Advanced Synthesis of 2-Amino-1,3,4-Thiadiazoles and Selenodiazoles for Commercial Scale-Up
Advanced Synthesis of 2-Amino-1,3,4-Thiadiazoles and Selenodiazoles for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for safer, more efficient, and scalable synthetic methodologies. Patent CN114805243B, published in June 2023, introduces a transformative approach to the preparation of 1,3,4-thiadiazole and 1,3,4-selenodiazole derivatives, which are critical scaffolds in modern medicinal chemistry and agrochemical development. This intellectual property discloses a robust protocol utilizing benzenesulfonylhydrazone and potassium thiocyanate or potassium selenocyanate as key starting materials, activated by N-chlorosuccinimide (NCS) under acidic conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward, replacing hazardous reagents with benign alternatives while maintaining high yields and structural diversity. The strategic implementation of this pathway allows for the streamlined production of bioactive compounds, including the successful synthesis of furidiazine, demonstrating its practical utility in complex molecule assembly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,3,4-thiadiazole and 1,3,4-selenodiazole rings has been plagued by significant operational and safety challenges that hinder industrial adoption. Traditional routes predominantly rely on hydrazine hydrate to introduce the nitrogen atoms at the 3 and 4 positions, coupled with volatile and malodorous sulfur or selenium sources such as isothiocyanates, thiosemicarbazides, or isoselenocyanates. As illustrated in prior art like Formula 1, these reagents not only possess intense, unpleasant odors that complicate workplace safety but also exhibit high toxicity, particularly hydrazine hydrate, which is a known carcinogen. Furthermore, earlier attempts to utilize odorless sulfur sources like potassium thiocyanate often necessitated extreme reaction conditions, such as heating in sealed tubes at temperatures exceeding 100°C for prolonged periods up to 12 hours. Such harsh parameters severely limit the application prospects of these reactions, creating bottlenecks in commercial scale-up of complex heterocyclic scaffolds due to pressure vessel requirements and energy inefficiency.
The Novel Approach
In stark contrast to these legacy limitations, the methodology disclosed in CN114805243B offers a remarkably mild and operationally simple alternative that leverages inexpensive and odorless inorganic salts. By employing benzenesulfonylhydrazone alongside potassium thiocyanate (for sulfur) or potassium selenocyanate (for selenium) in the presence of N-chlorosuccinimide and a Lewis acid, the reaction proceeds smoothly under standard reflux conditions or even at room temperature. 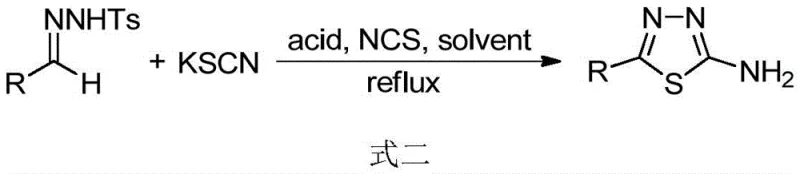 This novel approach eliminates the need for toxic hydrazine hydrate and foul-smelling organic isothiocyanates, thereby drastically improving the working environment and reducing the burden on waste treatment systems. The versatility of this system is evident in its ability to accommodate a wide range of substituents, including alkyl, aryl, and heteroaryl groups, ensuring that it serves as a universal platform for generating high-purity 1,3,4-thiadiazole derivatives required for downstream drug discovery and agricultural chemical manufacturing.
This novel approach eliminates the need for toxic hydrazine hydrate and foul-smelling organic isothiocyanates, thereby drastically improving the working environment and reducing the burden on waste treatment systems. The versatility of this system is evident in its ability to accommodate a wide range of substituents, including alkyl, aryl, and heteroaryl groups, ensuring that it serves as a universal platform for generating high-purity 1,3,4-thiadiazole derivatives required for downstream drug discovery and agricultural chemical manufacturing.
