Advanced Manufacturing of Oxiracetam: A Novel Route for High-Purity Nootropic Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for cognitive enhancers, and the preparation of oxiracetam (4-hydroxylethylpyrrolidone-2-acetamide) stands as a critical challenge in nootropic drug manufacturing. Patent CN101693685A discloses a groundbreaking method that fundamentally shifts the production paradigm by utilizing chloroacetoacetic ester and aminoacetonitrile hydrochloride as primary raw materials. This innovative approach addresses the longstanding inefficiencies of traditional synthesis, offering a pathway that combines hydrogenation, substitution, and cyclization steps to achieve the objective product with remarkable efficiency. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize supply chains for brain function improving agents. The technical breakthrough lies not just in the chemical transformation but in the strategic selection of starting materials that are both inexpensive and easily accessible, thereby laying the foundation for substantial cost reduction in pharmaceutical intermediates manufacturing.
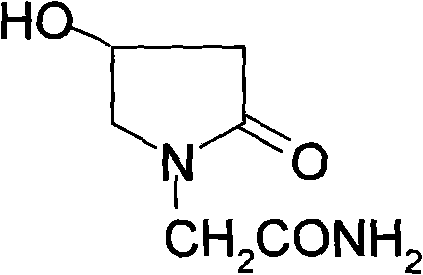
The limitations of conventional methods have long plagued the production of high-purity oxiracetam, creating bottlenecks for reliable pharmaceutical intermediates suppliers. Prior art, such as the method described in Japanese Patent JP62026267, relies on the direct reaction of 3-hydroxy-4-halo butanoic acid derivatives with glycine amide equivalents. This legacy approach is fraught with severe drawbacks, primarily the generation of complex side reaction products that complicate downstream purification. The resulting product mixture often fails to reach the stringent purity requirements necessary for medicinal use, necessitating extra care and extensive processing that drives up costs. Furthermore, the reaction time in these conventional methods is excessively long, often exceeding 20 hours, which severely limits throughput and increases energy consumption. The ultimate yield in these older processes is very low, rendering them economically unviable for satisfying the needs of modern commercial scale production where efficiency and speed are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on 3-hydroxy-4-halo butanoic acid derivatives introduces significant variability into the manufacturing process, making it difficult to maintain consistent quality control standards required by global regulatory bodies. The complexity of the reaction mixture means that impurities are structurally similar to the target molecule, making them notoriously difficult to remove without sacrificing overall yield. This impurity profile poses a risk to patient safety and complicates the validation process for new drug applications. Additionally, the prolonged reaction times tie up reactor capacity, reducing the overall asset utilization of the manufacturing facility. For supply chain heads, this translates to longer lead times and reduced flexibility in responding to market demand fluctuations. The inability to satisfy commercial scale production needs with these legacy methods forces manufacturers to seek alternative, more efficient synthetic strategies that can deliver high-purity oxiracetam consistently.
The Novel Approach
In stark contrast, the novel approach detailed in CN101693685A utilizes chloroacetoacetic ester as a versatile building block, initiating a sequence that is both chemically elegant and industrially practical. By employing a reduction step followed by substitution and cyclization, the process avoids the messy side reactions associated with the direct coupling of halo-acids. The technical process is simple, requiring standard unit operations that are easily implemented in existing multipurpose chemical plants. Compared with the prior art, the yield rate is significantly higher, with total recovery rates exceeding 60% in optimized embodiments, which is a dramatic improvement over the low yields of previous methods. The product quality is demonstrably better, with purity levels reaching above 98%, thus being more suitable for the industrialized scale production required by major pharmaceutical companies. This shift in methodology directly supports the goal of cost reduction in API manufacturing by minimizing waste and maximizing output per batch.
Mechanistic Insights into Reductive Cyclization Strategy
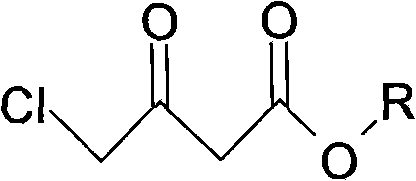
The core of this synthesis lies in the precise control of the reduction and cyclization steps, which dictate the stereochemistry and purity of the final pyrrolidone ring. The process begins with the reduction of chloroacetoacetic ester using a hydroborate agent, specifically potassium borohydride, at controlled low temperatures ranging from -5°C to 15°C, with optimal results observed between 3°C and 8°C. This selective reduction converts the ketone functionality into a secondary alcohol without affecting the ester or the chloro group, yielding a chloro-3-hydroxybutyrate intermediate. The choice of alcoholic solvent, such as ethanol or methanol, is critical here, as it solubilizes the reactants while stabilizing the intermediate species. Following this, the substitution reaction with aminoacetonitrile hydrochloride or sulfate occurs at elevated temperatures between 60°C and 100°C. This step introduces the nitrogen atom necessary for the ring closure, forming an amino-ester intermediate that is poised for cyclization.
