Scalable Total Synthesis of Piperonal: A Strategic Alternative for Global Flavor and Fragrance Supply Chains
Introduction to Advanced Piperonal Synthesis Technology
The global demand for high-purity Piperonal, also known as Heliotropin or 3,4-methylenedioxybenzaldehyde, continues to surge across the flavor, fragrance, and pharmaceutical sectors. Traditionally, the industry has relied heavily on semi-synthetic routes derived from safrole, a natural essential oil component that faces increasing regulatory scrutiny and supply chain volatility. However, a groundbreaking technological shift is emerging through the complete synthesis method detailed in patent CN102329297B. This innovative approach bypasses natural precursors entirely, utilizing a robust three-step chemical sequence starting from readily available petrochemical derivatives. By leveraging Friedel-Crafts acylation followed by controlled chlorination and oxidative cleavage, this method delivers a product with exceptional purity profiles and consistent quality.
The significance of this technology lies in its ability to decouple production from agricultural variables and regulatory bottlenecks associated with safrole. For R&D directors and procurement strategists, this represents a critical opportunity to secure a more resilient supply chain. The process is characterized by mild reaction conditions and the use of common industrial reagents, which significantly lowers the barrier to entry for large-scale manufacturing. As we analyze the structural integrity and synthetic pathway of this molecule, it becomes clear that this total synthesis route offers a superior alternative for modern industrial applications requiring rigorous quality control.
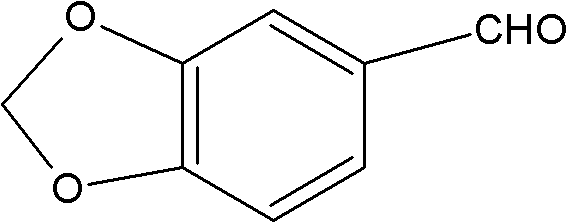
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Piperonal has been dominated by the oxidation of isosafrole, which is itself derived from the isomerization of safrole extracted from sassafras or other essential oils. This legacy pathway suffers from inherent vulnerabilities, primarily due to the classification of safrole as a List I chemical in many jurisdictions because of its potential use in illicit drug synthesis. Consequently, manufacturers face stringent reporting requirements, import/export restrictions, and unpredictable raw material pricing driven by harvest yields and geopolitical factors. Furthermore, the oxidation steps in the traditional route often involve harsh reagents like chromium trioxide or ozone, which generate significant hazardous waste and pose environmental compliance challenges. These factors collectively increase the total cost of ownership and introduce substantial supply chain risks for downstream users in the fine chemical industry.
The Novel Approach
In stark contrast, the novel total synthesis method described in CN102329297B introduces a paradigm shift by utilizing 1,3-benzodioxole and acetic anhydride as primary starting materials. This route completely eliminates the dependency on controlled natural precursors, thereby streamlining procurement and reducing regulatory overhead. The process employs a strategic sequence where the aromatic ring is first functionalized via acylation, followed by a precise side-chain modification through chlorination. This allows for a highly controlled transformation into the target aldehyde functionality without the formation of complex byproducts typical of direct oxidation methods. The result is a streamlined workflow that not only enhances operational safety but also ensures a consistent, high-yield output that is independent of seasonal agricultural fluctuations, offering a distinct competitive advantage in the global market.
Mechanistic Insights into Friedel-Crafts Acylation and Oxidative Cleavage
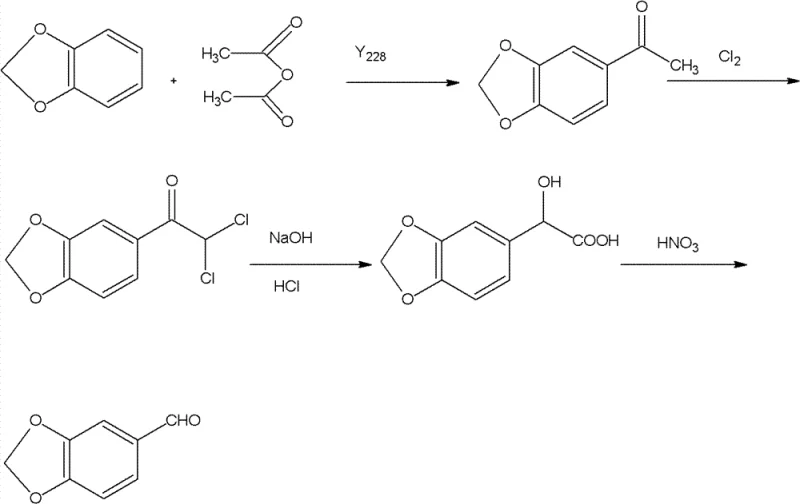
The core of this synthesis relies on a sophisticated interplay of electrophilic aromatic substitution and oxidative degradation mechanisms. The initial step involves a Friedel-Crafts acylation where 1,3-benzodioxole reacts with acetic anhydride in the presence of a Lewis acid catalyst, such as zinc chloride or aluminum chloride. This reaction selectively installs an acetyl group at the para-position relative to the electron-donating oxygen atoms of the dioxole ring, forming 3,4-methylenedioxyacetophenone. The choice of dichloroethane as a solvent and the temperature range of 40°C to 85°C are critical parameters that optimize the reaction kinetics while minimizing polyacylation or ring-opening side reactions. This step establishes the carbon skeleton required for the subsequent transformations, demonstrating high regioselectivity which is essential for maintaining high purity in the final product.
