Advanced Resolution Technology for S-(+)-Ketoprofen: Scalable Manufacturing and Cost Optimization
The pharmaceutical industry continuously seeks robust methodologies for producing single-enantiomer drugs, particularly for non-steroidal anti-inflammatory drugs (NSAIDs) where the eutomer exhibits superior efficacy and safety profiles. Patent CN102010327B introduces a groundbreaking resolution method for (±)-2-(3-benzoyl)-phenylpropionic acid, commonly known as Ketoprofen, addressing critical limitations in existing chiral separation technologies. This innovation focuses on the isolation of S-(+)-Ketoprofen, the biologically active isomer responsible for the therapeutic effects, while minimizing the toxicological risks associated with the racemate. The core of this technology lies in the utilization of a novel, cost-effective chiral amine resolving agent that facilitates the formation of diastereomeric salts with high selectivity. By shifting away from expensive enzymatic processes or complex synthetic routes, this patent offers a pathway to high-purity API intermediates that is both economically viable and chemically efficient. The structural integrity of the target molecule, S-(+)-2-(3-benzoyl)-phenylpropionic acid, is preserved throughout the mild reaction conditions, ensuring minimal degradation and high overall yield.
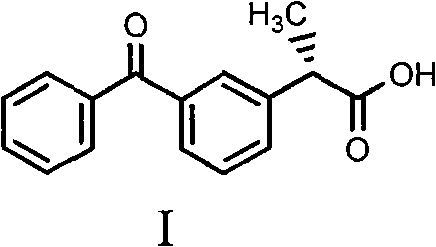
For R&D directors evaluating process feasibility, the chemical architecture of the target compound is paramount. As illustrated in the structure above, the presence of the chiral center at the alpha-position relative to the carboxylic acid group dictates the biological activity. The patent specifically targets the S-(+) configuration, which has been clinically proven to possess potent analgesic and anti-inflammatory properties without the severe gastrointestinal and renal side effects often attributed to the R-(-) enantiomer or the racemic mixture. Achieving high enantiomeric excess in this scaffold is not merely a regulatory requirement but a critical safety imperative. The described method leverages specific intermolecular interactions between the carboxylic acid moiety of the substrate and the amine group of the resolving agent to differentiate between the enantiomers during the crystallization phase. This precision in molecular recognition is the cornerstone of the process's success, allowing for the production of high-purity pharmaceutical intermediates that meet global pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure Ketoprofen has been plagued by significant technical and economic hurdles that hinder efficient commercial manufacturing. Traditional asymmetric synthesis routes, such as those employing Sharpless epoxidation followed by hydrogenolysis, involve multiple reaction steps that cumulatively result in low overall yields and exorbitant production costs. Furthermore, enzymatic kinetic resolution methods, while highly selective, rely on lipase preparations that are prohibitively expensive and difficult to source in the quantities required for industrial-scale operations. Previous chemical resolution attempts using resolving agents like N-octyl-D-glucosamine or complex chiral amino acid derivatives have suffered from poor resolution efficiency, often yielding single-round separation rates as low as 40% or failing to achieve optical purities exceeding 97%. These inefficiencies translate directly into increased waste generation, higher solvent consumption, and extended processing times, creating a bottleneck for supply chain reliability. Additionally, many prior art resolving agents are structurally complex, requiring multi-step syntheses themselves, which further compounds the cost burden and introduces additional points of failure in the manufacturing process.
