Scalable Synthesis of 3-Fluorinated Alkyl Pyrazole Carboxylic Acids for Agrochemical Production
The global demand for high-performance succinate dehydrogenase inhibitor (SDHI) fungicides has driven significant innovation in the synthesis of their key building blocks. Patent CN103351339A discloses a robust and industrially viable preparation method for 3-fluorinated alkyl-1-substituted pyrazole-4-carboxylic acids, which serve as critical precursors for major agrochemical active ingredients such as Isopyrazam, Sedaxane, and Penthiopyrad. This technology addresses the longstanding challenge of efficiently removing the chlorine atom at the 5-position of the pyrazole ring without compromising the integrity of the sensitive difluoromethyl group or the carboxylic acid functionality. By utilizing a catalytic hydrogenation strategy under mild alkaline conditions, the process achieves high conversion rates and exceptional purity profiles, making it an attractive route for large-scale manufacturing. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the nuances of this dechlorination technology is essential for securing a stable supply chain for next-generation crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing 3-fluorinated pyrazole carboxylic acids often rely on harsh chemical conditions that can lead to significant impurity formation and yield loss. Conventional dehalogenation strategies might employ stoichiometric reducing agents or extreme temperatures that risk defluorination or decarboxylation, resulting in complex impurity spectra that are difficult and costly to purge during downstream processing. Furthermore, older methodologies frequently require anhydrous organic solvents and expensive noble metal catalysts in high loadings, which drastically inflates the cost of goods sold (COGS) and complicates waste management protocols. The inability to effectively control the reaction selectivity often necessitates multiple recrystallization steps, extending the production cycle time and reducing the overall throughput of the manufacturing facility. These inefficiencies create bottlenecks in the supply chain, particularly when scaling up to meet the volumetric demands of the global agrochemical market.
The Novel Approach
The methodology outlined in the patent introduces a streamlined catalytic hydrogenation process that operates under remarkably mild conditions, typically between 25°C and 80°C and at hydrogen pressures ranging from 1 atm to 30 atm. This approach leverages the synergistic effect of a heterogeneous catalyst, such as palladium carbon or Raney nickel, in the presence of a mild base to facilitate the selective cleavage of the carbon-chlorine bond. The versatility of the solvent system is a key advantage, allowing the reaction to proceed efficiently in water, alcohols, ethers, or esters, thereby providing manufacturers with the flexibility to optimize for cost or environmental compliance. By maintaining the reaction temperature well below the decomposition threshold of the difluoromethyl moiety, the process ensures that the structural integrity of the molecule is preserved, leading to a crude product with an HPLC content often exceeding 98%. This significant improvement in selectivity minimizes the need for extensive purification, directly translating to cost reduction in agrochemical intermediates manufacturing.
Mechanistic Insights into Catalytic Hydrodechlorination
The core of this synthetic breakthrough lies in the mechanism of catalytic hydrodechlorination on the metal surface. When the 5-chloro-pyrazole derivative interacts with the activated catalyst (Pd/C or Raney-Ni) under a hydrogen atmosphere, the chlorine atom is displaced by a hydrogen atom via an oxidative addition-reductive elimination cycle. The presence of an alkali base, such as sodium hydroxide or potassium carbonate, plays a dual role: it neutralizes the hydrochloric acid generated in situ, preventing acid-catalyzed degradation of the pyrazole ring, and it may also assist in activating the catalyst surface or the substrate for more efficient hydrogen uptake. The choice of catalyst loading, typically ranging from 0.1% to 20% by weight relative to the substrate, allows for fine-tuning the reaction kinetics to balance between reaction speed and catalyst consumption. This mechanistic understanding is crucial for process chemists aiming to replicate the high purity levels reported in the patent embodiments, where residual starting material is consistently driven below 0.2%.
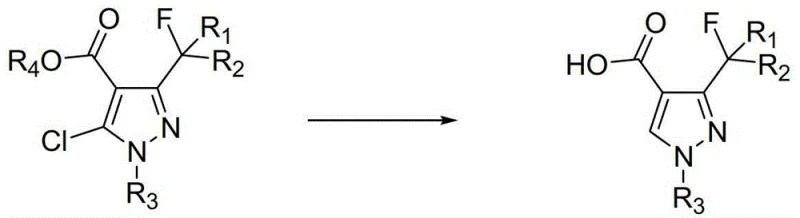
Furthermore, the stability of the difluoromethyl group under these specific hydrogenation conditions is a testament to the selectivity of the catalyst system. Unlike non-selective hydrogenation catalysts that might reduce the double bonds within the heterocyclic ring or cleave the carbon-fluorine bonds, the optimized conditions described ensure that only the carbon-chlorine bond is targeted. This selectivity is vital for maintaining the biological activity of the final fungicide, as the fluorine atoms are often critical for metabolic stability and binding affinity within the target enzyme. The ability to run this reaction in aqueous media further suggests that the transition state is stabilized by the solvent environment, potentially through hydrogen bonding interactions that favor the desired pathway over side reactions. For technical teams evaluating this route, the mechanistic robustness offers a high degree of confidence in the reproducibility of the process across different batch sizes and reactor configurations.
