Advanced Synthesis of Heterocyclic Intermediates: A Scalable Route for Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries rely heavily on the efficient construction of complex heterocyclic scaffolds, which serve as the structural backbone for countless active ingredients. Patent CN1795178A introduces a transformative methodology for synthesizing heterocyclic compounds of formula I, addressing critical bottlenecks in traditional synthetic routes. This innovation centers on a mild, two-step sequence that converts readily available isothiocyanates and primary amines into valuable thiourea intermediates, which are subsequently cyclized under basic conditions using sulfonyl chlorides. Unlike legacy methods that often demand harsh acidic environments or toxic heavy metal catalysts, this process operates effectively at near-room temperature, typically between 0°C and 35°C, ensuring compatibility with sensitive functional groups such as esters and nitriles. The strategic advantage lies in the selective ring closure that proceeds with high specificity, generally yielding a single cyclization product while cleanly eliminating the thiourea sulfur atom, thereby streamlining the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of heterocycles from thiourea precursors has been plagued by significant operational and environmental drawbacks that hinder scalable manufacturing. Traditional protocols frequently necessitate the use of hazardous heavy metal derivatives, such as mercury(II) oxide or lead oxide, to facilitate the cyclization of 1-(2-hydroxyethyl)-3-arylthioureas into oxazolidin-2-ylidenearylamines. The presence of these toxic metals poses severe regulatory and safety challenges, as even trace amounts in the final active pharmaceutical ingredient (API) are unacceptable and require costly, multi-step removal processes. Furthermore, alternative acid-catalyzed pathways often demand elevated temperatures and high acid concentrations, creating drastic reaction conditions that can degrade sensitive functional groups like ketals or nitriles present in complex molecular architectures. Other methods utilizing methyl iodide introduce risks of competing side reactions at nucleophilic centers, while carbodiimide-based approaches suffer from the difficult and time-consuming removal of urea by-products, collectively inflating production costs and extending lead times.
The Novel Approach
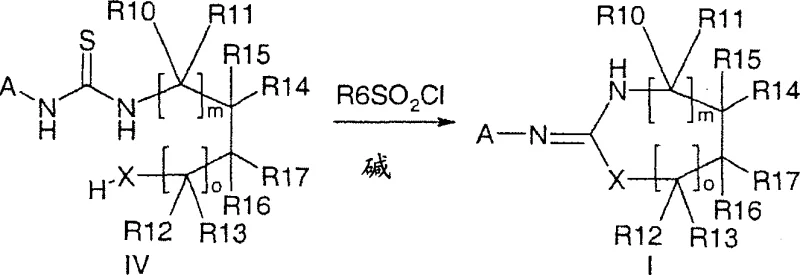
The novel approach detailed in the patent data circumvents these historical limitations by employing a sulfonyl chloride-mediated cyclization in the presence of a base, offering a robust and environmentally friendlier alternative. This method utilizes inexpensive and easy-to-handle reagents, specifically isothiocyanates and amino alcohols or diamines, to generate the desired heterocycles with variable ring sizes without the need for extreme thermal energy. A key differentiator is the ability to perform the reaction under mild conditions, typically around room temperature, which preserves the integrity of fragile functional groups and minimizes the formation of degradation by-products. Moreover, the process allows for the use of polymer-bound sulfonyl chlorides, which revolutionizes the work-up procedure by enabling product isolation through simple filtration and evaporation steps. This adaptability makes the synthesis suitable for both large-scale kilogram production and high-throughput robotic synthesis, providing a versatile platform for the rapid generation of structurally diverse analogs essential for drug discovery campaigns.
Mechanistic Insights into Sulfonyl Chloride-Mediated Cyclization
The core of this synthetic breakthrough lies in the activation of the thiourea intermediate via sulfonylation, which triggers an intramolecular nucleophilic attack to close the ring. Initially, the isothiocyanate reacts with the primary amine to form a thiourea derivative, establishing the necessary connectivity for cyclization. Upon treatment with a base, such as sodium hydroxide or potassium carbonate, and a sulfonyl chloride like p-toluenesulfonyl chloride, the thiourea nitrogen is activated, facilitating the displacement of the sulfur atom. This mechanism ensures that the ring closure proceeds very selectively, generally providing only one cyclization product, which is crucial for maintaining high stereochemical purity in chiral intermediates. The reaction pathway avoids the formation of stable metal-thiourea complexes that characterize older methods, instead relying on the transient formation of a sulfonamide-like species that collapses to release the heterocyclic core and a soluble sulfur-containing by-product that is easily washed away.
Controlling the impurity profile is paramount in the synthesis of pharmaceutical intermediates, and this mechanism offers inherent advantages in that regard. By operating at lower temperatures and avoiding strong Lewis acids, the process significantly reduces the risk of oligomerization or polymerization side reactions that often plague thiourea chemistry. The selectivity of the cyclization is further enhanced by the confinement of the thiourea intermediate, particularly when at least one aryl substituent is present on the nitrogen atom, which directs the ring closure to occur at the desired position. Additionally, the use of aqueous base solutions allows for effective pH control during the work-up, enabling the separation of the basic heterocyclic product from acidic by-products through simple phase separation. This clean reaction profile minimizes the burden on downstream purification units, such as chromatography columns, thereby reducing solvent consumption and waste generation while ensuring the final product meets stringent purity specifications required for clinical applications.