The Novel Approach
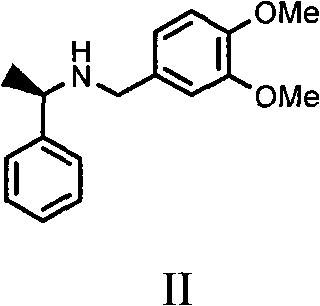
In stark contrast to these legacy technologies, the method disclosed in CN102010327B utilizes (R)-3,4-dimethoxy-N-(1-phenylethyl)-benzylamine as a resolving agent, representing a paradigm shift in cost-effective chiral separation. This specific amine is characterized by its structural simplicity and ease of synthesis, typically derived from readily available starting materials like 3,4-dimethoxybenzaldehyde and alpha-phenethylamine via a straightforward reductive amination. The novelty of this approach lies not only in the chemical identity of the resolver but also in its unprecedented application to the Ketoprofen substrate, where it demonstrates superior discrimination between enantiomers compared to previously tested amines. The process operates under mild conditions using common alcoholic solvents, eliminating the need for cryogenic temperatures or exotic catalysts. By optimizing the molar ratios and solvent systems, specifically employing isopropanol with a controlled amount of water, the method ensures the preferential crystallization of the desired diastereomeric salt. This results in a streamlined workflow that drastically reduces the number of unit operations, minimizes solvent waste, and significantly enhances the overall throughput of the production line, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks.
Mechanistic Insights into Diastereomeric Salt Formation
The success of this resolution strategy is fundamentally rooted in the stereoselective formation of diastereomeric salts, a process driven by the specific three-dimensional arrangement of the resolving agent. The resolving agent, (R)-3,4-dimethoxy-N-(1-phenylethyl)-benzylamine, possesses a rigid chiral framework augmented by electron-donating methoxy groups on the aromatic ring. These methoxy substituents play a crucial role in modulating the electronic environment and steric bulk around the amine nitrogen, thereby influencing the strength and geometry of the ionic bond formed with the carboxylate anion of the Ketoprofen enantiomers. When the racemic acid reacts with the chiral amine, two distinct diastereomeric salts are generated: one involving the S-(+) acid and the other involving the R-(-) acid. Due to the differences in their crystal lattice energies and solubility profiles, one salt—specifically the complex containing the desired S-(+) enantiomer—exhibits significantly lower solubility in the chosen solvent system. This differential solubility allows for the selective precipitation of the target salt upon cooling or antisolvent addition, effectively separating it from the mother liquor which retains the unwanted enantiomer.
Following the initial salt formation, the process incorporates a refinement step that further elevates the optical purity to exceptional levels. The crude diastereomeric salt can be recrystallized from alcoholic solvents, potentially with the addition of a small amount of fresh resolving agent to drive the equilibrium towards the desired crystal form. This iterative purification capability is a key advantage, as it allows manufacturers to tune the final enantiomeric excess to meet strict specifications, often achieving values of 99.8% ee or higher. Once the high-purity salt is isolated, the final step involves acid dissociation. Treatment with a strong mineral acid, such as sulfuric acid or hydrochloric acid, protonates the amine, breaking the ionic bond and releasing the free S-(+)-Ketoprofen into the organic phase. This acid-base extraction is a standard, robust unit operation that ensures the complete recovery of the product while allowing for the recycling of the valuable chiral amine from the aqueous layer. The entire mechanistic sequence is designed to maximize material efficiency and minimize impurity carryover, ensuring a clean final product profile.
How to Synthesize S-(+)-Ketoprofen Efficiently
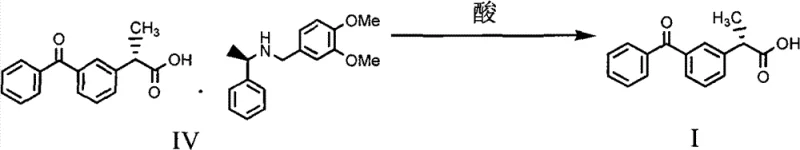
The practical implementation of this resolution technology involves a series of well-defined operational steps that balance reaction kinetics with crystallization thermodynamics. The process begins with the dissolution of the racemic substrate in a heated alcoholic solvent, followed by the controlled addition of the chiral resolving agent. The introduction of water as an antisolvent is a critical parameter that must be carefully managed to induce nucleation without causing oiling out or amorphous precipitation. Detailed protocols regarding temperature gradients, stirring rates, and aging times are essential to ensure the growth of large, filterable crystals that trap minimal mother liquor. For a comprehensive understanding of the specific operating parameters, including exact molar ratios and solvent volumes, please refer to the standardized synthesis guide below.