How to Synthesize 3-Difluoromethyl-1-methylpyrazole-4-carboxylic Acid Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity, requiring standard hydrogenation equipment such as an autoclave rather than specialized exotic reactors. The process begins with the dissolution or suspension of the chloro-precursor in the chosen solvent system, followed by the addition of the base and the catalyst. Once the reactor is sealed and purged with hydrogen, the reaction proceeds autonomously with monitoring via HPLC to determine the endpoint, which is typically achieved within 3 to 15 hours depending on the specific solvent and temperature profile selected. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry and workup procedures required to achieve the reported 99% HPLC purity. This straightforward protocol minimizes the training burden on plant operators and reduces the risk of human error during the charging and discharging phases of the batch cycle.
- Charge an autoclave with the chloro-pyrazole starting material, water or organic solvent, and a base such as sodium hydroxide or potassium carbonate.
- Add the catalyst (10% Palladium on Carbon or Raney Nickel) and pressurize the reactor with hydrogen gas to 1-30 atm.
- Heat the mixture to 25-80°C until reaction completion, then filter to recover the catalyst and acidify the filtrate to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial strategic advantages for organizations focused on cost reduction in agrochemical intermediates manufacturing and supply chain resilience. The ability to utilize water as a primary solvent in several embodiments eliminates the need for expensive and hazardous organic solvents, significantly lowering raw material costs and simplifying wastewater treatment protocols. Moreover, the catalysts employed, particularly Raney Nickel, are considerably more economical than many specialized homogeneous catalysts, and the heterogeneous nature of both Pd/C and Raney-Ni allows for easy filtration and potential recycling, further driving down the variable cost per kilogram. The mild reaction conditions (low pressure and temperature) reduce the energy consumption of the process and lower the safety risks associated with high-pressure hydrogenation, facilitating easier regulatory approval and insurance underwriting for manufacturing sites.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the use of inexpensive, commodity-grade reagents like sodium hydroxide and common solvents like methanol or ethanol drastically simplify the bill of materials. By avoiding the use of stoichiometric metallic reductants which generate large amounts of metal salt waste, the process reduces waste disposal costs and environmental levies. The high yield and purity reported in the embodiments mean that less starting material is wasted, maximizing the atom economy of the transformation and ensuring that the cost of goods remains competitive even in a volatile raw material market.
- Enhanced Supply Chain Reliability: The reliance on widely available catalysts and solvents ensures that the supply chain is not vulnerable to shortages of niche reagents. The robustness of the reaction across a wide range of temperatures and pressures provides manufacturing flexibility, allowing production to continue even if specific utility conditions fluctuate slightly. This operational flexibility is critical for maintaining continuous supply to downstream formulators, reducing the risk of stockouts that could disrupt the agricultural season. Additionally, the short reaction times observed in organic solvent systems (as low as 3 hours) enable faster turnaround times for production batches, increasing the overall capacity of the manufacturing asset.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in liter-scale autoclaves with parameters that translate linearly to industrial reactors. The option to run the reaction in water aligns perfectly with green chemistry principles, reducing the volatile organic compound (VOC) emissions associated with the manufacturing process. This environmental profile makes the technology future-proof against tightening environmental regulations, ensuring long-term operational continuity without the need for costly retrofits to emission control systems. The simple workup involving filtration and acidification avoids complex distillation or chromatography steps, making the scale-up from pilot plant to commercial production seamless and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrodechlorination technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for process evaluation. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their existing supply chains and product portfolios.
Q: What catalysts are suitable for the hydrodechlorination of pyrazole intermediates?
A: The patent specifies that both 10% Palladium on Carbon (Pd/C) and Raney Nickel are highly effective catalysts for this transformation, offering flexibility in cost and activity profiles.
Q: Can this reaction be performed in aqueous media?
A: Yes, the process is versatile and supports aqueous systems using water as the primary solvent, which significantly reduces environmental impact and solvent recovery costs compared to purely organic systems.
Q: What is the typical purity achievable with this method?
A: Experimental embodiments demonstrate that HPLC content can reach approximately 99%, with residual starting material (chloro-derivative) reduced to less than 0.2%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Difluoromethyl Pyrazole Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the efficacy and safety of final agrochemical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is executed with precision. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 3-fluorinated alkyl pyrazole carboxylic acid meets the exacting standards required for SDHI fungicide synthesis. Our commitment to quality assurance means that our clients can rely on consistent batch-to-batch reproducibility, minimizing the risk of formulation issues or regulatory delays.
We invite you to contact our technical procurement team to discuss how we can support your specific volume requirements and timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can lower your total landed cost. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-purity 3-difluoromethyl pyrazole carboxylic acid reliably and efficiently. Partner with us to secure a sustainable and cost-effective supply chain for your next-generation crop protection solutions.