Mechanistic Insights into NCS-Promoted Cyclization
The success of this transformation hinges on the synergistic interaction between the oxidant N-chlorosuccinimide (NCS) and the Lewis acid catalyst, which facilitates the activation of the sulfur or selenium source and the subsequent cyclization event. In the thiadiazole synthesis, the Lewis acid, such as boron trifluoride etherate, likely coordinates with the hydrazone nitrogen, increasing its electrophilicity and promoting nucleophilic attack by the thiocyanate anion. Simultaneously, NCS acts as a mild chlorinating and oxidizing agent, facilitating the removal of the sulfonyl group and driving the oxidative cyclization to form the stable five-membered heterocyclic ring. This mechanism avoids the formation of unstable intermediates often seen in hydrazine-based routes, leading to cleaner reaction profiles and simplified purification processes. For the selenodiazole variant, the use of scandium triflate or similar Lewis acids at room temperature suggests an even lower activation energy barrier, allowing the reaction to proceed without external heating, which is a rare and valuable feature for selenium chemistry that typically requires vigorous conditions.
From an impurity control perspective, this mechanistic pathway offers distinct advantages by minimizing side reactions associated with free hydrazine or volatile isothiocyanates. The use of solid potassium salts ensures a controlled release of the chalcogen source, preventing the rapid, uncontrolled exotherms that can lead to polymerization or decomposition byproducts. Moreover, the specific stoichiometry defined in the patent, such as a molar ratio of benzenesulfonylhydrazone to potassium thiocyanate to acid to NCS of 1:2:0.5:2, is optimized to drive the reaction to completion while suppressing over-oxidation or chlorination of the aromatic rings. This precision in reagent balancing is crucial for achieving the stringent purity specifications demanded by regulatory bodies for API intermediates, ensuring that the final product contains minimal levels of genotoxic impurities or heavy metal residues that might arise from alternative transition-metal catalyzed methods.
How to Synthesize 2-Amino-1,3,4-Thiadiazole Efficiently
To implement this cutting-edge technology in a laboratory or pilot plant setting, operators must adhere to a precise sequence of addition and environmental controls to maximize yield and safety. The process begins with the dissolution of the benzenesulfonylhydrazone precursor in a suitable alcohol solvent like tert-butanol under an inert argon atmosphere to prevent moisture interference. Following this, the odorless potassium thiocyanate and the oxidant NCS are introduced, followed by the careful addition of the Lewis acid catalyst to initiate the cyclization. 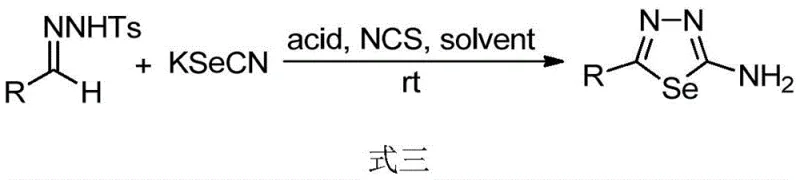 The reaction mixture is then heated to reflux until monitoring indicates the complete disappearance of the starting hydrazone, typically within a timeframe of 0.5 to 4 hours depending on the substrate electronics. Detailed standardized synthesis steps see the guide below.
The reaction mixture is then heated to reflux until monitoring indicates the complete disappearance of the starting hydrazone, typically within a timeframe of 0.5 to 4 hours depending on the substrate electronics. Detailed standardized synthesis steps see the guide below.
- Dissolve benzenesulfonylhydrazone in tert-butanol under argon protection and add potassium thiocyanate.
- Introduce N-chlorosuccinimide (NCS) and a Lewis acid catalyst such as BF3·OEt2 to the reaction mixture.