Following acylation, the process advances through a chlorination and hydrolysis sequence that effectively converts the methyl ketone into an aldehyde. The acetophenone intermediate undergoes alpha-chlorination with chlorine gas in acetic acid at low temperatures (20°C to 40°C) to yield a gem-dichloro species. This dichloro intermediate is then subjected to alkaline hydrolysis using liquid caustic soda, which facilitates the formation of a mandelic acid derivative. Finally, oxidative cleavage using nitric acid converts the alpha-hydroxy acid into the target aldehyde. This multi-step conversion is mechanistically elegant as it avoids over-oxidation to the carboxylic acid, a common pitfall in aldehyde synthesis. The careful control of pH during acidification and the specific concentration of nitric acid ensure that the reaction stops precisely at the aldehyde stage, preserving the integrity of the sensitive methylenedioxy ring.
How to Synthesize Piperonal Efficiently
Implementing this synthesis route requires precise control over reaction stoichiometry and thermal conditions to maximize yield and safety. The patent outlines a reproducible protocol where the molar ratios of reagents are tightly defined, ensuring that the catalytic cycles function optimally without excess waste. For technical teams looking to adopt this methodology, the process begins with the preparation of the acylated intermediate, followed by the critical chlorination step which must be monitored to prevent over-chlorination. The final oxidation stage demands careful temperature management to handle the exothermic nature of nitric acid reactions. Detailed standardized operating procedures for each unit operation, including workup and purification via vacuum rectification, are essential for translating this laboratory-scale success into commercial reality.
- Perform Friedel-Crafts acylation of 1,3-benzodioxole with acetic anhydride using zinc chloride or aluminum chloride catalyst in dichloroethane at 40-85°C.
- Conduct low-temperature chlorination of the resulting 3,4-methylenedioxyacetophenone in acetic acid solvent to form the dichloro intermediate.
- Hydrolyze the dichloro compound with caustic soda, acidify to isolate mandelic acid derivative, and oxidize with nitric acid to yield final Piperonal.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this total synthesis technology offers profound benefits that extend beyond simple chemical transformation. By shifting the raw material base from regulated botanical extracts to bulk petrochemical commodities, manufacturers can achieve significant cost reduction in flavor and fragrance intermediate manufacturing. The elimination of safrole removes the need for expensive compliance programs and specialized storage facilities required for controlled substances. Furthermore, the use of common reagents like acetic anhydride, chlorine, and nitric acid ensures that supply chains are robust and less susceptible to the geopolitical disruptions that often affect niche natural products. This stability allows for better long-term planning and inventory management, crucial for maintaining continuous production lines in high-volume facilities.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the low cost and high availability of the starting materials. Unlike safrole, which commands a premium due to extraction costs and regulatory scarcity, 1,3-benzodioxole and acetic anhydride are produced on a massive global scale, leading to inherently lower input costs. Additionally, the process avoids the use of precious metal catalysts or exotic oxidants, relying instead on inexpensive Lewis acids and mineral acids. The high yields reported in the patent examples, often exceeding 80% overall, further contribute to cost efficiency by maximizing the throughput of raw materials and minimizing waste disposal expenses associated with low-efficiency reactions.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by decoupling production from agricultural sources. Natural essential oil supplies are prone to variability due to weather patterns, crop diseases, and changing land-use policies in producing countries. In contrast, the synthetic route described here relies on a steady stream of industrial chemicals that are available year-round from multiple suppliers worldwide. This diversification of the supply base reduces the risk of stockouts and price spikes, ensuring that downstream customers in the pharmaceutical and food industries receive their orders on time. The ability to scale production up or down based on market demand without being constrained by harvest cycles provides a level of agility that is invaluable in dynamic markets.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by utilizing solvents and reagents that are easier to recover and recycle. The reaction conditions are mild, reducing energy consumption for heating and cooling compared to high-temperature pyrolysis or harsh oxidation methods. Moreover, the waste streams generated are primarily inorganic salts and acidic waters, which can be treated using standard effluent treatment protocols found in most modern chemical parks. This simplifies the permitting process for new facilities and reduces the environmental footprint of the manufacturing operation. The scalability is further evidenced by the straightforward unit operations involved—mixing, heating, filtration, and distillation—which are easily replicated in large-scale reactors without requiring specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific process parameters and advantages outlined in the patent documentation, providing clarity for stakeholders evaluating this route for potential adoption. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production portfolios.
Q: What are the primary advantages of this total synthesis route over traditional safrole-based methods?
A: This method avoids the regulatory restrictions and supply volatility associated with safrole, a controlled precursor. It utilizes commodity chemicals like acetic anhydride and chlorine, ensuring a more stable and compliant supply chain for industrial manufacturing.
Q: What purity levels can be achieved with this specific oxidative cleavage process?
A: The patent data indicates that through vacuum rectification and alcohol recrystallization, the final Piperonal product achieves GC purity levels exceeding 99.6%, meeting stringent requirements for fine fragrance and pharmaceutical applications.
Q: Is the chlorination step safe for large-scale commercial production?
A: Yes, the process specifies mild reaction temperatures between 20°C and 40°C for the chlorination step. This controlled low-temperature regime minimizes thermal risks and side reactions, making it highly suitable for safe scale-up in standard chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperonal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving needs of the global fine chemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering Piperonal with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest international standards for flavor and pharmaceutical applications. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing a secure foundation for your supply chain.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific business needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this synthetic route. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable, high-quality supply of Piperonal that drives value and innovation in your product formulations.
