Advanced Synthesis of Urea Derivatives via Carbonate Activation for Commercial Scale-up
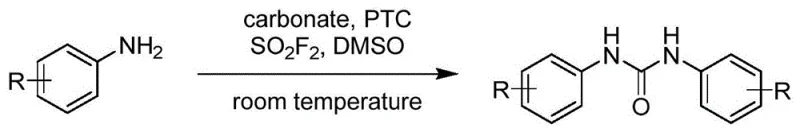
The landscape of organic synthesis is constantly evolving towards safer, more sustainable, and cost-effective methodologies, particularly for high-volume chemical building blocks. A significant breakthrough in this domain is documented in patent CN114213286B, which discloses a novel synthetic method for urea derivatives. This technology represents a paradigm shift from traditional hazardous routes to a mild, efficient process utilizing sodium carbonate as a C1 carbonyl synthon. By activating sodium carbonate with sulfuryl fluoride (SO2F2) in the presence of a phase transfer catalyst, this method enables the direct coupling of amines to form urea derivatives under remarkably mild conditions. For R&D directors and procurement managers in the fine chemical and pharmaceutical sectors, this innovation offers a compelling alternative to legacy processes, promising enhanced safety profiles and streamlined manufacturing workflows without compromising on yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of urea derivatives has relied heavily on highly reactive and dangerous intermediates such as phosgene (carbonyl chloride) or isocyanates. These traditional reagents pose severe safety hazards due to their toxicity and instability, often necessitating specialized equipment and rigorous containment protocols that drive up capital expenditure. Furthermore, the high reactivity of phosgene frequently leads to the formation of numerous side products, complicating the downstream purification process and reducing overall atom economy. More recent alternatives involving carbon monoxide (CO) or carbon dioxide (CO2) have attempted to mitigate some safety concerns but introduce new challenges, typically requiring expensive transition metal catalysts like palladium, ruthenium, or nickel, along with specialized ligands. These metal-catalyzed processes often demand high temperatures and pressures, increasing energy consumption and introducing the risk of heavy metal contamination in the final product, which is a critical quality attribute for pharmaceutical intermediates.
The Novel Approach
In stark contrast to these conventional limitations, the methodology described in patent CN114213286B utilizes sodium carbonate, a ubiquitous and inexpensive inorganic salt, as the carbonyl source. The core innovation lies in the activation of this stable carbonate by SO2F2, which generates a reactive species capable of coupling with amines at room temperature (25-30°C). This approach completely eliminates the need for toxic phosgene and expensive transition metal catalysts, thereby drastically simplifying the reaction setup and reducing raw material costs. The reaction conditions are exceptionally mild, operating effectively in common polar aprotic solvents like DMSO or DMF. Moreover, the process demonstrates excellent functional group tolerance, accommodating a wide range of aromatic, heterocyclic, and aliphatic amines. The simplicity of the workup, often involving merely pouring the reaction mixture into water to precipitate the product, underscores the operational efficiency of this novel route, making it highly attractive for large-scale commercial adoption.
Mechanistic Insights into SO2F2-Mediated Carbonylation
The mechanistic pathway of this transformation is both elegant and efficient, centering on the activation of the carbonate anion. In the presence of a phase transfer catalyst such as 15-crown-5, the sodium carbonate becomes soluble and reactive in the organic phase. The introduction of SO2F2 acts as a powerful activator, likely forming a transient fluorosulfonate or similar activated carbonate intermediate that possesses sufficient electrophilicity to react with the nucleophilic amine. This activation step is crucial as it bypasses the thermodynamic stability of the carbonate ion without requiring harsh thermal conditions. The reaction proceeds through a nucleophilic attack by the amine on the activated carbonyl carbon, followed by the elimination of leaving groups to form the urea linkage. This mechanism avoids the radical pathways or high-energy intermediates associated with metal-catalyzed carbonylations, resulting in a cleaner reaction profile with fewer by-products. The absence of transition metals also means there is no risk of metal-induced decomposition of sensitive substrates, ensuring higher fidelity in the synthesis of complex molecular architectures.
Impurity control is inherently superior in this system due to the chemoselectivity of the activation process. Traditional methods often suffer from over-reaction or polymerization side reactions, especially with highly reactive isocyanates. However, the controlled generation of the reactive species from sodium carbonate and SO2F2 ensures that the concentration of the active intermediate remains low and steady, favoring the desired bimolecular coupling over competing degradation pathways. The patent data indicates that for aromatic amines, the product often precipitates directly from the reaction mixture upon aqueous workup, leaving soluble impurities such as excess salts and catalysts in the mother liquor. This self-purifying characteristic significantly reduces the burden on downstream processing units, minimizing solvent usage and waste generation. For aliphatic amines, where the product may remain soluble, standard extraction and chromatography techniques yield high-purity materials, demonstrating the versatility of the method across different substrate classes while maintaining a clean impurity profile.