- Reflux the mixture until completion, then extract with ethyl acetate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation strategies that directly impact the bottom line. By shifting away from volatile, malodorous, and highly toxic reagents like hydrazine hydrate and isothiocyanates, facilities can significantly reduce the costs associated with specialized containment infrastructure, personal protective equipment, and hazardous waste disposal. The reliance on commodity chemicals such as potassium thiocyanate and N-chlorosuccinimide ensures a stable and cost-effective supply chain, insulating production schedules from the volatility often seen with niche organic reagents. Furthermore, the elimination of high-pressure sealed tube requirements allows for the utilization of standard glass-lined reactors, facilitating a seamless transition from gram-scale discovery to multi-ton commercial production without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and difficult-to-handle organic sulfur sources with inexpensive inorganic salts. Since potassium thiocyanate and potassium selenocyanate are bulk commodities, their procurement costs are substantially lower than custom-synthesized isothiocyanates, leading to direct material cost savings. Additionally, the mild reaction conditions, particularly the room temperature protocol for selenodiazoles, result in significant energy conservation by eliminating the need for prolonged heating cycles. The simplified workup procedure, involving standard extraction and crystallization rather than complex distillation or high-vacuum purification, further reduces utility consumption and labor hours, contributing to a leaner and more profitable manufacturing model for high-value heterocyclic intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents that are not subject to the same regulatory restrictions as hydrazine derivatives. Potassium thiocyanate and NCS are produced by multiple global suppliers, reducing the risk of single-source dependency and ensuring continuous availability even during market disruptions. The robustness of the reaction across a broad substrate scope means that a single standardized protocol can be applied to synthesize a diverse library of intermediates, simplifying inventory management and reducing the need for specialized training for different chemical transformations. This flexibility allows manufacturers to respond rapidly to changing market demands for specific API precursors without retooling production lines or sourcing new, exotic starting materials.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this technology aligns perfectly with green chemistry principles by minimizing the generation of hazardous waste and avoiding the use of carcinogenic substances. The absence of strong odors improves site-wide air quality and community relations, while the aqueous workup generates waste streams that are easier to treat compared to those containing organic sulfides or selenium volatiles. The scalability is inherently high because the reaction does not rely on sensitive catalysts that might deactivate on large scales or require strict exclusion of oxygen beyond standard inertion. This makes the process ideal for commercial scale-up of complex heterocyclic scaffolds, ensuring that production can be ramped up to meet global demand while maintaining full compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, providing clarity for stakeholders evaluating its potential integration into their existing portfolios. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, highlighting the practical benefits over traditional methods. Understanding these nuances is essential for making informed decisions about process development and vendor selection in the competitive landscape of fine chemical intermediates.
Q: What are the safety advantages of using potassium thiocyanate over isothiocyanates?
A: Potassium thiocyanate is a solid, odorless, and low-toxicity reagent, whereas traditional isothiocyanates are volatile liquids with pungent, irritating odors that pose significant inhalation risks and require specialized containment equipment.
Q: Does this method require high-pressure sealed tubes for reaction?
A: No, unlike previous iodine-promoted methods that required sealed tubes at temperatures exceeding 100°C, this novel protocol operates efficiently under standard reflux conditions or even at room temperature for selenodiazoles, greatly simplifying equipment requirements.
Q: What is the substrate scope for this cyclization reaction?
A: The method demonstrates broad compatibility with various substrates including alkyl, phenyl, substituted phenyl, furyl, thienyl, and pyridyl groups, making it highly versatile for synthesizing diverse pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-1,3,4-Thiadiazole Supplier
As the pharmaceutical and agrochemical industries continue to demand higher purity and more sustainable manufacturing practices, NINGBO INNO PHARMCHEM stands ready to leverage this advanced technology for your specific project needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 2-amino-1,3,4-thiadiazole or selenodiazole meets the exacting standards required for GMP manufacturing. Our commitment to quality assurance means that you can rely on us not just as a vendor, but as a strategic partner dedicated to the success of your drug development pipeline.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages this method offers over your current supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make data-driven decisions that optimize both your R&D timelines and your overall production costs. Let us help you secure a reliable supply of high-quality intermediates that drive your business forward.
