Scalable Manufacturing of S-Pregabalin via Novel Nitro-Reduction Pathway for Global Pharma Supply
The pharmaceutical industry continuously seeks robust manufacturing routes for high-volume neurological agents, and patent CN101300224A presents a transformative approach to synthesizing (S)-Pregabalin, a critical active pharmaceutical ingredient. This specific intellectual property details a novel methodology that bypasses the severe safety limitations associated with traditional asymmetric hydrogenation techniques requiring high-pressure carbon monoxide. By leveraging a nitro-diester precursor strategy, the disclosed technology enables a safer, more controllable reaction environment that is inherently better suited for rigorous quality control standards demanded by global regulatory bodies. The strategic shift away from toxic cyanide reagents and extreme pressure conditions represents a significant evolution in process chemistry, directly addressing the growing need for sustainable and safe pharmaceutical intermediates production. For procurement leaders and technical directors, understanding this underlying chemical architecture is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality without supply chain disruptions caused by hazardous material handling restrictions.
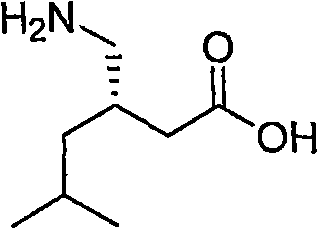
Historically, the manufacturing landscape for gabapentinoids has been constrained by methodologies that introduce substantial operational risks and complexity at the commercial scale. Conventional approaches, such as those utilizing rhodium-catalyzed asymmetric hydrogenation, often necessitate the use of carbon monoxide at elevated pressures, creating significant engineering bottlenecks and safety liabilities for production facilities. Furthermore, alternative routes involving the conjugate addition of hydrogen cyanide to unsaturated imides rely on reagents that are acutely toxic and require specialized containment infrastructure, drastically increasing capital expenditure and operational overhead. These legacy methods also frequently suffer from issues related to catalyst recovery and the formation of difficult-to-remove metal impurities, which can compromise the final purity profile of the active ingredient. In contrast, the novel approach detailed in the reference patent utilizes a nitro-reduction pathway that operates under much milder conditions, typically employing standard hydrogenation catalysts like palladium on carbon or Raney nickel at pressures ranging from 1 to 5 atmospheres. This fundamental shift in reaction design not only mitigates safety risks but also simplifies the downstream processing requirements, facilitating a more streamlined workflow for cost reduction in API manufacturing while maintaining high stereochemical integrity.
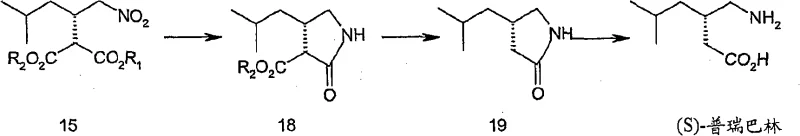
The mechanistic elegance of this synthesis lies in the selective reduction of the nitro group within the diester framework, followed by a carefully controlled decarboxylation sequence that preserves the crucial chiral center. The process initiates with the hydrogenation of the nitro-diester precursor, where the nitro functionality is efficiently converted to a primary amine without affecting the ester groups, yielding a stable amino-diester intermediate. Subsequent thermal treatment, often facilitated by copper salts or alkali halides in polar aprotic solvents, induces a selective mono-decarboxylation that sets the stage for the final structural arrangement. This step is critical because it avoids the racemization risks often associated with harsh basic or acidic conditions found in older resolution-based methods. The reaction kinetics are managed through precise temperature control, typically maintaining the mixture between 60°C and 100°C, which ensures complete conversion while minimizing the formation of thermal degradation byproducts. Understanding these mechanistic nuances allows R&D teams to optimize impurity profiles and ensure that the resulting high-purity Pregabalin meets the stringent specifications required for central nervous system therapeutics.
To implement this synthesis efficiently, technical teams must adhere to a standardized protocol that maximizes yield while ensuring operator safety and environmental compliance throughout the production cycle. The procedure begins with the preparation of the reaction vessel under an inert atmosphere, followed by the careful addition of the nitro-diester substrate and the chosen hydrogenation catalyst in a suitable alcohol solvent. Once the reduction phase is complete and the intermediate is isolated, the subsequent decarboxylation step requires precise monitoring of temperature and reaction time to prevent over-reaction or side-product formation. Detailed standard operating procedures for each unit operation, from filtration of the spent catalyst to the final crystallization of the product, are essential for maintaining batch-to-batch consistency. Adhering to these validated steps ensures that the commercial scale-up of complex pharmaceutical intermediates proceeds smoothly, minimizing the risk of costly batch failures or deviations that could impact supply continuity.
