Revolutionizing Biphenyl Production: High-Selectivity Hydrogenation of Dibenzofuran for Global Markets
The global demand for high-purity biphenyl (BP) continues to surge across pharmaceutical, agrochemical, and polymer sectors, driving an urgent need for more efficient and sustainable synthesis routes. Traditional methods often struggle with energy intensity and byproduct management, creating bottlenecks for reliable supply chains. Addressing these challenges, patent CN107445786B introduces a groundbreaking method for preparing biphenyl through the catalytic hydrogenation of dibenzofuran (DBF). This technology leverages a specialized fluidized bed reactor system coupled with novel molybdenum-based catalysts, specifically MoO3, Mo2C, MoP, or Mo2N. By converting low-value dibenzofuran, a byproduct of coal tar processing, into high-value biphenyl, this process not only optimizes resource utilization but also achieves exceptional conversion rates exceeding 96% with biphenyl selectivity reaching up to 99.3%. For procurement and R&D leaders, this represents a paradigm shift towards cost-effective and environmentally compliant manufacturing of critical fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of biphenyl has relied heavily on the catalytic cracking of pure benzene at extremely high temperatures, typically ranging between 500°C and 680°C. This thermal cracking process is inherently energy-intensive, requiring substantial fuel input to maintain the necessary reaction conditions, which directly inflates operational expenditures. Moreover, the harsh reaction environment promotes the formation of undesirable byproducts, most notably terphenyl, which complicates the downstream purification process. The presence of these impurities necessitates complex distillation sequences to achieve the stringent purity standards required for pharmaceutical and electronic applications, thereby increasing both capital investment in separation equipment and ongoing maintenance costs. Additionally, the reliance on pure benzene as a feedstock exposes manufacturers to volatile petrochemical market prices, introducing significant financial risk and supply chain instability for long-term production planning.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes dibenzofuran, an abundant and underutilized component found in coal tar washing oil, as the primary feedstock. This method operates under significantly milder conditions, with reaction temperatures optimized between 320°C and 400°C and pressures as low as 0.1 MPa, drastically reducing energy consumption compared to benzene cracking. The core innovation lies in the use of molybdenum-based catalysts within a fluidized bed reactor, which facilitates excellent gas-solid contact and heat transfer. This setup allows for precise control over the hydrogenation pathway, favoring the cleavage of the C-O bond in dibenzofuran to form biphenyl while minimizing the hydrogenation of the aromatic rings to cyclohexylbenzene. The result is a streamlined process that delivers high-purity biphenyl with minimal byproduct formation, effectively bypassing the economic and technical hurdles associated with traditional synthesis routes.
Mechanistic Insights into MoO3-Catalyzed Hydrogenation
The exceptional performance of this synthesis route is fundamentally rooted in the unique electronic and structural properties of the molybdenum oxide (MoO3) catalyst. Unlike traditional noble metal or sulfided catalysts, MoO3 exhibits a specific affinity for the oxygen-containing heterocyclic structure of dibenzofuran. The mechanism involves the adsorption of the dibenzofuran molecule onto the active sites of the catalyst surface, where the interaction weakens the C-O bonds within the furan ring. Under the influence of activated hydrogen species generated on the catalyst surface, the C-O bond undergoes hydrogenolysis, effectively removing the oxygen atom as water and leaving the biphenyl skeleton intact. This selective bond cleavage is critical; it prevents the saturation of the phenyl rings, which would otherwise lead to the formation of cyclohexylbenzene (CHB), a difficult-to-separate impurity that degrades product quality.
Furthermore, the fluidized bed configuration plays a pivotal role in maintaining catalyst activity and selectivity over extended periods. As the reaction proceeds, coke deposition can occur on the catalyst surface, potentially blocking active sites. However, the continuous circulation of the catalyst between the reactor and the regenerator unit allows for the periodic removal of these carbonaceous deposits using air oxidation. This regeneration cycle restores the catalyst's surface area and active site density, ensuring consistent performance without the need for frequent shutdowns or catalyst replacement. The synergy between the MoO3 active phase and the dynamic reactor environment creates a robust system capable of sustaining high conversion rates and selectivity, providing a reliable foundation for large-scale industrial application.
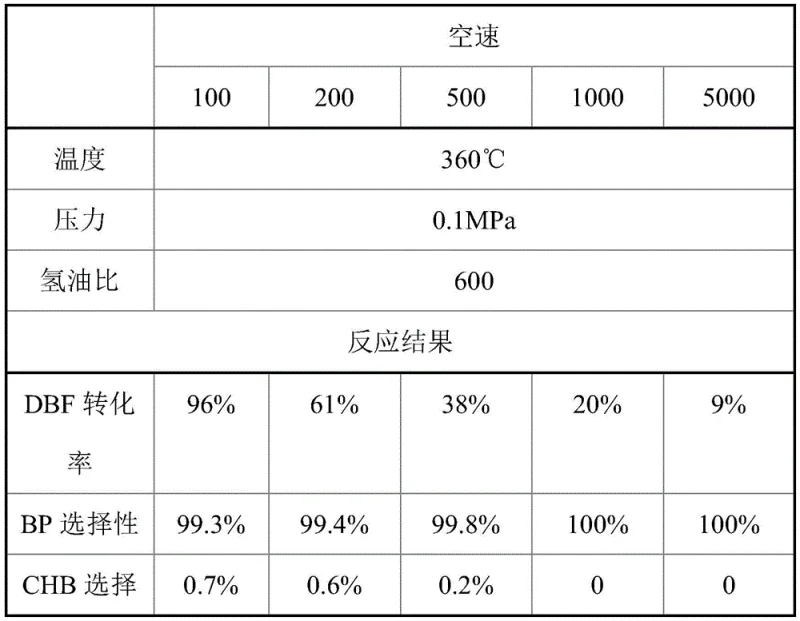
Experimental data underscores the superiority of the MoO3 catalyst compared to other molybdenum variants such as Mo2C, Mo2N, and MoP. As illustrated in the comparative analysis, MoO3 achieves a dibenzofuran conversion rate of 96% with a biphenyl selectivity of 99.3% under optimized conditions of 360°C and 0.1 MPa. In comparison, other catalysts show lower activity and selectivity, with MoP exhibiting a biphenyl selectivity of only 90.8% and higher formation of CHB byproducts. This data confirms that the specific oxidation state and surface chemistry of MoO3 are optimal for directing the reaction pathway towards the desired biphenyl product, minimizing waste and maximizing yield efficiency.