How to Synthesize Heterocyclic Compounds Efficiently
The synthesis of these high-value heterocyclic compounds is designed for operational simplicity and scalability, making it an ideal candidate for technology transfer from laboratory to commercial plant. The process begins with the formation of the thiourea intermediate in a solvent like tetrahydrofuran (THF) at ambient temperature, followed by the direct addition of base and sulfonyl chloride to effect cyclization in a potential one-pot sequence. This streamlined workflow eliminates the need for isolating unstable intermediates, reducing handling time and exposure to potentially hazardous materials. For a comprehensive understanding of the specific stoichiometric ratios, solvent choices, and work-up procedures tailored to your specific substrate, please refer to the detailed standardized synthesis steps provided in the guide below.
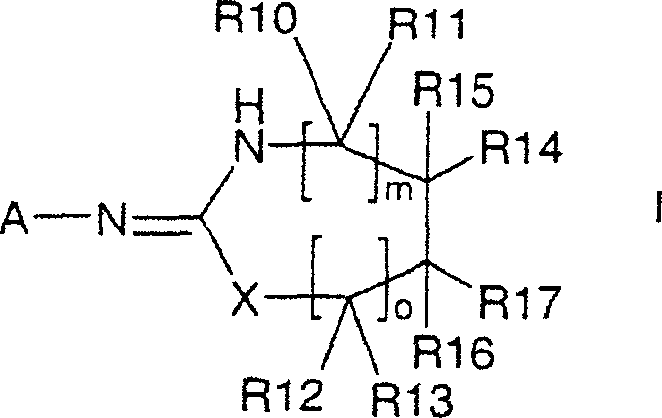
- React an isothiocyanate of formula II with a primary amine of formula III in a solvent like THF to form a thiourea intermediate of formula IV.
- Treat the thiourea intermediate with a base (e.g., sodium hydroxide) and a sulfonyl chloride (e.g., p-toluenesulfonyl chloride) to induce cyclization.
- Isolate the final heterocyclic compound of formula I through extraction, filtration, or crystallization, optionally utilizing polymer-bound reagents for simplified purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process delivers substantial value by addressing key pain points in the supply chain of complex organic intermediates. The elimination of heavy metal catalysts not only aligns with increasingly strict environmental regulations but also removes the need for expensive scavenging resins or complex filtration systems dedicated to metal removal. This simplification of the downstream processing directly translates to reduced operational expenditures and a smaller environmental footprint, appealing to sustainability-focused procurement strategies. Furthermore, the reliance on commodity chemicals like sulfonyl chlorides and common bases ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents that can disrupt production schedules.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the drastic simplification of the purification workflow. By avoiding the use of toxic heavy metals, manufacturers save significantly on the costs associated with hazardous waste disposal and the specialized equipment required for metal clearance. Additionally, the ability to use polymer-bound reagents allows for the recovery and potential reuse of solid supports, further driving down the cost of goods sold (COGS). The mild reaction conditions also reduce energy consumption, as there is no need for prolonged heating or cryogenic cooling, contributing to overall process efficiency and lower utility costs in large-scale production facilities.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain continuity by utilizing widely available starting materials that are not subject to the geopolitical or logistical constraints often seen with exotic catalysts. The tolerance of the reaction to various functional groups means that a single set of conditions can be applied to a broad library of substrates, reducing the need for multiple bespoke synthetic routes and simplifying inventory management. This flexibility allows suppliers to respond more rapidly to changes in demand, ensuring consistent delivery of high-quality intermediates without the delays typically caused by complex optimization studies or reagent shortages.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is straightforward due to the exothermic nature of the reaction being manageable under controlled addition rates and the absence of gas evolution or hazardous by-products. The use of aqueous work-ups and organic solvents like ethyl acetate or ethers facilitates easy phase separation and solvent recovery, aligning with green chemistry principles. This compliance with environmental standards reduces the regulatory burden on manufacturing sites and minimizes the risk of production shutdowns due to non-compliance, ensuring a reliable long-term supply of critical pharmaceutical building blocks for global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this heterocyclic synthesis technology. These insights are derived directly from the experimental data and mechanistic understanding presented in the patent literature, aiming to clarify the practical application of the method for process chemists and project managers. Understanding these nuances is essential for evaluating the feasibility of adopting this route for specific target molecules and for anticipating potential challenges during scale-up activities.
Q: What are the advantages of using sulfonyl chloride over heavy metal catalysts for heterocycle synthesis?
A: The use of sulfonyl chloride eliminates the need for toxic heavy metals like mercury or lead oxides, ensuring the final product is free from undesirable metal traces and simplifying the purification process significantly.
Q: Can this process be adapted for automated or parallel synthesis?
A: Yes, the mild reaction conditions and the option to use polymer-bound sulfonyl chlorides allow for simple filtration and evaporation steps, making the process highly suitable for robot-assisted and parallel synthesis platforms.
Q: What types of heterocyclic rings can be formed using this method?
A: This versatile method supports the formation of various ring sizes, including 5-membered rings like oxazolidines and thiazolidines, as well as larger saturated carbocyclic rings fused with nitrogen, oxygen, or sulfur atoms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and commercialization. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and robust. We are committed to delivering high-purity heterocyclic intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall manufacturing costs.
