Optimized Industrial Synthesis of Celecoxib Intermediates for Commercial Scale-Up
Optimized Industrial Synthesis of Celecoxib Intermediates for Commercial Scale-Up
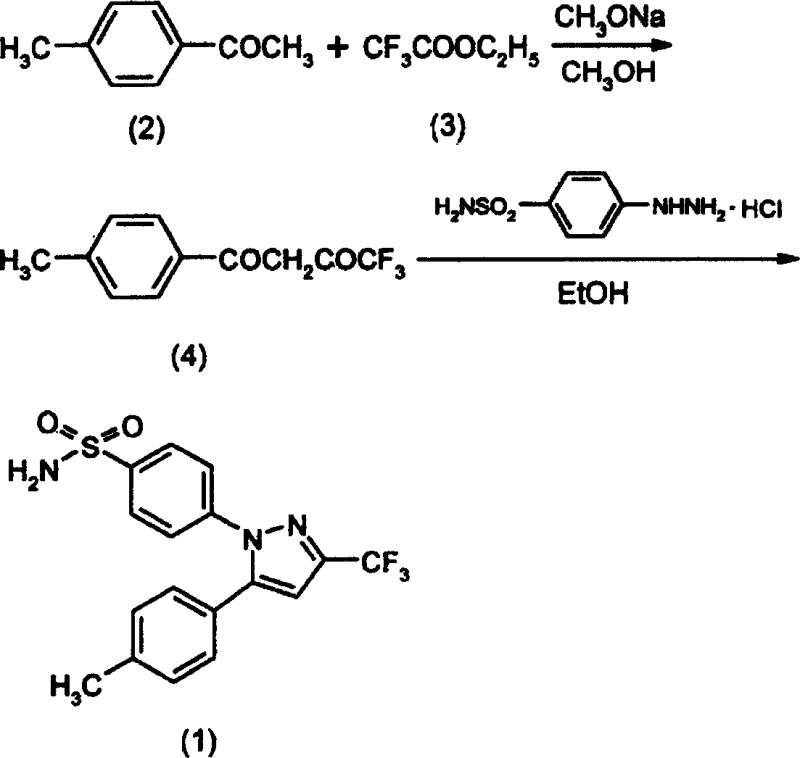
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-value active pharmaceutical ingredients (APIs), particularly for widely prescribed non-steroidal anti-inflammatory drugs (NSAIDs) like Celecoxib. Patent CN1152865C presents a significant technological advancement in the synthesis of YM 177, commonly known as Celecoxib, by addressing critical bottlenecks in catalyst handling and solvent toxicity. This intellectual property outlines a refined Claisen condensation strategy that utilizes in-situ generation of sodium methoxide, thereby bypassing the logistical and stability challenges associated with storing commercial sodium methoxide powder. Furthermore, the patent introduces a greener purification protocol that substitutes hazardous chlorinated solvents with benign alcohol-based or ester-based systems. For global procurement teams and R&D directors, this methodology represents a pivotal shift towards more sustainable and cost-effective pharmaceutical intermediate manufacturing, ensuring a reliable supply chain for this essential COX-2 inhibitor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Celecoxib, such as those disclosed in earlier international patents like WO97/11704, rely heavily on the use of pre-prepared solid sodium methoxide as a catalyst for the initial condensation step. This approach introduces substantial operational inefficiencies because solid sodium methoxide is highly hygroscopic and prone to rapid degradation upon exposure to atmospheric moisture, leading to inconsistent catalytic activity and poor batch-to-batch repeatability. Additionally, the conventional purification process necessitates the use of methylene chloride and n-hexane for recrystallization, solvents that are not only expensive but also pose severe health risks to operators and significant environmental compliance burdens due to their toxicity and volatile organic compound (VOC) emissions. The requirement for vacuum concentration and pulverization of the catalyst further increases the workload and energy consumption, creating a complex workflow that is difficult to scale safely in large-scale API manufacturing facilities without rigorous safety controls.
The Novel Approach
In stark contrast, the novel approach detailed in CN1152865C streamlines the production workflow by generating the sodium methoxide catalyst directly within the reaction vessel through the reaction of metallic sodium with absolute methanol. This in-situ generation ensures the catalyst is fresh, highly active, and free from moisture-induced degradation, thereby enhancing the reliability of the Claisen condensation between p-methylacetophenone and ethyl trifluoroacetate. Moreover, the innovation extends to the downstream processing, where the recrystallization of the final product is achieved using ethanol/water or ethyl acetate/petroleum ether mixtures. These solvent systems are not only significantly cheaper and less toxic than methylene chloride but also yield products with superior crystal morphology and color. This dual optimization of both the catalytic initiation and the purification stages results in a total yield of 47-48%, demonstrating that environmental sustainability and high efficiency can be achieved simultaneously in commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into In-Situ Catalyzed Claisen Condensation
The core chemical transformation in this synthesis is the Claisen condensation, a fundamental carbon-carbon bond-forming reaction that links the p-methylacetophenone and ethyl trifluoroacetate to form the critical 1,3-diketone intermediate. Mechanistically, the in-situ generated methoxide ion acts as a strong base to deprotonate the alpha-carbon of the ketone, creating a nucleophilic enolate that attacks the carbonyl carbon of the ester. The use of freshly generated methoxide ensures a high concentration of active base without the interference of carbonate impurities often found in aged commercial samples, which drives the equilibrium towards the formation of the desired beta-diketone species. Following this condensation, the intermediate undergoes a cyclization reaction with sulfonamide-phenylhydrazine hydrochloride in ethanol. This step involves the nucleophilic attack of the hydrazine nitrogen on the diketone carbonyls, followed by dehydration to close the pyrazole ring, ultimately yielding the target Celecoxib structure with high regioselectivity.
