule. With fewer unit operations and a reduced reliance on complex multi-column setups, the risk of equipment failure or process deviation is significantly lowered, ensuring consistent delivery timelines. The ability to process dilute crude solutions directly without concentration steps also allows for greater flexibility in upstream synthesis scheduling, reducing bottlenecks and enhancing the overall agility of the manufacturing supply chain. This reliability is crucial for maintaining the continuity of supply for essential medications.
- Scalability and Environmental Compliance: The method is inherently designed for industrial scale-up, utilizing standard preparative chromatography systems that can be easily expanded to meet increasing demand. The reduction in hazardous waste generation aligns with strict environmental regulations, facilitating easier permitting and reducing the likelihood of regulatory interruptions. The aqueous effluent can be directly subjected to sewage treatment and recycling, supporting sustainability goals and reducing the facility's environmental footprint. This compliance advantage positions the manufacturer as a preferred partner for global pharmaceutical companies seeking sustainable sourcing options.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced octreotide refining technology. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for stakeholders evaluating this process. Understanding these details is essential for making informed decisions about technology adoption and supplier qualification. The information below clarifies the operational parameters and quality outcomes associated with this method.
Q: How does the new refining method reduce environmental impact?
A: The process utilizes aqueous mobile phases during enrichment and salt conversion stages, significantly reducing the generation of hazardous organic waste liquid compared to traditional methods.
Q: What is the purity level achievable with this chromatography technique?
A: The method achieves high purity levels, with HPLC analysis showing results exceeding 99.5% for the refined octreotide product.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the one-step continuous production design and use of robust packing materials make it highly scalable for industrial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octreotide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of peptide manufacturing, leveraging advanced refining technologies like the one described in CN109942678B to deliver superior quality products. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the rigorous demands of global pharmaceutical markets. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of octreotide meets the highest standards of safety and efficacy. Our commitment to continuous improvement and process optimization allows us to offer competitive solutions that balance cost, quality, and sustainability for our partners.
We invite you to collaborate with us to optimize your supply chain and secure a stable source of high-quality peptide intermediates. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your manufacturing efficiency. Let us help you navigate the complexities of peptide production with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
