Advanced Synthesis of Triazole Heterocycles: A Scalable Route for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Triazole Heterocycles: A Scalable Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust synthetic pathways that balance high purity with economic viability, particularly for complex heterocyclic scaffolds used in next-generation therapeutics. Patent CN112300122A introduces a significant advancement in the synthesis of (Z)-N-((4H-1,2,4-triazol-4-yl)methylene)-3-(pyridin-4-yl)aniline, a versatile triazole heterocyclic compound with potent potential as an intermediate for anti-inflammatory, antibacterial, and antitumor agents. This innovation addresses critical bottlenecks in traditional manufacturing by replacing energy-intensive microwave protocols with a温和 (mild) thermal approach that maintains exceptional yield and selectivity. The structural integrity of the final molecule, featuring a pyridine ring linked to a triazole moiety via an imine bridge, is crucial for its bioactivity and metal-coordination capabilities, making it a high-value target for R&D teams focused on SCO (Spin Crossover) materials and medicinal chemistry.
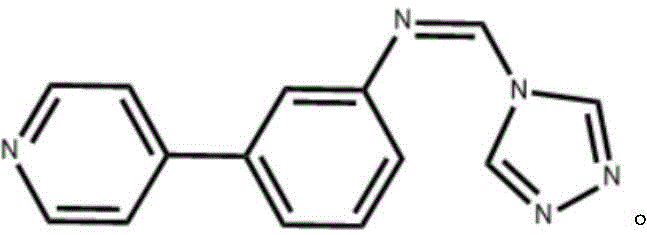
For procurement managers and supply chain directors, the implications of this patent extend beyond mere chemical novelty; it represents a tangible opportunity for cost reduction in API manufacturing. By utilizing readily available starting materials such as pyridine-4-boronic acid and 3-bromobenzaldehyde, the supply chain risk is minimized compared to routes requiring exotic reagents. Furthermore, the elimination of complex purification steps typically associated with microwave-assisted synthesis translates directly into reduced processing time and lower operational expenditures. As a reliable pharmaceutical intermediate supplier, understanding these process efficiencies is key to securing long-term contracts with multinational corporations that demand both consistency and competitive pricing in their raw material sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazole heterocyclic compounds has relied heavily on methods that impose significant logistical and financial burdens on manufacturers. Conventional protocols often necessitate the use of microwave radiation to drive Suzuki coupling reactions, which, while effective on a small laboratory scale, present severe challenges for commercial scale-up of complex heterocycles. The requirement for specialized microwave reactors limits batch sizes and introduces safety hazards related to pressure buildup and uneven heating in larger vessels. Additionally, traditional aldehyde-amine condensation reactions frequently employ harsh conditions that can lead to the degradation of sensitive functional groups, resulting in lower overall yields and a complex impurity profile that requires extensive chromatographic purification. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, creating friction in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112300122A offers a streamlined alternative that prioritizes operational simplicity without compromising chemical performance. The novel approach utilizes a standard thermal heating method at a moderate temperature of 80°C, effectively removing the dependency on expensive and capacity-limited microwave equipment. This shift allows for the use of standard glass-lined or stainless steel reactors, facilitating a smoother transition from pilot plant to full-scale production. Moreover, the reaction conditions are optimized to minimize side reactions, ensuring that the crude product possesses high content and purity right from the initial isolation. This reduction in downstream processing requirements not only accelerates the manufacturing timeline but also significantly lowers solvent consumption and waste generation, aligning with modern green chemistry principles that are increasingly mandated by regulatory bodies.
