Advancing Pharmaceutical Intermediate Manufacturing via Electrochemical Oxidation Technology
Introduction to Advanced Electrochemical Synthesis
The pharmaceutical industry continuously seeks robust and environmentally benign methodologies for constructing heterocyclic scaffolds essential for drug discovery and development. Patent CN112981436B introduces a groundbreaking electrochemical oxidation method for synthesizing 2-amino-1,3,4-thiadiazole derivatives, a privileged structure found in numerous bioactive molecules. This technology utilizes aryl isothiocyanates and simple aldehyde hydrazones as starting materials, employing 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) as a catalytic mediator within an undivided electrolytic cell. The significance of this innovation lies in its ability to bypass the severe safety and environmental limitations associated with classical thiadiazole formation, offering a pathway that is both mild and highly efficient.
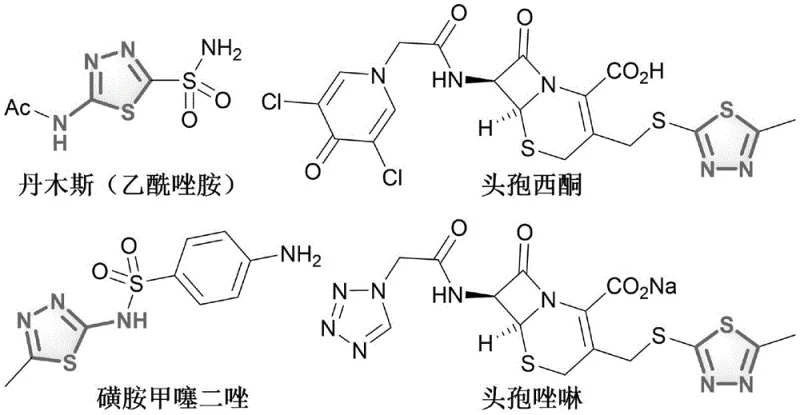
The 1,3,4-thiadiazole core is a critical pharmacophore present in widely used medications such as acetazolamide, cefazedone, sulfamethylthiadiazole, and cefazolin. As depicted in the structural examples below, the versatility of this heterocycle allows for diverse substitution patterns that modulate biological activity. By providing a cleaner synthetic route to these derivatives, this patent addresses the growing demand for reliable pharmaceutical intermediate supplier capabilities that align with green chemistry principles while maintaining high purity standards required for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3,4-thiadiazole derivatives has relied heavily on hazardous reagents that pose significant challenges for modern manufacturing facilities. The first conventional method involves the cyclization of hydrazides using phosphorus pentasulfide (P2S5). While effective in the laboratory, P2S5 is unstable in the presence of moisture, hydrolyzing rapidly to produce phosphoric acid and hydrogen sulfide gas. The release of hydrogen sulfide, known for its rotten egg odor and extreme toxicity, creates severe occupational health risks and necessitates expensive scrubbing systems, making it unsuitable for large-scale industrial production.
Alternative traditional routes utilize carbon disulfide (CS2) reacting with amino hydrazones or thiohydrazides. Carbon disulfide is extremely flammable and possesses strong irritant properties, presenting substantial fire hazards and environmental risks. Furthermore, methods employing substituted thiosemicarbazides often require dehydration using concentrated sulfuric acid or phosphoric acid, leading to low reaction yields and difficult waste disposal issues. These legacy processes result in high manufacturing costs due to the need for specialized containment, extensive safety protocols, and complex downstream purification to remove toxic byproducts and residual heavy metals.
The Novel Approach
In stark contrast, the electrochemical oxidation method described in the patent offers a transformative solution by replacing chemical oxidants with electrons. This novel approach utilizes a constant current electrolysis setup where the oxidation power is precisely controlled by the applied current, eliminating the need for stoichiometric amounts of dangerous oxidizing agents. The reaction proceeds in a mixed solvent system of acetonitrile and water (9:1), which not only serves as a proton source but also enhances the solubility of various substrates. The use of DDQ as a redox mediator ensures that the oxidation potential is tuned specifically for the transformation, preventing over-oxidation of sensitive functional groups.
