Advanced Manufacturing of Difluoromethylpyrazole Intermediates for Global Agrochemical Supply Chains
Advanced Manufacturing of Difluoromethylpyrazole Intermediates for Global Agrochemical Supply Chains
The global demand for high-performance fungicides continues to drive innovation in the synthesis of heterocyclic intermediates, particularly those containing fluorine motifs which enhance metabolic stability and bioavailability. Patent CN101535274B introduces a transformative methodology for preparing difluoromethyl-substituted pyrazol-4-yl carboxylates, a critical scaffold in modern crop protection chemistry. Unlike traditional approaches that struggle with the high cost and scarcity of difluorinated starting materials, this invention leverages a clever reductive defluorination strategy starting from abundant trifluoro-precursors. By employing a metal-mediated silylation followed by hydrazine cyclization, the process achieves exceptional regioselectivity and yield. This technical breakthrough not only resolves long-standing synthetic bottlenecks but also offers a robust pathway for reliable agrochemical intermediate supplier networks seeking to optimize their manufacturing portfolios.
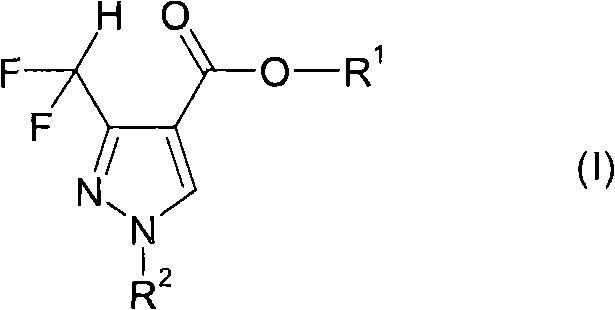
The core innovation lies in the structural versatility of Formula I, where R1 and R2 groups can be extensively modified to tune the physicochemical properties of the final active ingredient. The ability to access these structures efficiently allows pharmaceutical and agrochemical companies to accelerate their pipeline development. For procurement teams, understanding the underlying chemistry is crucial, as it dictates the availability and pricing of the raw materials. This patent effectively decouples the production of valuable difluoromethyl-pyrazoles from the volatile market of specialized difluoro-reagents, establishing a more stable foundation for cost reduction in fungicide manufacturing. The following analysis details the mechanistic advantages and commercial implications of adopting this superior synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-difluoromethyl-pyrazole derivatives has been hindered by the reliance on 2,2-difluoroacetoacetate as the primary building block, as described in prior art such as WO 92/12970. This starting material is notoriously expensive and difficult to procure in bulk quantities, creating a significant barrier to entry for large-scale production. Furthermore, the conventional pathway often involves multiple steps including hydrolysis and conversion to acid chlorides before final amidation, which cumulatively erodes overall yield and increases waste generation. The economic inefficiency is compounded by the safety hazards associated with handling reactive fluorinating agents required to install the difluoromethyl group at later stages. For supply chain heads, these factors translate into extended lead times and vulnerability to raw material shortages, making the conventional route unsustainable for meeting the growing global demand for high-purity OLED material and agrochemical precursors.
The Novel Approach
In stark contrast, the method disclosed in CN101535274B circumvents these issues by utilizing 4,4,4-trihalo-substituted acetoacetate derivatives, specifically trifluoro-analogues, which are commodity chemicals with established supply chains. The process initiates with a reductive reaction using metals from Groups 1, 2, 3, 4, or 12, such as magnesium or zinc, in the presence of a chlorosilane. This unique combination facilitates a controlled defluorination that generates a reactive silylated enol intermediate in situ. Subsequent reaction with hydrazines proceeds with high regioselectivity to directly afford the desired pyrazole-4-carboxylates. This streamlined approach eliminates the need for expensive difluoro-starting materials and harsh fluorination steps, thereby drastically simplifying the operational complexity. For a reliable agrochemical intermediate supplier, this translates to a more resilient production model capable of supporting commercial scale-up of complex polymer additives and fine chemicals with greater consistency.
Mechanistic Insights into Reductive Silylation and Cyclization
The heart of this technology is the metal-mediated reductive silylation step, which converts the trifluoromethyl ketone moiety into a difluoromethylene unit while simultaneously protecting the enol functionality. When the trifluoroacetoacetate derivative (Formula II) is treated with magnesium and trimethylsilyl chloride in an aprotic solvent like DMF or NMP, a single-electron transfer occurs. This generates a radical anion that undergoes defluorination to form a silylated enol species, designated as Formula A in the patent documentation. This intermediate is crucial because it stabilizes the reactive carbon center against premature hydrolysis or side reactions. The presence of the silyl group directs the subsequent nucleophilic attack by the hydrazine, ensuring that cyclization occurs predominantly at the 4-position rather than the 3-position. This mechanistic control is vital for R&D directors focused on impurity profiles, as it minimizes the formation of regioisomers that are difficult to separate.
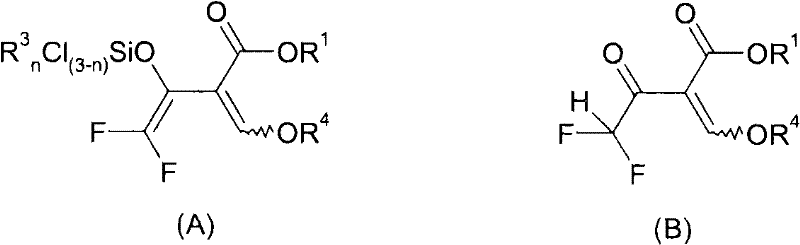
Following the formation of the silylated intermediate, the reaction mixture is treated with a hydrazine derivative (Formula III). Depending on the specific conditions, the silyl group may be hydrolyzed to form intermediate B before cyclization, or the cyclization may proceed directly. The patent highlights that performing the reaction in the presence of water or protic solvents during the second step facilitates the removal of the silyl protecting group and drives the condensation reaction to completion. The choice of metal is also critical; magnesium is particularly effective when the leaving group X is fluorine, while zinc is preferred for chlorine or bromine variants. This flexibility allows chemists to fine-tune the reaction kinetics based on the specific substrate. Understanding these nuances is essential for optimizing the commercial scale-up of complex heterocycles, ensuring that the process remains robust even when transitioning from laboratory glassware to multi-ton reactors.
