Advanced Synthesis of Triazole Heterocyclic Compounds for Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust synthetic routes for heterocyclic compounds that serve as critical building blocks for next-generation therapeutics. Patent CN112300122A introduces a significant advancement in the synthesis of triazole heterocyclic compounds, specifically detailing the preparation of (Z)-N-((4H-1,2,4-triazol-4-yl)methylene)-3-(pyridin-4-yl)aniline. This compound represents a sophisticated molecular architecture combining a triazole moiety with a pyridine-substituted phenyl ring, offering enhanced potential for metal coordination and biological activity. The disclosed methodology moves away from traditional, energy-intensive microwave-assisted protocols, favoring a more scalable approach using standard heating in a biphasic solvent system. For R&D directors and procurement specialists, this shift implies a transition towards more economically viable and environmentally considerate manufacturing processes. The strategic integration of a pyridine ring not only diversifies the chemical space available for drug discovery but also aligns with the growing demand for multifunctional intermediates capable of acting as anti-inflammatory, antibacterial, and antitumor agents. Understanding the nuances of this patent is essential for stakeholders aiming to secure a reliable pharmaceutical intermediate supplier for complex heterocyclic scaffolds.
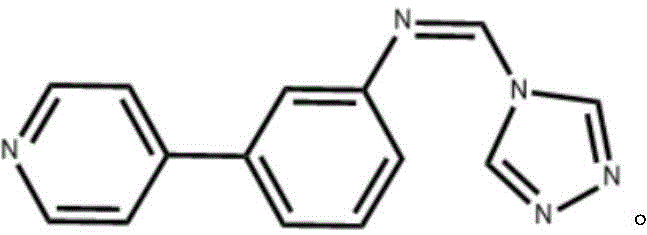
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazole heterocyclic compounds has relied heavily on conditions that pose significant challenges for large-scale industrial application. Conventional protocols often necessitate the use of microwave radiation to drive Suzuki coupling reactions, typically employing polar aprotic solvents like DMF mixed with water. While effective on a laboratory scale, these methods introduce substantial complications when transitioning to commercial production. The reliance on microwave technology limits batch sizes and increases capital expenditure for specialized equipment. Furthermore, the use of DMF raises concerns regarding toxicity and waste disposal, complicating regulatory compliance and environmental safety protocols. Additionally, traditional aldehyde-amine condensation reactions frequently require refluxing in ethanol with strong acids, often leading to side reactions or requiring extensive purification steps to remove unreacted starting materials. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In contrast, the methodology outlined in the patent presents a streamlined and economically superior alternative that addresses these inherent inefficiencies. The novel approach utilizes a Suzuki coupling reaction conducted in a 1,4-dioxane and water mixture with a volume ratio of 4:1, heated to a moderate temperature of 80°C. This eliminates the need for microwave irradiation, allowing for the use of standard reactor vessels that are easily scalable from pilot plants to full commercial production. The choice of solvent system is particularly advantageous as it balances solubility requirements with safety profiles, reducing the environmental burden associated with toxic solvent disposal. Following the coupling step, the subsequent condensation with 4-amino-1,2,4-triazole is performed under similarly mild conditions in ethanol with catalytic sulfuric acid. This two-step sequence ensures high selectivity and yield while minimizing the formation of impurities. By optimizing reaction parameters such as molar ratios and temperature, the process achieves a level of efficiency that significantly lowers the overall cost of goods sold, making it an attractive option for cost reduction in API manufacturing.