How to Synthesize Urea Derivatives Efficiently
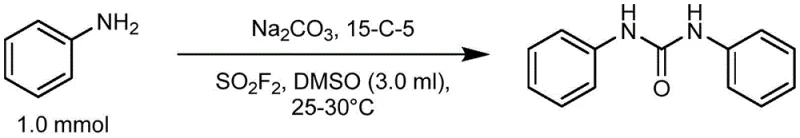
The practical implementation of this synthesis route is straightforward and robust, designed to facilitate easy technology transfer from the laboratory to pilot and commercial scales. The process begins with the preparation of a homogeneous mixture containing the amine substrate, anhydrous sodium carbonate, and a catalytic amount of a phase transfer agent in a suitable solvent. The reaction is initiated by the controlled introduction of SO2F2 gas, which can be sourced commercially or generated in situ, allowing for flexibility in supply chain management. Monitoring the reaction progress is easily achieved via TLC or HPLC, with typical completion times ranging from 16 to 24 hours at ambient temperature. The detailed standardized synthesis steps, including specific molar ratios and workup procedures for various amine substrates, are outlined in the guide below to ensure reproducibility and optimal yield.
- Dissolve the amine compound, anhydrous sodium carbonate, and a phase transfer catalyst (such as 15-crown-5) in an organic solvent like DMSO to form a mixed system.
- Introduce sulfuryl fluoride (SO2F2) gas into the mixture at 25-30°C and stir for 16-24 hours until the reaction is complete.
- Pour the resulting solid-liquid mixture into water to precipitate the solid product, then filter, wash with water, and dry to obtain the pure urea derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers profound advantages that directly impact the bottom line and supply chain resilience for manufacturers of pharmaceutical intermediates and fine chemicals. The substitution of expensive and hazardous reagents with commodity chemicals like sodium carbonate creates a substantial cost advantage in raw material procurement. Additionally, the elimination of transition metal catalysts removes the need for costly metal scavenging steps and rigorous testing for residual metals, which are mandatory regulatory requirements for drug substances. This simplification of the purification train not only reduces processing time but also lowers the consumption of solvents and consumables, contributing to a more sustainable and economically viable manufacturing process. The mild reaction conditions further translate to reduced energy costs, as there is no need for heating or high-pressure equipment, allowing the reaction to be run in standard glass-lined or stainless steel reactors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of sodium carbonate, which is significantly cheaper than phosgene, isocyanates, or specialized organometallic reagents. By avoiding the use of precious metal catalysts such as palladium or ruthenium, the process eliminates a major cost driver associated with catalyst recovery and loss. Furthermore, the simplified workup procedure, which often requires only filtration and washing for aromatic substrates, drastically reduces labor hours and solvent waste disposal costs. This lean manufacturing approach allows for a lower cost of goods sold (COGS), providing a competitive edge in pricing for high-volume urea derivatives while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by relying on widely available, non-regulated raw materials. Sodium carbonate and common phase transfer catalysts are commodity chemicals with robust global supply networks, minimizing the risk of shortages that often plague specialized reagents. The ability to generate SO2F2 in situ from readily available precursors further de-risks the supply chain by reducing dependence on external gas suppliers. This autonomy in reagent generation ensures continuous production capability even during logistical disruptions. Moreover, the stability of the starting materials allows for long-term storage without degradation, facilitating better inventory management and reducing the pressure for just-in-time delivery of sensitive intermediates.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than traditional methods, aligning with increasingly stringent global regulations on chemical manufacturing. The absence of toxic phosgene eliminates the need for complex scrubbing systems and emergency containment protocols, simplifying facility permitting and operation. The reduction in heavy metal usage means less hazardous waste is generated, easing the burden on wastewater treatment facilities and lowering disposal costs. The process is inherently scalable, as the exothermic nature of the reaction is manageable at room temperature, preventing thermal runaway risks associated with high-temperature processes. This combination of safety, sustainability, and scalability makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this urea synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process engineers and quality assurance teams. Understanding these details is crucial for assessing the feasibility of adopting this method for specific product lines and for integrating it into existing manufacturing frameworks. The clarity provided here aims to reduce uncertainty and accelerate the decision-making process for technical stakeholders evaluating this innovative synthetic route.
Q: What are the advantages of using sodium carbonate over phosgene for urea synthesis?
A: Sodium carbonate is a stable, inexpensive, and non-toxic solid C1 source, whereas phosgene is a hazardous gas requiring strict safety measures. This method eliminates the need for toxic reagents and complex handling protocols.
Q: Does this synthesis method require transition metal catalysts?
A: No, this protocol operates without expensive transition metal catalysts like palladium or nickel. It relies on phase transfer catalysis and SO2F2 activation, which simplifies purification and reduces heavy metal contamination risks.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the reaction proceeds at room temperature with simple workup procedures such as filtration or extraction. The use of common solvents and reagents facilitates easy scale-up from laboratory to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Urea Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the carbonate activation route described in patent CN114213286B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that for our partners in the pharmaceutical and agrochemical industries, consistency and compliance are non-negotiable, and our infrastructure is designed to deliver high-purity urea derivatives that meet the most demanding global regulatory standards.
We invite you to collaborate with us to leverage this cost-effective and sustainable technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating how this route can optimize your manufacturing budget. Please contact our technical procurement team to request specific COA data for our existing urea derivative portfolio or to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive efficiency and innovation in your supply chain, ensuring reliable access to critical chemical building blocks.