How to Synthesize Difluoromethylpyrazolyl Carboxylates Efficiently
Implementing this synthesis requires careful attention to solvent dryness and reagent stoichiometry to maximize the formation of the silylated intermediate. The process begins with the preparation of the enol ether precursor (Formula II) from tri(halo)acetoacetates and orthoformates, a well-established condensation reaction. Once Formula II is secured, it is introduced to a suspension of activated metal and chlorosilane in a dry polar aprotic solvent. Temperature control is paramount during this exothermic addition, typically maintained between -10°C and 60°C to prevent decomposition. After the reductive step is complete, excess silane is removed, and the crude intermediate is reacted with the hydrazine component. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Prepare the 4,4,4-trihalo-substituted acetoacetate derivative (Formula II) by reacting tri(halo)acetoacetate with an orthoformate.
- React Formula II with a chlorosilane and a reducing metal (Magnesium or Zinc) in an aprotic solvent to form a silylated enol intermediate.
- Treat the reaction mixture with a hydrazine derivative (Formula III) to effect cyclization and form the final pyrazole carboxylate (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented process offers profound strategic benefits for organizations managing the procurement of specialty chemicals and agrochemical intermediates. The primary advantage stems from the substitution of scarce, high-cost difluoro-reagents with abundant trifluoro-commodities. This shift fundamentally alters the cost structure of the synthesis, removing a major bottleneck that has historically constrained production capacity. By relying on raw materials that are produced on a massive industrial scale for other applications, manufacturers can secure long-term supply contracts at stable prices. This stability is critical for supply chain heads who must mitigate the risks associated with volatile raw material markets. Furthermore, the simplified reaction sequence reduces the number of unit operations required, leading to lower energy consumption and reduced solvent usage, which aligns with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive 2,2-difluoroacetoacetate represents a direct and significant saving in bill of materials. Since the new route utilizes 4,4,4-trifluoroacetoacetate, which is a widely available bulk chemical, the input costs are substantially lower. Additionally, the high regioselectivity of the cyclization step reduces the burden on downstream purification processes such as chromatography or repeated crystallization. This efficiency gain means that less material is lost as waste, further driving down the cost per kilogram of the final active pharmaceutical ingredient or agrochemical. The avoidance of specialized fluorinating agents also removes the need for costly corrosion-resistant equipment, allowing the use of standard stainless steel reactors.
- Enhanced Supply Chain Reliability: Sourcing difluorinated building blocks often involves dealing with a limited number of niche suppliers, creating single points of failure in the supply chain. In contrast, trifluoroacetoacetates and orthoformates are produced by numerous chemical manufacturers globally, ensuring a diversified and resilient supply base. This abundance reduces the risk of production stoppages due to raw material shortages. Moreover, the reaction conditions described in the patent are mild and do not require extreme pressures or cryogenic temperatures, making the process easier to transfer between different manufacturing sites. This flexibility ensures continuity of supply even if one facility faces operational challenges, a key consideration for reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reagents and solvents that are compatible with existing large-scale infrastructure. The use of magnesium or zinc as reducing agents generates inorganic salts as byproducts, which are easier to handle and dispose of compared to the heavy metal waste often associated with transition metal catalysis. The high conversion rates reported in the patent examples indicate that the reaction proceeds cleanly, minimizing the generation of hazardous organic waste streams. This environmental profile simplifies regulatory compliance and reduces the costs associated with waste treatment. For companies aiming to expand their production capacity, this route offers a clear path to increasing output without proportional increases in environmental footprint or capital expenditure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and claims within the patent specification, providing a reliable reference for process chemists and engineers. Understanding these details helps in assessing the feasibility of integrating this technology into existing manufacturing workflows.
Q: Why is the trifluoro-route superior to the conventional difluoro-route?
A: Conventional methods rely on 2,2-difluoroacetoacetate, which is expensive and difficult to source. This patent utilizes readily available 4,4,4-trifluoroacetoacetates, significantly lowering raw material costs while maintaining high regioselectivity through a controlled reductive defluorination mechanism.
Q: What is the role of the silane compound in this synthesis?
A: The silane compound, such as trimethylsilyl chloride, reacts with the metal-reduced intermediate to form a stable silylated enol (Formula A). This prevents premature hydrolysis and controls the subsequent cyclization with hydrazine, ensuring high yields of the desired 4-carboxylate isomer over the 3-carboxylate byproduct.
Q: Can this process be scaled for industrial production?
A: Yes, the process operates under atmospheric pressure and moderate temperatures (-10°C to 60°C), utilizing common solvents like DMF or NMP. The use of standard reducing metals like magnesium or zinc makes it highly amenable to large-scale commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylpyrazolyl Carboxylates Supplier
The technological advancements detailed in CN101535274B represent a significant leap forward in the efficient production of fluorinated heterocycles. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier agrochemical and pharmaceutical companies. We understand that moving from a patent concept to a commercial reality requires not just chemical expertise but also robust project management and supply chain coordination.
We invite you to contact our technical procurement team to discuss how we can support your specific needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need specific COA data to validate our capabilities, we are ready to assist. Let us collaborate to optimize your route feasibility assessments and secure a sustainable supply of these critical intermediates for your future projects.