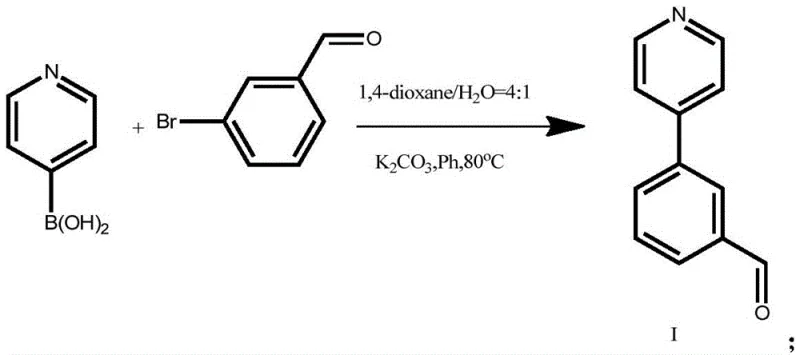
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Condensation
The core of this synthetic strategy lies in the precise execution of the palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. In the first step, pyridine-4-boronic acid reacts with 3-bromobenzaldehyde in the presence of tetrakis(triphenylphosphine)palladium(0) and potassium carbonate. The mechanism involves the oxidative addition of the aryl bromide to the palladium center, followed by transmetallation with the boronic acid species activated by the base. This forms a biaryl palladium complex which subsequently undergoes reductive elimination to yield the desired 3-(pyridin-4-yl)benzaldehyde intermediate. The use of a 1.2:1.2:1 molar ratio of boronic acid to base to aldehyde ensures complete conversion of the limiting reagent, while the 0.05 equivalent of catalyst loading strikes a balance between reaction rate and cost efficiency. The reaction is maintained under an inert atmosphere to prevent catalyst deactivation, ensuring consistent performance over the 2-hour reaction period. This mechanistic understanding is crucial for R&D teams aiming to replicate the process with high fidelity and minimal batch-to-batch variation.
Following the isolation of the aldehyde intermediate, the synthesis proceeds via an acid-catalyzed condensation reaction to form the final imine linkage. The aldehyde group of the intermediate reacts with the amino group of 4-amino-1,2,4-triazole in an ethanolic solution. Concentrated sulfuric acid acts as a proton donor, activating the carbonyl carbon of the aldehyde towards nucleophilic attack by the amine. This leads to the formation of a hemiaminal intermediate, which subsequently loses a water molecule to generate the stable C=N double bond characteristic of the target triazole heterocyclic compound. The reaction is driven to completion by heating at 80°C for at least 3 hours, ensuring that equilibrium favors the product side. The resulting precipitate can be easily filtered and washed, demonstrating a self-purifying aspect of the reaction that simplifies downstream processing. This mechanistic pathway highlights the robustness of the chemistry, offering a clear route to high-purity products suitable for sensitive pharmaceutical applications.
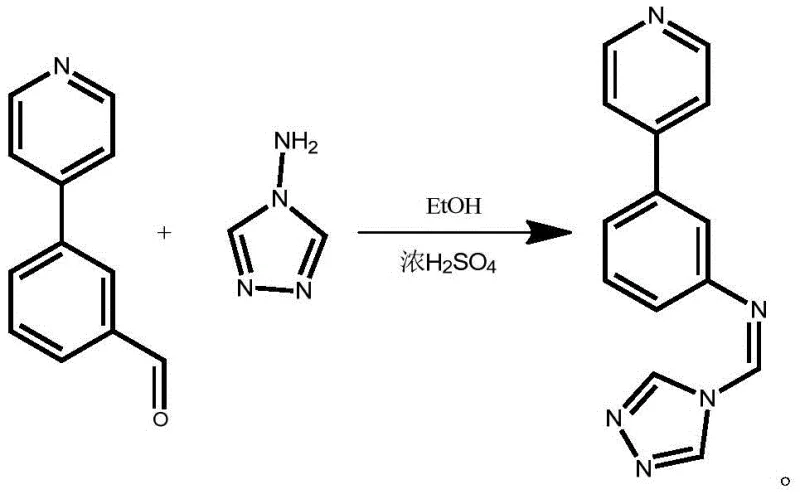
How to Synthesize (Z)-N-((4H-1,2,4-triazol-4-yl)methylene)-3-(pyridin-4-yl)aniline Efficiently
Executing this synthesis requires careful attention to stoichiometry and reaction conditions to maximize yield and purity. The process begins with the preparation of the reaction mixture for the Suzuki coupling, where precise molar ratios of pyridine-4-boronic acid, potassium carbonate, and 3-bromobenzaldehyde are combined with the palladium catalyst in the dioxane-water solvent system. Maintaining an inert atmosphere throughout this stage is critical to preserve catalyst activity. Once the coupling is complete, the workup involves filtration through celite to remove palladium residues, followed by extraction and drying to isolate the aldehyde intermediate as a yellow oily liquid. The second stage involves dissolving this intermediate in ethanol and adding 4-amino-1,2,4-triazole along with concentrated sulfuric acid. The mixture is then heated to promote condensation, after which the product precipitates upon cooling. Detailed standardized synthesis steps see the guide below.
