Scaling Novel Imidazopyridine Derivatives for Next-Generation Antihypertensive Therapies
Scaling Novel Imidazopyridine Derivatives for Next-Generation Antihypertensive Therapies
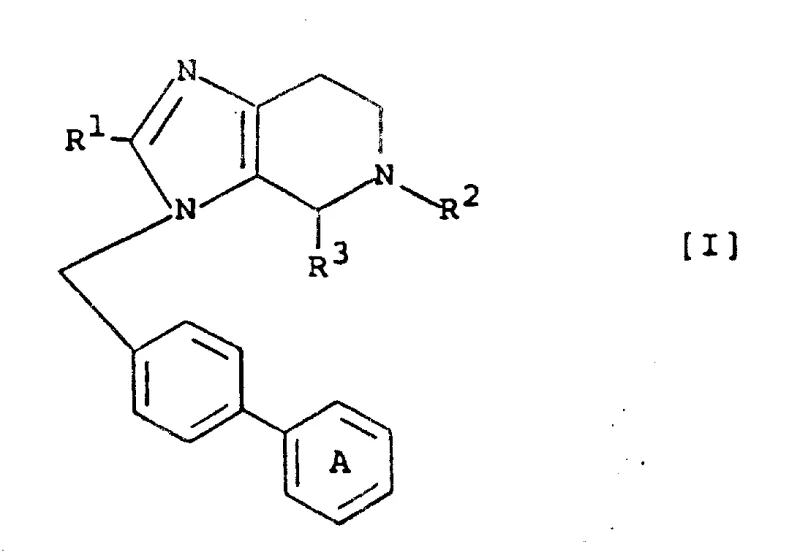
The pharmaceutical landscape for cardiovascular disease management is continuously evolving, driven by the need for more potent and selective angiotensin II receptor antagonists. Patent CN1070912A introduces a significant advancement in this field through the disclosure of novel imidazopyridine derivatives characterized by a fused bicyclic core structure. Unlike traditional monocyclic imidazole-based inhibitors, these compounds incorporate a pyridine ring fused to the imidazole nucleus, creating a rigid scaffold that enhances binding affinity and metabolic stability. The patent details a robust synthetic methodology that allows for extensive structural diversification at multiple positions, enabling the fine-tuning of pharmacological properties. This technical breakthrough represents a critical opportunity for generic drug manufacturers and innovators seeking to develop next-generation antihypertensive agents with superior efficacy profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of angiotensin II receptor blockers has relied heavily on monocyclic imidazole scaffolds, such as those found in early generation drugs like losartan. While effective, these traditional structures often face challenges related to metabolic clearance and off-target interactions due to their conformational flexibility. The synthesis of these older analogs frequently requires harsh conditions for introducing the critical biphenyl-tetrazole pharmacophore, often leading to lower yields and difficult purification processes. Furthermore, the limited structural rigidity of monocyclic systems can result in suboptimal receptor occupancy, necessitating higher dosages to achieve therapeutic blood pressure control. These limitations drive up the cost of goods and complicate the regulatory pathway due to the formation of complex impurity profiles that must be strictly controlled.
The Novel Approach
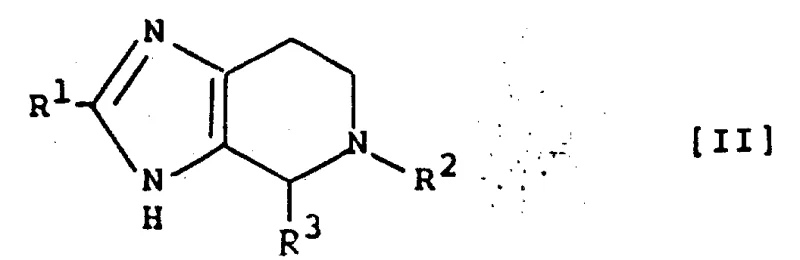
The methodology outlined in the patent data presents a transformative approach by utilizing a condensed imidazopyridine framework. This fused ring system inherently restricts molecular rotation, locking the pharmacophore into a bioactive conformation that maximizes interaction with the AT1 receptor. The synthetic strategy employs a modular design where the core heterocycle is constructed first, followed by late-stage functionalization. This allows for the introduction of diverse substituents at the N-position and the C-position of the pyridine ring without compromising the integrity of the sensitive tetrazole moiety. By shifting from a linear assembly to a convergent synthesis involving key intermediates like Formula II and Formula III, the process significantly reduces the number of isolation steps. This convergence not only improves overall yield but also simplifies the downstream processing, making it highly attractive for industrial scale-up.
Mechanistic Insights into Condensation and N-Alkylation Strategies
The core construction of the imidazopyridine skeleton relies on a sophisticated condensation reaction between an amino-ethyl substituted imidazole precursor and a glyoxylic acid derivative. This cyclization step is pivotal, as it forms the new carbon-nitrogen bonds required to close the pyridine ring. The reaction typically proceeds under acidic conditions or via thermal activation in high-boiling solvents, facilitating the dehydration necessary for aromatization. Following the formation of the bicyclic core, the critical N-alkylation step attaches the pharmacologically essential biphenyl-tetrazole group. This transformation is achieved through nucleophilic substitution, where the deprotonated nitrogen of the imidazopyridine attacks an activated benzyl halide carrying the protected tetrazole ring.
