Advanced Aqueous Resolution Technology for High-Purity S-(+)-Ketoprofen Intermediates
Advanced Aqueous Resolution Technology for High-Purity S-(+)-Ketoprofen Intermediates
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable methods for producing single-enantiomer active pharmaceutical ingredients (APIs), particularly for non-steroidal anti-inflammatory drugs (NSAIDs) where stereochemistry dictates efficacy and safety profiles. Patent CN107759466B introduces a groundbreaking resolution method for (±)-2-(3-benzoyl)-phenylpropionic acid, commonly known as Ketoprofen, specifically targeting the therapeutically superior S-(+) enantiomer. This technology represents a significant leap forward in chiral separation science by replacing hazardous organic solvents with water and drastically optimizing the stoichiometry of the resolving agent. For R&D directors and process chemists, this patent offers a compelling alternative to traditional enzymatic or chromatographic separations, providing a pathway to high-purity intermediates with reduced environmental footprint and enhanced economic viability for large-scale manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of S-(+)-Ketoprofen has been plagued by significant technical and economic hurdles associated with conventional resolution strategies. Traditional methods often rely heavily on expensive lipase enzymes for kinetic resolution, which, while selective, suffer from prohibitive costs and limited scalability due to enzyme stability issues and the requirement for specialized reaction conditions. Furthermore, classical chemical resolution techniques typically employ chiral amines in a 1:1 molar ratio with the racemic acid, necessitating the use of large quantities of costly resolving agents that must be recovered and recycled, adding complexity and energy consumption to the downstream processing. Many existing protocols also depend on volatile organic compounds (VOCs) as solvents, creating substantial safety hazards, environmental compliance burdens, and waste disposal challenges that conflict with modern green chemistry principles and increasing regulatory scrutiny on pharmaceutical manufacturing emissions.
The Novel Approach
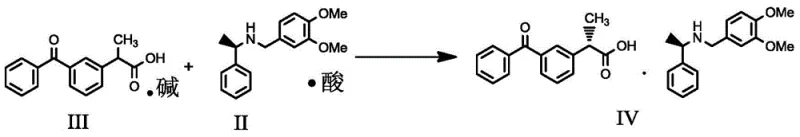
In stark contrast to these legacy processes, the methodology disclosed in CN107759466B utilizes a highly efficient diastereomeric salt formation strategy conducted entirely in an aqueous medium. The core innovation lies in the use of (R)-3,4-dimethoxy-N-(1-phenethyl)-benzylamine as a resolving agent, which exhibits exceptional selectivity for the S-(+) enantiomer of Ketoprofen when formulated as an acid salt. Crucially, this process operates with a sub-stoichiometric amount of the resolving agent, utilizing a molar ratio of substrate to resolving agent of approximately 1:0.5, which effectively halves the consumption of this critical chiral auxiliary compared to standard practices. The reaction proceeds smoothly in water at moderate temperatures between 50°C and 80°C, eliminating the need for flammable organic solvents and simplifying the isolation of the diastereomeric salt through straightforward filtration, thereby streamlining the entire production workflow.
Mechanistic Insights into Aqueous Diastereomeric Salt Crystallization
The success of this resolution process is fundamentally rooted in the precise thermodynamic and kinetic control of diastereomeric salt crystallization within an aqueous environment. When the racemic Ketoprofen sodium salt interacts with the chiral (R)-3,4-dimethoxy-N-(1-phenethyl)-benzylamine acetate in water, distinct diastereomeric salts are formed in situ. Due to the specific three-dimensional arrangement of the methoxy groups on the benzyl ring and the phenethyl moiety of the resolving agent, the salt formed with the S-(+) enantiomer of Ketoprofen possesses significantly lower solubility in hot water compared to its R-(-) counterpart. This differential solubility drives the selective precipitation of the S-(+) diastereomeric salt upon cooling or concentration, effectively pulling the equilibrium towards the desired product while leaving the unwanted R-(-) enantiomer in the mother liquor. The presence of water as the solvent enhances this selectivity by leveraging hydrogen bonding networks that stabilize the crystal lattice of the target diastereomer, a phenomenon that is often less pronounced in organic media.
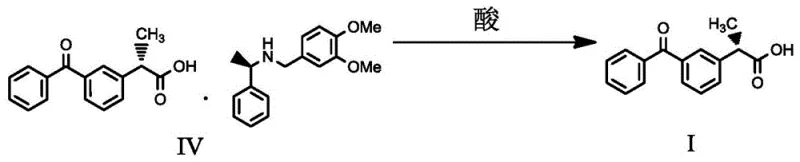
Following the isolation of the diastereomeric salt, the final step involves the liberation of the free acid through acidification, a process that must be carefully managed to maintain optical integrity. The addition of a strong inorganic acid, such as hydrochloric or sulfuric acid, protonates the carboxylate group of the Ketoprofen, breaking the ionic bond with the chiral amine and precipitating the S-(+)-Ketoprofen free acid. This step is critical for ensuring that no racemization occurs under acidic conditions, and the patent specifies controlled temperature ranges during acidification to prevent thermal degradation or epimerization. The resulting product demonstrates exceptional optical purity, with enantiomeric excess (ee) values consistently exceeding 99.8%, validating the robustness of the chiral recognition mechanism and the effectiveness of the crystallization parameters defined in the patent claims for producing pharmaceutical-grade intermediates.