Mechanistic Insights into Suzuki Coupling and Aldehyde-Amine Condensation
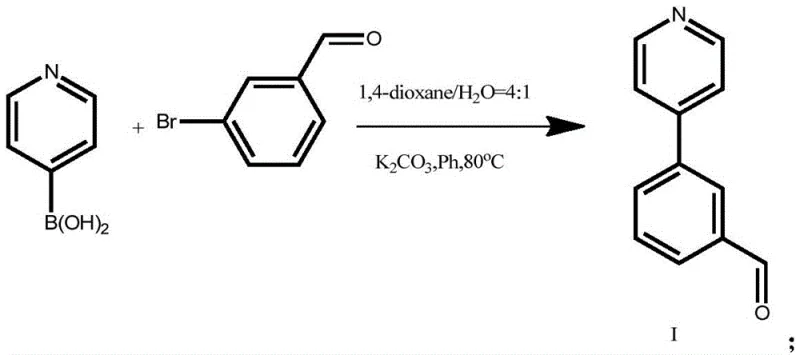
The core of this synthetic strategy lies in a two-step sequence that leverages well-understood yet highly optimized organic transformations. The first step involves a Suzuki-Miyaura cross-coupling reaction, a cornerstone of modern medicinal chemistry, which links the pyridine and benzene rings. In this specific embodiment, pyridine-4-boronic acid reacts with 3-bromobenzaldehyde in the presence of a palladium catalyst, specifically tetrakis(triphenylphosphine)palladium(0), and potassium carbonate as a base. The choice of a 1,4-dioxane and water solvent system (4:1 ratio) is critical; it provides the necessary solubility for the organic substrates while allowing the inorganic base to function effectively in the aqueous phase. This biphasic environment facilitates the transmetallation step of the catalytic cycle, ensuring high conversion rates while maintaining the stability of the aldehyde functionality against unwanted oxidation or reduction.
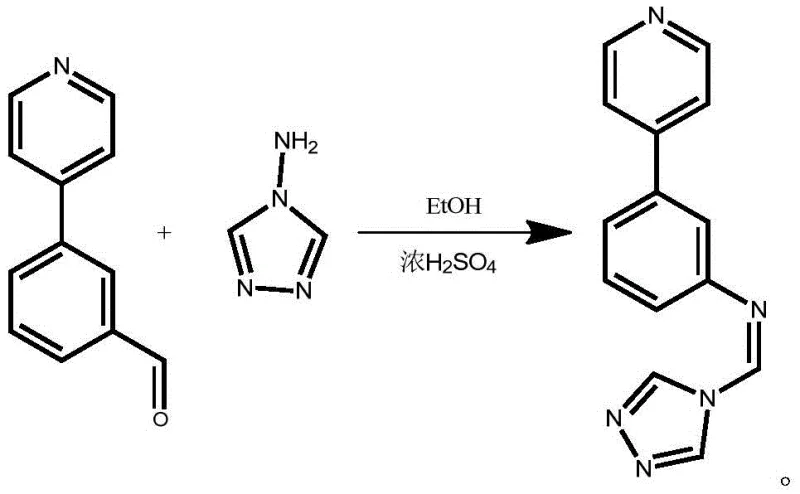
Following the successful construction of the biaryl aldehyde intermediate, the second step employs an acid-catalyzed aldehyde-amine condensation to install the triazole moiety. This reaction proceeds by mixing the intermediate with 4-amino-1,2,4-triazole in ethanol, using concentrated sulfuric acid to activate the carbonyl group towards nucleophilic attack by the amine. The mechanism involves the formation of a hemiaminal intermediate which subsequently dehydrates to form the stable imine (Schiff base) linkage. The use of ethanol as a solvent in this step is particularly advantageous for commercial operations due to its low toxicity and ease of removal via distillation. The reaction is driven to completion at 80°C over a period of three hours, ensuring that the equilibrium favors the formation of the desired (Z)-isomer, which is essential for the compound's biological activity and coordination properties.
How to Synthesize (Z)-N-((4H-1,2,4-triazol-4-yl)methylene)-3-(pyridin-4-yl)aniline Efficiently
To achieve optimal results in the laboratory or pilot plant, strict adherence to the molar ratios and temperature profiles specified in the patent is essential. The process begins with the careful preparation of the Suzuki coupling mixture under an inert atmosphere to prevent catalyst deactivation by oxygen. Following the coupling, the workup procedure involves filtration through celite to remove palladium black and inorganic salts, followed by extraction and washing steps that are designed to maximize recovery while minimizing impurity carryover. The subsequent condensation step requires precise control of the acid catalyst amount to avoid hydrolysis of the product while ensuring sufficient activation of the aldehyde. Detailed standardized synthesis steps see the guide below.