How to Synthesize Biphenyl Efficiently
Implementing this synthesis route requires careful attention to reactor dynamics and parameter optimization to fully realize the benefits of the MoO3 catalyst system. The process begins with the preparation of the catalyst, followed by its introduction into the fluidized bed riser where it mixes with preheated hydrogen and the dibenzofuran feed. Maintaining the correct space velocity and hydrogen-to-oil ratio is crucial for ensuring sufficient residence time for the reaction while preventing excessive hydrogenation. The detailed standardized synthesis steps, including specific temperature ramps, pressure controls, and regeneration cycles, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the MoO3 catalyst and load it into the fluidized bed reactor system, ensuring the riser and settler units are aligned for continuous circulation.
- Heat the dibenzofuran feed liquid to 200-300°C and introduce it into the catalyst bed driven by low-pressure hydrogen with a molar ratio of 5-100.
- Maintain reaction conditions at 320-400°C and 0.1-3.0 MPa, then separate products via cyclone separator and purify biphenyl through rectification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dibenzofuran hydrogenation technology offers transformative advantages in terms of cost structure and supply security. By shifting the feedstock base from high-value pure benzene to low-cost coal tar derivatives, manufacturers can significantly decouple their production costs from fluctuating crude oil markets. This strategic pivot not only stabilizes the cost of goods sold but also opens up new avenues for margin expansion in a competitive global market. The simplified downstream processing, resulting from high selectivity and low byproduct formation, further contributes to operational efficiency by reducing utility consumption and waste disposal fees associated with complex purification trains.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the use of regenerable molybdenum-based systems drastically reduce catalyst procurement and replacement costs. Additionally, the mild reaction conditions lower energy requirements for heating and compression, leading to substantial savings in utility bills. The high selectivity minimizes the loss of raw materials to byproducts, ensuring that a greater proportion of the feedstock is converted into saleable high-purity biphenyl, thereby optimizing the overall material balance and reducing the effective cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: Sourcing dibenzofuran from coal tar processing provides a stable and abundant supply of raw materials that is less susceptible to the geopolitical and logistical disruptions often affecting benzene supply chains. The ability to regenerate the catalyst on-site ensures continuous operation without long lead times for catalyst delivery, mitigating the risk of production stoppages. This reliability is critical for meeting the just-in-time delivery requirements of downstream pharmaceutical and agrochemical customers who depend on uninterrupted access to high-quality intermediates.
- Scalability and Environmental Compliance: The fluidized bed reactor design is inherently scalable, allowing for seamless transition from pilot plant to full commercial production capacities ranging from hundreds of kilograms to hundreds of metric tons annually. The process generates minimal hazardous waste due to the high atom economy and the absence of toxic solvents or heavy metal contaminants in the final product. This aligns with increasingly stringent environmental regulations, reducing the compliance burden and enhancing the corporate sustainability profile of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biphenyl synthesis technology. These insights are derived directly from the patent specifications and experimental data to provide clarity on process feasibility and performance metrics. Understanding these details is essential for stakeholders evaluating the integration of this method into existing production portfolios.
Q: What represents the primary advantage of using Dibenzofuran over Benzene for Biphenyl synthesis?
A: Utilizing dibenzofuran, a low-value component derived from coal tar washing oil, significantly reduces raw material costs compared to pure benzene. Furthermore, this pathway avoids the formation of terphenyl byproducts common in benzene cracking, simplifying downstream purification and enhancing overall yield.
Q: How does the MoO3 catalyst performance compare to traditional CoMo catalysts?
A: The proprietary MoO3 catalyst demonstrates superior activity and selectivity, achieving dibenzofuran conversion rates up to 96% and biphenyl selectivity reaching 99.3%. In contrast, traditional supported CoMo catalysts typically exhibit much lower selectivity around 60%, leading to higher waste generation and separation costs.
Q: Is the catalyst regeneration process feasible for continuous industrial operation?
A: Yes, the process utilizes a fluidized bed design where the catalyst circulates between the reactor and a regenerator. Air is used to regenerate the catalyst in the regenerator unit, allowing for continuous reuse without significant loss of activity, which ensures long-term operational stability and reduced catalyst consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the fine chemicals sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the MoO3-catalyzed hydrogenation of dibenzofuran can be successfully translated into robust manufacturing operations. We are committed to delivering high-purity biphenyl that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch conforms to the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this route offers for your specific application. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to serve as your trusted partner in securing a sustainable and cost-efficient supply of high-value chemical intermediates.