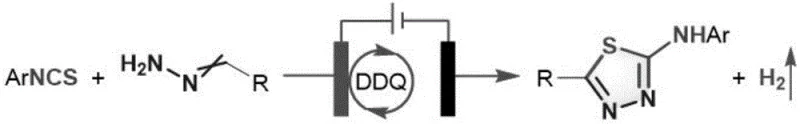
This methodology demonstrates exceptional substrate tolerance, accommodating electron-rich and electron-deficient aryl isothiocyanates as well as various aldehyde hydrazones, including those derived from heterocyclic and aliphatic aldehydes. The reaction conditions are remarkably mild, typically operating at 35°C under an argon atmosphere, which preserves the integrity of thermally labile compounds. By avoiding transition metal catalysts, the process simplifies the removal of metal impurities, a critical factor for regulatory compliance in drug substance manufacturing. This shift from chemical to electrochemical driving force represents a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into DDQ-Mediated Electrochemical Cyclization
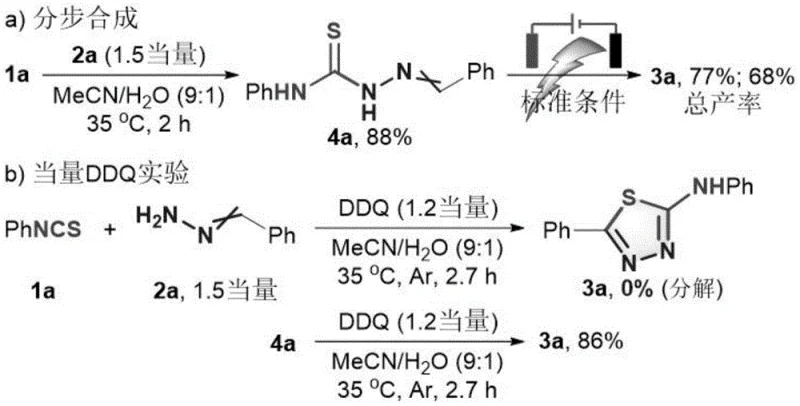
Understanding the mechanistic pathway is crucial for optimizing the synthesis of high-purity OLED material or pharmaceutical precursors. The reaction initiates with a nucleophilic addition between the aryl isothiocyanate and the aldehyde hydrazone to form a thiosemicarbazide intermediate. Spectroscopic analysis confirms that this addition step is rapid, consuming the majority of the isothiocyanate within the first 30 minutes. However, cyclic voltammetry studies reveal that this intermediate does not undergo direct oxidation at the anode under the standard reaction conditions, suggesting that a mediated electron transfer process is operative.
The DDQ catalyst plays a pivotal role as an electron shuttle. It interacts with the thiosemicarbazide intermediate, facilitating its oxidation at a lower potential than direct anodic oxidation would require. This interaction prevents the competitive oxidative decomposition of the starting materials or the product, which was observed when attempting the reaction with stoichiometric DDQ without electricity. The electrochemical regeneration of the active oxidant species at the anode allows the catalytic cycle to continue with high Faradaic efficiency, reported to be around 62% in optimized conditions. This mechanism ensures that the electrons flowing through the cell are effectively utilized to drive the cyclization rather than side reactions.
Impurity control is inherently built into this mechanism. Since the oxidation is mediated and potential-controlled, the formation of over-oxidized byproducts is minimized compared to harsh chemical oxidants like permanganate or chromate. The absence of heavy metal catalysts further reduces the risk of metal-complexed impurities that are difficult to purge. The reaction produces hydrogen gas at the cathode as the sole byproduct of the reduction half-reaction, which simply vents from the system, leaving the organic phase free from inorganic salt contamination. This clean reaction profile is essential for achieving the stringent purity specifications required for clinical-grade intermediates.
How to Synthesize 2-Amino-1,3,4-Thiadiazole Efficiently
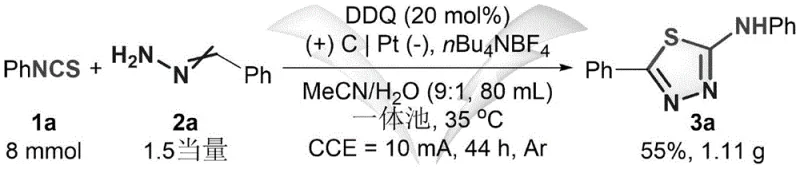
The practical implementation of this electrochemical protocol requires careful attention to electrode configuration and reaction parameters to ensure reproducibility and yield. The patent outlines a standardized procedure using an undivided cell equipped with a graphite rod anode and a platinum sheet cathode. The electrolyte system typically employs tetrabutylammonium tetrafluoroborate (nBu4NBF4) dissolved in a degassed mixture of acetonitrile and water. Maintaining a constant current of 10 mA at a controlled temperature of 35°C is critical for maximizing the Faradaic yield. Detailed operational guidelines regarding electrode polishing, solvent degassing, and workup procedures are essential for transferring this technology from the laboratory to pilot scales.