- React racemic (±)-2-(3-benzoyl)-phenylpropionic acid with the chiral resolving agent (R)-3,4-dimethoxy-N-(1-phenylethyl)-benzylamine in an alcoholic solvent such as isopropanol.
- Induce crystallization of the diastereomeric salt by adding water and cooling, followed by filtration to isolate the solid salt complex.
- Dissociate the purified salt using a mineral acid like sulfuric acid and extract the free S-(+)-Ketoprofen into an organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this resolution method offers transformative benefits that directly impact the bottom line and operational resilience. The primary driver of value is the substantial reduction in raw material costs achieved by replacing expensive, specialized resolving agents with a simple, commercially accessible amine. Unlike enzymatic resolvers that require cold chain logistics and have limited shelf lives, this chemical resolving agent is stable, easy to store, and can be sourced from multiple suppliers, thereby mitigating supply risk. Furthermore, the use of commodity solvents like isopropanol and sulfuric acid eliminates the need for hazardous or regulated reagents, simplifying waste disposal and reducing environmental compliance costs. The robustness of the crystallization process ensures consistent batch-to-batch quality, reducing the incidence of failed batches and the associated costs of rework or disposal. This reliability translates into shorter lead times for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of costly enzymes and complex chiral auxiliaries results in a drastic simplification of the bill of materials. By utilizing a resolving agent that is synthesized from inexpensive bulk chemicals, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the high recovery yield of the product from the salt dissociation step means that less starting material is wasted, further enhancing the economic efficiency of the process. The ability to recover and recycle the chiral amine from the aqueous waste stream adds another layer of cost savings, creating a closed-loop system that minimizes raw material consumption.
- Enhanced Supply Chain Reliability: The reliance on widely available chemical feedstocks ensures that production is not vulnerable to the supply bottlenecks often associated with niche biocatalysts or custom-synthesized chiral ligands. The process uses standard reactor equipment and filtration systems found in most multipurpose chemical plants, meaning that production can be easily scaled or transferred between facilities without significant capital investment. This flexibility strengthens the supply chain by allowing for diversified manufacturing locations and reducing dependency on single-source providers for critical reagents.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without losing efficiency or purity. The use of green solvents like isopropanol and the generation of minimal hazardous waste align with modern sustainability goals and regulatory requirements. The straightforward workup procedure reduces energy consumption associated with solvent removal and purification, contributing to a lower carbon footprint for the manufacturing operation. This environmental compatibility facilitates smoother regulatory approvals and enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What is the primary advantage of the resolving agent used in CN102010327B?
A: The resolving agent, (R)-3,4-dimethoxy-N-(1-phenylethyl)-benzylamine, is significantly cheaper and easier to synthesize compared to traditional enzymatic or complex amino acid-based resolving agents, offering a distinct cost advantage for large-scale production.
Q: What optical purity can be achieved with this resolution method?
A: The patented process demonstrates the capability to achieve an enantiomeric excess (ee%) of greater than 99.8%, meeting the stringent quality requirements for active pharmaceutical ingredients (APIs) intended for clinical use.
Q: Is this process suitable for industrial scale-up?
A: Yes, the method utilizes common solvents like isopropanol and standard unit operations such as crystallization and acid-base extraction, making it highly adaptable for commercial scale-up from pilot plants to multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(+)-Ketoprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality chiral intermediates that meet the rigorous demands of the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify enantiomeric excess and impurity profiles for every batch. Our facility is equipped to handle the specific solvent systems and crystallization requirements of this patented resolution method, guaranteeing a reliable supply of S-(+)-Ketoprofen that adheres to international quality standards.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral resolution can add value to your pharmaceutical development pipeline.