How to Synthesize S-(+)-Ketoprofen Efficiently
The synthesis of S-(+)-Ketoprofen via this patented aqueous resolution route offers a streamlined protocol that balances high yield with operational simplicity, making it highly attractive for process development teams aiming to optimize manufacturing workflows. The procedure begins with the preparation of the resolving agent salt and the substrate salt separately in water, followed by their combination under controlled thermal conditions to induce selective crystallization of the target diastereomer. Detailed standardized operating procedures regarding mixing rates, seeding strategies, and filtration temperatures are essential to replicate the high purity and yield reported in the patent examples. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to implement this technology in a pilot or production setting, please refer to the structured guide below.
- Prepare the resolving agent salt by reacting (R)-3,4-dimethoxy-N-(1-phenethyl)-benzylamine with acetic acid in water.
- React the resolving agent salt with racemic ketoprofen sodium salt in water at 60-65°C to form the diastereomeric salt precipitate.
- Filter the diastereomeric salt and treat with a strong inorganic acid to liberate the pure S-(+)-Ketoprofen free acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this aqueous resolution technology presents a multitude of tangible benefits that directly address the pressing needs for cost containment and supply security in the pharmaceutical sector. By shifting the solvent system from expensive, regulated organic chemicals to ubiquitous and inexpensive water, manufacturers can achieve substantial reductions in raw material expenditures and waste treatment costs, while simultaneously mitigating the risks associated with solvent supply chain volatility. Furthermore, the drastic reduction in the loading of the chiral resolving agent—cutting the requirement by nearly half compared to conventional methods—translates into immediate and significant savings on one of the most costly inputs in the synthesis of chiral APIs, thereby improving the overall gross margin profile of the final active ingredient without compromising on quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of organic solvents and the optimized stoichiometry of the chiral auxiliary. Traditional resolution methods often require a 1:1 molar ratio of resolving agent to substrate, meaning that for every kilogram of product, an equivalent mass of expensive chiral amine is tied up in the process; by reducing this ratio to 0.5:1, the material cost for the resolving agent is effectively halved, leading to drastic improvements in the cost of goods sold (COGS). Additionally, the use of water removes the capital and operational expenses associated with solvent recovery distillation columns, explosion-proof infrastructure, and hazardous waste incineration, further enhancing the financial attractiveness of this route for large-scale commercial production.
- Enhanced Supply Chain Reliability: Relying on water as the primary reaction medium significantly de-risks the supply chain by removing dependence on petrochemical-derived solvents, which are subject to frequent price fluctuations and geopolitical supply disruptions. The resolving agent, (R)-3,4-dimethoxy-N-(1-phenethyl)-benzylamine, is synthesized from readily available starting materials, ensuring a stable and secure supply base that can support continuous manufacturing campaigns without the bottleneck risks often associated with specialized enzymes or exotic chiral ligands. This robustness ensures consistent lead times for high-purity API intermediates, allowing downstream drug product manufacturers to maintain reliable inventory levels and meet market demand without interruption.
- Scalability and Environmental Compliance: The inherent safety and simplicity of an aqueous process make it exceptionally well-suited for commercial scale-up, as it avoids the heat transfer limitations and safety hazards typical of exothermic reactions in organic solvents. From an environmental standpoint, the process aligns perfectly with green chemistry mandates by generating minimal hazardous waste and eliminating VOC emissions, which simplifies the permitting process for new manufacturing facilities and reduces the long-term liability associated with environmental compliance. This sustainability profile not only future-proofs the manufacturing asset against tightening regulations but also enhances the brand value of the final pharmaceutical product by appealing to increasingly eco-conscious stakeholders and healthcare providers.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this resolution technology into their existing portfolios, we have compiled a set of frequently asked questions based on the specific technical disclosures and experimental data found within the patent literature. These inquiries address common concerns regarding solvent compatibility, chiral purity thresholds, and the practical implications of the reduced resolving agent loading on process design. Understanding these nuances is critical for project managers and chemical engineers who are tasked with assessing the risk-reward profile of adopting new synthetic routes for critical pharmaceutical intermediates in a regulated environment.
Q: What are the primary advantages of using water as a solvent in this resolution process?
A: Using water eliminates the need for volatile and toxic organic solvents, significantly reducing environmental impact and solvent recovery costs while improving operational safety.
Q: How does this method improve cost efficiency compared to traditional resolution techniques?
A: The process utilizes a sub-stoichiometric amount of the resolving agent (approximately 0.5 equivalents), effectively halving the consumption of this expensive chiral auxiliary compared to standard 1:1 molar ratio methods.
Q: What level of optical purity can be achieved with this specific resolution technology?
A: The patented method consistently achieves an enantiomeric excess (ee) of greater than 99.8%, ensuring the production of high-purity S-(+)-Ketoprofen suitable for stringent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketoprofen Supplier
The technological advancements detailed in patent CN107759466B underscore the immense potential for optimizing the production of S-(+)-Ketoprofen, yet translating such laboratory-scale innovations into robust commercial reality requires a partner with deep process engineering expertise and state-of-the-art manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this domain, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of aqueous resolution are fully realized in practice. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of chiral intermediate meets the exacting standards required by global regulatory bodies, thus providing our partners with absolute confidence in the quality and consistency of their supply chain.
We invite procurement leaders and R&D strategists to engage with us to explore how this advanced resolution technology can be tailored to your specific production needs, offering a pathway to significant operational efficiencies and cost optimizations. By initiating a dialogue with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient process for your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive value and competitiveness in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