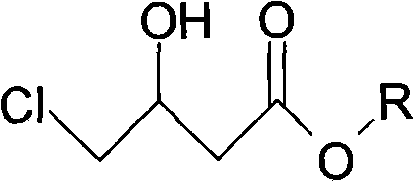
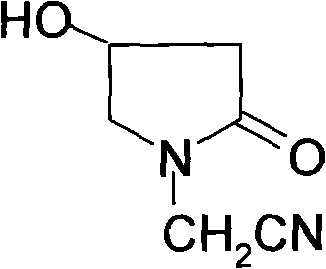
The subsequent cyclization and dealcoholization step is where the pyrrolidone ring is formed, creating the core scaffold of the oxiracetam molecule. Conducted in an alcoholic solvent at temperatures between 65°C and 85°C, this intramolecular reaction closes the ring to form 4-hydroxypyrrolidone-2-acetonitrile. The mechanism involves the nucleophilic attack of the amine nitrogen on the ester carbonyl, followed by the elimination of the alcohol moiety. Finally, the nitrile group is hydrolyzed under mild alkaline conditions using mineral alkalis like sodium hydroxide or potassium hydroxide at 40°C to 60°C. This hydrolysis converts the nitrile into the primary amide functionality found in the final product. Impurity control is inherently built into this pathway; by avoiding the harsh conditions and unstable intermediates of the old method, the formation of polymeric byproducts and structural isomers is minimized. The use of specific molar ratios, such as 1:4.5 for the borohydride to ester, ensures complete conversion while preventing over-reduction or decomposition, thereby securing a clean impurity profile.
How to Synthesize Oxiracetam Efficiently
The synthesis of oxiracetam via this patented route offers a streamlined protocol that balances reaction kinetics with operational simplicity, making it ideal for process chemists looking to implement robust manufacturing procedures. The detailed standardized synthesis steps involve a sequential four-stage process that transforms cheap bulk chemicals into a high-value nootropic intermediate. The initial reduction sets the stage for high yield, followed by a tandem substitution-cyclization sequence that builds molecular complexity rapidly. The final hydrolysis is gentle, preserving the integrity of the sensitive hydroxyl group on the pyrrolidone ring. For a comprehensive guide on executing this synthesis with precision, refer to the standardized protocol below.
- Reduce chloroacetoacetic ester with potassium borohydride in alcoholic solvent at 3-8°C to form chloro-3-hydroxybutyrate.
- Perform substitution reaction with aminoacetonitrile hydrochloride at 65-85°C to generate the amino-ester intermediate.
- Execute dealcoholization and cyclization at 65-85°C to form 4-hydroxypyrrolidone-2-acetonitrile.
- Hydrolyze the nitrile intermediate with mineral alkali at 50°C to obtain the final oxiracetam product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative advantages that extend beyond mere chemical yield. The primary benefit is the drastic simplification of the raw material portfolio; chloroacetoacetic ester and aminoacetonitrile salts are commodity chemicals with stable global supply chains, unlike the specialized and often expensive 3-hydroxy-4-halo butanoic acid derivatives required by older methods. This shift significantly mitigates supply risk and ensures continuity of supply even during market fluctuations. Furthermore, the operational simplicity of the process reduces the burden on manufacturing teams, allowing for faster batch turnover and better utilization of capital equipment. The elimination of complex purification steps required to remove stubborn impurities from the old process translates directly into lower operating expenses and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the use of inexpensive starting materials and the high efficiency of the reaction sequence. By eliminating the need for expensive, hard-to-source precursors and reducing the reaction time from over 20 hours to a fraction of that, the overall cost of goods sold is substantially lowered. The high yield of the reduction step (approximately 88%) and the robust total recovery of over 60% mean that less raw material is wasted per kilogram of final product. Additionally, the avoidance of transition metal catalysts or exotic reagents removes the cost associated with catalyst recovery and heavy metal testing, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key raw materials are widely produced industrial chemicals rather than niche fine chemicals. This abundance ensures that procurement teams can negotiate better pricing and secure long-term contracts without fear of supply interruption. The shorter reaction times also mean that production lead times are significantly reduced, allowing the supply chain to be more responsive to sudden spikes in demand for nootropic formulations. The robustness of the process against minor variations in conditions ensures consistent output quality, reducing the risk of batch failures that could disrupt the supply of critical API intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard solvents like ethanol and water which are environmentally benign and easy to recover. The absence of hazardous byproducts and the use of mild reaction conditions simplify waste treatment protocols, ensuring compliance with increasingly strict environmental regulations. The ability to run the substitution and cyclization steps successively in the same solvent system reduces solvent consumption and energy usage for distillation. This green chemistry profile not only lowers environmental fees but also aligns with the sustainability goals of major pharmaceutical partners, making the manufacturer a preferred vendor in a eco-conscious market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxiracetam synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and product quality. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing lines or for procurement teams assessing supplier qualifications.
Q: What are the advantages of this oxiracetam synthesis method over prior art?
A: Unlike the conventional method using 3-hydroxy-4-halo butanoic acid which suffers from complex side reactions and low purity, this novel route utilizes cheap chloroacetoacetic ester. It offers a simpler technical process, significantly higher total recovery (over 60%), and superior product quality suitable for industrial scale production.
Q: What are the critical reaction conditions for the reduction step?
A: The reduction of chloroacetoacetic ester requires strict temperature control between 3°C and 8°C using potassium borohydride in an alcoholic solvent. Maintaining this low temperature range is crucial for maximizing the yield of the chloro-3-hydroxybutyrate intermediate, which was achieved at approximately 88% in optimized embodiments.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the patent explicitly states the method is more suitable for industrialized scale production compared to prior art. The use of readily available raw materials, simple operational steps, and the elimination of prolonged reaction times (reducing from over 20 hours in old methods) facilitates efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of effective cognitive health solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of oxiracetam meets the highest international standards. Our commitment to technical excellence allows us to deliver complex nootropic intermediates with the consistency and reliability that global pharmaceutical partners demand.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your product portfolio and drive value for your organization.