- Perform Suzuki coupling between pyridine-4-boronic acid and 3-bromobenzaldehyde using Pd(PPh3)4 catalyst in 1,4-dioxane/water at 80°C.
- Isolate the intermediate aldehyde compound via filtration, extraction with dichloromethane, and drying over anhydrous magnesium sulfate.
- React the isolated aldehyde intermediate with 4-amino-1,2,4-triazole in ethanol with concentrated sulfuric acid at 80°C to form the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The elimination of microwave reactors and the use of common, commercially available solvents like 1,4-dioxane and ethanol drastically simplify the infrastructure requirements for production. This accessibility translates directly into reduced capital expenditure and lower operational overheads. Furthermore, the mild reaction conditions minimize the risk of thermal runaway or hazardous incidents, enhancing workplace safety and reducing insurance liabilities. The simplicity of the workup procedure, which relies on filtration and washing rather than complex chromatography, significantly shortens the production cycle time. This efficiency allows for faster turnover of batches, enabling suppliers to respond more agilely to market demands and reducing lead time for high-purity pharmaceutical intermediates. The overall process design prioritizes economic viability without compromising on the quality of the final product.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive and energy-intensive microwave technology with conventional heating methods. By utilizing a standard oil bath or heating mantle at 80°C, facilities can leverage existing reactor fleets without needing specialized upgrades. Additionally, the catalyst loading is kept low at 0.05 equivalents, which minimizes the consumption of precious palladium resources, a significant factor given the volatility of precious metal prices. The solvent system, while requiring careful handling, is recyclable and less problematic than high-boiling polar solvents like DMF, reducing waste treatment costs. These cumulative factors result in a substantially lower cost per kilogram of the final active intermediate, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including pyridine-4-boronic acid and 3-bromobenzaldehyde, are commodity chemicals with well-established global supply chains. This abundance ensures that production is not vulnerable to the shortages often associated with exotic or highly specialized reagents. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or mixing rates, leading to consistent batch quality and reliable delivery schedules. For supply chain planners, this predictability is invaluable, as it reduces the need for excessive safety stock and allows for leaner inventory management. The ability to source materials easily and produce consistently strengthens the overall resilience of the supply network against external disruptions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial scale is straightforward due to the absence of specialized equipment constraints. The use of a biphasic solvent system facilitates heat transfer and mixing in large vessels, ensuring uniform reaction conditions even at multi-ton scales. From an environmental perspective, the avoidance of DMF and the use of ethanol for the final crystallization step align with green chemistry principles, simplifying regulatory approvals and waste disposal. The precipitation of the final product allows for efficient solid-liquid separation, minimizing solvent usage in the purification stage. These attributes make the process highly attractive for manufacturers aiming to expand capacity while maintaining strict adherence to environmental, health, and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this triazole heterocyclic compound. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their development pipelines. The answers reflect the practical realities of implementing this chemistry in a GMP-compliant environment.
Q: What are the key advantages of this synthesis method over conventional microwave-assisted routes?
A: This method eliminates the need for expensive microwave equipment and harsh DMF solvents, utilizing a milder 1,4-dioxane/water system at standard heating temperatures, which significantly simplifies scale-up and reduces operational costs.
Q: How does the inclusion of the pyridine ring benefit the biological activity of the compound?
A: The pyridine ring enhances the molecule's ability to coordinate with metal ions, potentially improving its bioactivity as an anti-inflammatory, antibacterial, or antitumor agent compared to standard triazole derivatives.
Q: Is the purification process complex for commercial production?
A: No, the process is designed for simplicity; the final product precipitates upon cooling and can be easily collected by filtration and washing with ethanol, avoiding complex chromatographic purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-N-((4H-1,2,4-triazol-4-yl)methylene)-3-(pyridin-4-yl)aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this triazole synthesis, guaranteeing a consistent supply of material that meets your exacting standards. By partnering with us, you gain access to a supply chain that prioritizes reliability, quality, and technical excellence.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current sourcing strategy or need to evaluate the feasibility of this route for your specific application, we are here to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your requirements. Let us collaborate to accelerate your drug development timeline with our superior manufacturing capabilities and dedicated customer support.