Mechanistically, the use of strong bases such as sodium hydride or alkali metal alkoxides ensures complete deprotonation of the imidazopyridine nitrogen, generating a highly reactive nucleophile. The choice of solvent, typically polar aprotic media like dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), stabilizes the transition state and enhances the solubility of the ionic intermediates. Subsequent acylation reactions allow for the introduction of various carbonyl-containing side chains at the remaining nitrogen or carbon centers. These acylations often utilize activated acid derivatives such as acid chlorides or anhydrides, sometimes mediated by coupling agents like dicyclohexylcarbodiimide (DCC) or EDC/HOBt to prevent racemization and ensure high conversion. The ability to perform these modifications on the fully formed scaffold provides exceptional control over the final physicochemical properties of the active pharmaceutical ingredient.
Impurity control is managed through the strategic use of protecting groups, particularly for the tetrazole ring, which is susceptible to alkylation at multiple nitrogen atoms. The patent describes the use of trityl or silyl protecting groups to direct the alkylation specifically to the desired position, thereby minimizing the formation of regioisomers. Post-reaction deprotection is carried out under mild acidic or basic conditions that do not degrade the sensitive ester or carboxylic acid functionalities elsewhere in the molecule. Final purification is routinely achieved using silica gel column chromatography or recrystallization from appropriate solvent systems, ensuring that the final product meets stringent purity specifications required for clinical applications. This rigorous control over the reaction pathway ensures a clean impurity profile, which is a critical factor for regulatory approval.
How to Synthesize Imidazopyridine Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to ensure reproducibility and safety. The process begins with the preparation of the amino-imidazole starting material, followed by cyclization with glyoxylic acid esters to form the imidazopyridine core. Subsequent steps involve the careful alkylation with biphenyl tetrazole bromides and final acylation or hydrolysis to yield the target carboxylic acid or ester derivatives. Each step must be monitored closely to prevent over-reaction or decomposition of the thermally sensitive tetrazole ring. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Preparation of the core imidazopyridine skeleton through the condensation of substituted imidazole precursors with glyoxylic acid esters under acidic or basic conditions.
- N-Alkylation of the imidazopyridine nitrogen using activated biphenyl tetrazole derivatives, typically employing sodium hydride or potassium carbonate in polar aprotic solvents.
- Final functionalization via acylation or hydrolysis to introduce diverse side chains, followed by purification using silica gel chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route described in this patent offers substantial advantages over legacy manufacturing processes. The reliance on commodity chemicals and standard reagents means that raw material sourcing is stable and less susceptible to geopolitical disruptions. The elimination of transition metal catalysts in the key bond-forming steps removes the need for expensive and time-consuming heavy metal scavenging processes, which are often a bottleneck in API production. This simplification of the downstream processing directly translates to reduced manufacturing costs and shorter cycle times. Furthermore, the modular nature of the synthesis allows for the production of a library of analogs from a common advanced intermediate, providing flexibility to respond to market demands without retooling the entire production line.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts such as palladium or platinum, which are subject to volatile pricing and supply constraints. Instead, it utilizes cost-effective bases like sodium hydride and potassium carbonate, along with widely available solvents. The high atom economy of the condensation and alkylation steps minimizes waste generation, reducing disposal costs. Additionally, the ability to isolate stable intermediates allows for batch processing optimization, further driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The synthetic pathway is robust and tolerant to minor variations in reaction conditions, ensuring consistent quality across different production batches. The starting materials, including substituted imidazoles and biphenyl derivatives, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The process does not require cryogenic conditions or specialized high-pressure equipment, making it compatible with existing multipurpose reactor infrastructure in most chemical manufacturing facilities. This compatibility ensures rapid technology transfer and reliable supply continuity.
- Scalability and Environmental Compliance: The reactions are conducted in standard organic solvents that can be efficiently recovered and recycled, aligning with green chemistry principles and reducing the environmental footprint. The absence of toxic heavy metals simplifies wastewater treatment and regulatory compliance regarding residual metal limits in the final drug product. The scalability of the nucleophilic substitution and acylation steps has been demonstrated in similar chemical contexts, suggesting a smooth path from pilot plant to commercial scale production without significant engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these imidazopyridine derivatives. The answers are derived from the specific experimental data and claims found within the patent documentation, providing clarity on the feasibility and scope of the technology.
Q: What is the primary therapeutic application of these imidazopyridine derivatives?
A: These compounds function as potent angiotensin II receptor antagonists, specifically designed for the prevention and treatment of hypertension by inhibiting vasoconstriction and aldosterone secretion.
Q: How does the fused ring system improve upon traditional imidazole drugs?
A: The condensed imidazopyridine ring system offers enhanced structural rigidity and potentially improved metabolic stability compared to monocyclic imidazole analogs, allowing for optimized pharmacokinetic profiles.
Q: Are the synthetic routes scalable for commercial production?
A: Yes, the described methodologies utilize standard organic transformations such as nucleophilic substitution and amide coupling, which are well-established for scale-up from laboratory to multi-ton manufacturing without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazopyridine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing for complex pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of imidazopyridine derivatives meets the highest industry standards. Our commitment to quality and consistency makes us the preferred partner for global pharmaceutical companies seeking reliable supply chains for critical cardiovascular intermediates.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your regulatory filings and accelerate your time to market. Let us help you optimize your supply chain with high-quality, cost-effective solutions.