From a commercial perspective, the adoption of this nitro-reduction pathway offers profound advantages for procurement and supply chain management teams focused on long-term stability and cost efficiency. The elimination of high-pressure carbon monoxide and toxic cyanide reagents removes the need for expensive specialized containment systems and reduces the regulatory burden associated with handling hazardous materials, leading to substantial operational cost savings. Furthermore, the use of commodity chemicals such as ethanol, methanol, and common metal catalysts enhances supply chain resilience by reducing dependency on scarce or geopolitically sensitive reagents. The milder reaction conditions also translate to lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and aligning with modern sustainability goals in chemical manufacturing. By simplifying the purification process through effective crystallization and ion exchange techniques, manufacturers can achieve higher overall yields and reduce waste generation, further driving down the cost per kilogram of the final active ingredient. These factors collectively enhance the reliability of the supply chain, ensuring that partners can depend on a reliable pharmaceutical intermediates supplier to meet demanding production schedules without compromise.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive high-pressure reactor infrastructure required for carbon monoxide hydrogenation, significantly lowering capital investment and maintenance costs for production facilities. By utilizing widely available catalysts like palladium on carbon and Raney nickel, the method reduces raw material expenses and simplifies catalyst recovery protocols. The streamlined workflow minimizes the number of isolation steps, which decreases solvent consumption and waste disposal fees associated with complex multi-step syntheses. Additionally, the improved safety profile reduces insurance premiums and regulatory compliance costs, contributing to a more favorable overall economic model for large-scale production.
- Enhanced Supply Chain Reliability: Relying on common solvents and reagents that are readily available in the global chemical market mitigates the risk of supply disruptions caused by shortages of specialized precursors. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor variations in temperature or pressure compared to highly sensitive asymmetric catalysis methods. This flexibility enables suppliers to respond more quickly to fluctuations in market demand, ensuring consistent availability of critical intermediates for downstream API production. Moreover, the reduced hazard classification of the reagents simplifies logistics and transportation, allowing for faster and more cost-effective delivery to manufacturing sites worldwide.
- Scalability and Environmental Compliance: The mild operating parameters facilitate easy translation from laboratory bench scale to multi-ton commercial production without the need for significant process re-engineering or equipment modification. The absence of highly toxic byproducts simplifies effluent treatment processes, ensuring compliance with increasingly stringent environmental regulations regarding wastewater discharge and hazardous waste disposal. Efficient solvent recovery systems can be integrated seamlessly into the process flow, promoting a circular economy approach and minimizing the environmental impact of the manufacturing operation. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently asked questions regarding this technology often center on the specific impurity profiles and the regulatory acceptance of the novel intermediates generated during the synthesis. Technical stakeholders require assurance that the new pathway does not introduce genotoxic impurities or difficult-to-clear metal residues that could delay regulatory filings or complicate validation efforts. The data indicates that the selective nature of the decarboxylation step effectively controls the formation of regio-isomers, while the final ion exchange purification ensures that metal levels are well below ICH Q3D guidelines. Addressing these technical queries proactively helps build confidence among quality assurance teams and facilitates smoother technology transfer processes between development and commercial manufacturing units.
- Catalytic hydrogenation of the nitro-diester precursor using Pd/C or Raney Nickel under mild pressure to form the amino-diester intermediate.
- Thermal treatment with copper salts or alkali halides in polar solvents to induce selective decarboxylation and cyclization.
- Final hydrolysis and purification via ion exchange resin or crystallization to isolate pharmaceutical grade S-Pregabalin.
Frequently Asked Questions (FAQ)
Q: How does this process improve safety compared to conventional Pregabalin synthesis?
A: This method eliminates the need for high-pressure carbon monoxide and highly toxic hydrogen cyanide reagents used in older asymmetric hydrogenation or conjugate addition routes, significantly reducing operational hazards and waste treatment costs.
Q: What are the key impurities controlled in this nitro-reduction pathway?
A: The process utilizes specific thermal decarboxylation conditions and ion exchange purification to effectively remove over-reduced byproducts and residual metal catalysts, ensuring stringent purity specifications suitable for API manufacturing.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the reaction conditions operate at mild temperatures between 25°C and 100°C and moderate hydrogen pressures of 1 to 5 atmospheres, making it highly adaptable for multi-ton scale-up without requiring specialized high-pressure reactor infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Pregabalin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN101300224A to ensure the consistent supply of high-quality neurological therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging state-of-the-art facilities equipped to handle hydrogenation and thermal decarboxylation safely and efficiently. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of S-Pregabalin meets the exacting standards required by global pharmaceutical regulators. Our commitment to technical excellence ensures that we can navigate the complexities of chiral synthesis and impurity control, providing our partners with a secure and dependable source of critical active ingredients.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific supply chain requirements and cost optimization goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our implementation of this novel pathway can reduce your overall procurement costs while enhancing supply security. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to engineer a supply solution that drives value and innovation in your pharmaceutical development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