- Prepare the aldehyde hydrazone substrate by reacting the corresponding aldehyde with hydrazine hydrate in methanol at room temperature.
- Set up an undivided electrolytic cell with a graphite rod anode and platinum sheet cathode, adding the aryl isothiocyanate, hydrazone, DDQ catalyst, and nBu4NBF4 electrolyte in MeCN/H2O.
- Apply constant current electrolysis (10 mA) at 35°C under argon protection until the reaction reaches completion, then isolate the product via extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical synthesis route offers compelling strategic advantages beyond mere technical novelty. The elimination of hazardous reagents like phosphorus pentasulfide and carbon disulfide drastically simplifies the logistics of raw material sourcing and storage. Facilities no longer need to maintain specialized infrastructure for handling toxic gases or flammable liquids, which directly translates to reduced insurance premiums and lower capital expenditure on safety equipment. This simplification of the supply chain enhances overall reliability and reduces the risk of production stoppages due to regulatory inspections or safety incidents.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and stoichiometric chemical oxidants significantly lowers the bill of materials. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of silica gel and solvents during purification. The mild reaction conditions also imply lower energy costs for heating and cooling compared to processes requiring reflux or cryogenic temperatures. By streamlining the synthesis into a single electrochemical step with high atom economy, manufacturers can achieve substantial cost savings in the production of complex thiadiazole derivatives.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as aryl isothiocyanates and aldehydes ensures a robust supply base. Unlike specialized reagents that may have limited suppliers or long lead times, the inputs for this process are commodity chemicals with established global supply chains. The ability to run the reaction in an air atmosphere with only a slight decrease in yield, as noted in the patent data, further adds to the operational flexibility, allowing for production even if inert gas supplies are temporarily constrained.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this method to gram levels, indicating its potential for commercial scale-up of complex pharmaceutical intermediates. The use of an undivided cell simplifies reactor design, making it easier to adapt to flow chemistry or larger batch tanks. Environmentally, the process generates minimal waste; the primary byproduct is hydrogen gas, and the aqueous-organic solvent system is easier to treat than the acidic sludge generated by traditional acid-catalyzed dehydrations. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of thiadiazole derivatives. These answers are derived directly from the experimental data and findings presented in the patent literature, providing clarity on reaction scope, optimization, and practical application. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of this electrochemical method over traditional thiadiazole synthesis?
A: Unlike traditional methods requiring toxic phosphorus pentasulfide or flammable carbon disulfide, this electrochemical approach operates under mild conditions (35°C) without transition metal catalysts or stoichiometric chemical oxidants, significantly reducing environmental hazards and purification complexity.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent demonstrates successful scale-up to gram-level quantities (e.g., 8 mmol scale) with maintained efficiency. The use of simple electrode materials like graphite and platinum, along with a mixed aqueous-organic solvent system, supports potential adaptation for larger commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of DDQ in this electrochemical reaction?
A: DDQ (2,3-dichloro-5,6-dicyano-p-benzoquinone) acts as a redox mediator catalyst. It facilitates the oxidative cyclization of the thiosemicarbazide intermediate at the anode, improving Faradaic efficiency and preventing the direct oxidative decomposition of sensitive substrates that might occur with direct electrolysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-1,3,4-Thiadiazole Supplier
As the pharmaceutical landscape evolves towards greener and more efficient synthesis technologies, partnering with an experienced CDMO becomes increasingly vital. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced electrochemical methodologies to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for global regulatory submissions.
We invite you to explore how our electrochemical capabilities can optimize your supply chain for thiadiazole-based APIs. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how this innovative patent technology can enhance your product's competitiveness and sustainability profile in the global market.