Impurity control is intrinsically linked to the choice of solvents and the freshness of the catalyst in this mechanism. By avoiding the use of degraded sodium methoxide, the process minimizes side reactions such as hydrolysis of the ester starting material or incomplete condensation, which are common sources of yield loss in traditional methods. Furthermore, the switch to ethanol/water recrystallization leverages the differential solubility of the product versus potential organic impurities; the polar nature of the ethanol/water system effectively washes away non-polar byproducts while allowing the target molecule to crystallize in a pure, needle-shaped form. This precise control over the crystallization thermodynamics ensures that the final high-purity Celecoxib meets stringent pharmacopeial standards without requiring additional chromatographic purification steps, which are often cost-prohibitive at an industrial scale.
How to Synthesize Celecoxib Efficiently
The synthesis protocol described in the patent offers a practical roadmap for laboratories and pilot plants aiming to produce Celecoxib with improved safety and cost profiles. The process begins with the careful addition of sodium metal to anhydrous methanol to generate the catalytic solution, followed by the sequential addition of the ketone and ester substrates under reflux conditions. After the condensation is complete, the intermediate is isolated through acidification and extraction, dried, and then immediately subjected to the cyclization step with the hydrazine salt in ethanol. The final purification is achieved by dissolving the crude solid in hot ethyl acetate or ethanol and inducing crystallization through the gradual addition of a non-solvent like petroleum ether or water. For detailed standard operating procedures and specific stoichiometric ratios required to replicate this high-yielding process, please refer to the technical guide below.
- Generate sodium methoxide in-situ by reacting sodium metal with absolute methanol, then add p-methylacetophenone and ethyl trifluoroacetate for Claisen condensation.
- Isolate the diketone intermediate via acidification and extraction, then concentrate to obtain the crude yellow oil.
- React the intermediate with sulfonamide-phenylhydrazine hydrochloride in ethanol under reflux to form the pyrazole ring, followed by recrystallization using ethanol/water or ethyl acetate/petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN1152865C offers tangible strategic advantages that extend beyond simple chemical yield. The elimination of pre-packaged sodium methoxide removes a volatile and hazardous material from the supply chain, reducing storage costs and minimizing the risk of supply disruptions caused by the instability of the catalyst during transport. Furthermore, the substitution of expensive and regulated solvents like methylene chloride with commodity chemicals like ethanol and ethyl acetate drastically simplifies solvent recovery and waste management protocols. This shift not only lowers the direct material costs but also reduces the regulatory burden associated with handling hazardous air pollutants, making the entire manufacturing process more resilient to changing environmental regulations and more attractive for long-term cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost, high-toxicity solvents with low-cost, readily available alternatives. By utilizing ethanol/water or ethyl acetate/petroleum ether systems, manufacturers can avoid the premium pricing and strict disposal fees associated with chlorinated solvents. Additionally, the in-situ generation of the catalyst eliminates the markup charged by suppliers for processed solid sodium methoxide and removes the energy costs associated with its vacuum drying and pulverization. These cumulative savings contribute to a significantly lower cost of goods sold (COGS) without compromising the quality or yield of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved by removing the dependency on hygroscopic catalysts that require specialized storage conditions. Solid sodium methoxide often suffers from shelf-life issues, leading to variability in reaction performance if the material absorbs moisture during warehousing or shipping. By generating the catalyst on-demand from stable raw materials (sodium metal and methanol), the process ensures consistent reaction kinetics and product quality regardless of external storage conditions. This reliability allows for tighter production scheduling and reduces the need for safety stock, thereby optimizing inventory turnover and ensuring a steady flow of pharmaceutical intermediates to downstream formulation units.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is inherently more scalable because it relies on solvents with higher flash points and lower toxicity profiles. The removal of methylene chloride, a suspected carcinogen, significantly improves the safety of the working environment for plant operators and simplifies the permitting process for facility expansions. The use of ethanol and water also facilitates easier solvent recovery through distillation, allowing for high rates of recycling that further reduce waste generation. This alignment with green chemistry principles ensures that the manufacturing process remains compliant with increasingly strict global environmental standards, securing the long-term viability of the production site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the optimized synthesis of Celecoxib. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this specific methodology outperforms legacy processes in terms of safety, efficiency, and product quality. Understanding these nuances is critical for technical teams evaluating potential technology transfers or licensing opportunities for large-scale production.
Q: How does the in-situ catalyst generation improve process stability?
A: By generating sodium methoxide directly from sodium metal and methanol within the reactor, the process eliminates the need to purchase, store, and handle hygroscopic solid sodium methoxide, which often degrades and causes batch inconsistency.
Q: What are the environmental benefits of the new purification method?
A: The patented method replaces toxic methylene chloride and n-hexane with ethanol/water or ethyl acetate/petroleum ether systems, significantly reducing VOC emissions and operator exposure risks while lowering solvent disposal costs.
Q: Does this synthetic route affect the final yield of Celecoxib?
A: Yes, the optimized protocol achieves a total yield of 47-48%, which is comparable to or slightly better than the 46% yield reported in prior art methods, while simultaneously improving crystal morphology and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Celecoxib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative techniques described in patents like CN1152865C can be seamlessly translated into robust industrial operations. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Celecoxib intermediate meets the highest international standards for safety and efficacy. Our infrastructure is designed to handle complex heterocyclic chemistry with precision, making us an ideal partner for companies seeking to optimize their supply chain for anti-inflammatory medications.
We invite you to collaborate with us to leverage these technological advancements for your specific production needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both your product quality and your bottom line.