- Perform Suzuki coupling between pyridine-4-boronic acid and 3-bromobenzaldehyde using Pd(PPh3)4 catalyst in 1,4-dioxane/water at 80°C.
- Isolate the intermediate aldehyde compound via filtration, washing, and extraction to remove palladium residues and inorganic salts.
- Conduct aldehyde-amine condensation with 4-amino-1,2,4-triazole in ethanol using concentrated sulfuric acid catalyst at 80°C to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the technical improvements in this patent translate directly into strategic commercial benefits. The shift away from microwave technology and towards conventional heating infrastructure drastically simplifies the capital expenditure requirements for manufacturing facilities. This means that existing production lines can be utilized without the need for costly retrofits or the purchase of specialized equipment, thereby accelerating the time-to-market for new drug candidates utilizing this intermediate. Furthermore, the use of common solvents like ethanol and 1,4-dioxane, as opposed to more exotic or highly regulated solvents, simplifies procurement logistics and reduces the volatility of raw material costs. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations and regulatory changes.
- Cost Reduction in Manufacturing: The elimination of microwave reactors and the simplification of purification processes lead to substantial cost savings. By avoiding column chromatography and relying on crystallization or simple filtration, the consumption of silica gel and large volumes of elution solvents is drastically reduced. Additionally, the high yield and selectivity of the reaction minimize the loss of valuable starting materials, ensuring that the overall material cost per kilogram of the final product is significantly lower than that of conventional methods. This efficiency allows for more competitive pricing models when bidding for large-scale supply contracts.
- Enhanced Supply Chain Reliability: The starting materials, pyridine-4-boronic acid and 3-bromobenzaldehyde, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to minor variations in utility supplies or environmental conditions, ensuring consistent output quality. This reliability is crucial for pharmaceutical companies that require uninterrupted supply to maintain their own clinical trial timelines and commercial production schedules, thereby strengthening the partnership between the supplier and the end-user.
- Scalability and Environmental Compliance: The mild reaction temperatures and the use of less hazardous solvents make this process inherently safer and easier to scale. Large-scale exotherms are manageable at 80°C, unlike the rapid and intense heating of microwave processes. From an environmental perspective, the reduced solvent load and the avoidance of heavy metal contamination (through efficient palladium removal) simplify wastewater treatment and waste disposal. This alignment with green chemistry standards not only reduces regulatory compliance costs but also enhances the corporate social responsibility profile of the manufacturing operation, which is increasingly important for global partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity for technical teams evaluating the feasibility of adoption. Understanding these nuances is vital for making informed decisions about process integration and supplier selection.
Q: What are the key advantages of this synthesis method over conventional microwave-assisted routes?
A: This method eliminates the need for specialized microwave equipment and harsh reaction conditions, utilizing standard heating at 80°C which significantly simplifies reactor requirements and improves operational safety for large-scale production.
Q: How does the solvent system contribute to cost efficiency and environmental compliance?
A: By employing a 1,4-dioxane/water mixture and ethanol, the process reduces reliance on highly toxic polar aprotic solvents like DMF, thereby lowering waste disposal costs and aligning with stricter environmental regulations for pharmaceutical manufacturing.
Q: Is this process suitable for commercial scale-up of complex heterocycles?
A: Yes, the mild temperature profile (80°C) and robust catalyst system allow for straightforward translation from laboratory to pilot and commercial scales without the thermal management challenges associated with exothermic microwave reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-N-((4H-1,2,4-triazol-4-yl)methylene)-3-(pyridin-4-yl)aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry described in CN112300122A can be realized at the volumes your business demands. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay. Our dedication to quality assurance ensures that every batch of triazole heterocyclic compound we supply is consistent, reliable, and ready for immediate use in your downstream synthetic applications.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route. By leveraging our process optimization capabilities, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to scientific excellence and operational efficiency can drive value for your organization and support your goals in developing next-generation therapeutic agents.
