Scalable Synthesis of Fused-Ring Imidazopyridine Derivatives for Next-Generation Antihypertensive Therapies
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the need for more potent and stable angiotensin II receptor antagonists. Patent CN1039323C introduces a significant advancement in this field through the preparation of novel imidazopyridine derivatives. Unlike traditional antihypertensive agents that rely on single-ring imidazole nuclei, these fused-ring systems offer superior pharmacological profiles. The core innovation lies in the structural rigidity and electronic properties conferred by the imidazo[4,5-c]pyridine scaffold, which enhances receptor binding affinity. This technical breakthrough addresses the limitations of earlier generations of drugs, providing a robust foundation for developing next-generation hypertension treatments. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthesis is critical for ensuring consistent quality and supply continuity.
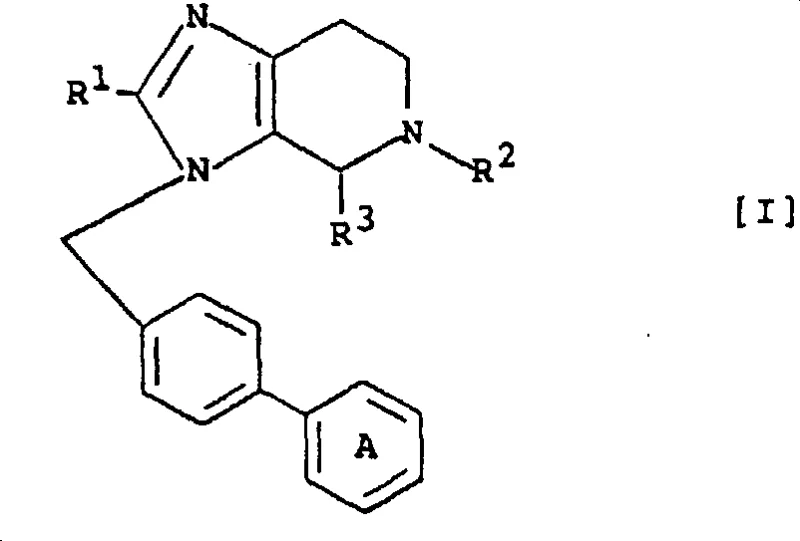
The transition from conventional single-ring imidazoles to these complex fused-ring derivatives represents a paradigm shift in medicinal chemistry design. Traditional methods often struggled with metabolic instability and lower bioavailability inherent to simpler heterocyclic structures. In contrast, the novel approach detailed in the patent utilizes a strategic cyclization strategy that locks the molecular conformation. This structural constraint minimizes non-productive metabolic pathways, thereby extending the half-life of the active compound in vivo. Furthermore, the versatility of the substitution pattern allows for fine-tuning of physicochemical properties such as solubility and lipophilicity. By moving away from the limitations of the past, manufacturers can achieve cost reduction in antihypertensive drug manufacturing through higher efficacy doses and reduced frequency of administration, ultimately benefiting the entire healthcare value chain.
Mechanistic Insights into N-Alkylation and Cyclization Strategies
The synthesis mechanism centers on the precise functionalization of the imidazole nitrogen followed by ring closure. The process typically initiates with the deprotonation of the imidazole precursor using strong bases such as sodium hydride or alkali metal alkoxides in polar aprotic solvents like dimethylformamide. This generates a highly reactive nucleophile that attacks electrophilic centers, such as biphenyl methyl halides, to form the crucial N-C bond. Temperature control is paramount during this exothermic phase, often maintained between -30°C and 50°C to prevent side reactions. Subsequent steps involve the introduction of various acyl groups or the formation of the pyridine ring through condensation with aldehydes or similar reagents. The ability to manipulate substituents at the R1, R2, and R3 positions allows for the generation of a diverse library of analogs, facilitating structure-activity relationship studies essential for drug optimization.
Impurity control is a critical aspect of this synthetic route, particularly regarding the formation of positional isomers. During the alkylation step, reaction at different nitrogen atoms can lead to regioisomeric mixtures that complicate downstream purification. The patent outlines rigorous purification protocols involving silica gel column chromatography and recrystallization to isolate the desired isomer with high purity. Additionally, the hydrolysis of ester groups to carboxylic acids must be carefully managed to avoid racemization at chiral centers. By employing mild hydrolytic conditions and monitoring reaction progress closely, manufacturers can ensure the stereochemical integrity of the final product. This attention to detail in impurity profiling is vital for meeting stringent regulatory standards required for high-purity imidazopyridine derivatives intended for clinical use.
How to Synthesize Imidazopyridine Derivatives Efficiently
The efficient production of these complex molecules requires a deep understanding of reaction kinetics and thermodynamic stability. The patented process offers a streamlined pathway that avoids the use of expensive transition metal catalysts, relying instead on widely available organic and inorganic reagents. Detailed operational parameters, including solvent ratios, stirring speeds, and addition rates, are optimized to maximize yield while minimizing waste generation. For research and development teams looking to replicate or scale this chemistry, adherence to the specified molar equivalents and temperature profiles is essential. The following guide outlines the standardized synthesis steps derived from the patent data, serving as a foundational reference for process development.
- Perform N-alkylation of the imidazole precursor using alkali metal hydrides or alkoxides in polar aprotic solvents like DMF at controlled temperatures ranging from -30°C to 50°C.
- Execute cyclization or acylation reactions using appropriate acylating agents or aldehydes, often requiring acid or base catalysis depending on the specific substituent requirements.
- Purify the final crude product through silica gel column chromatography or recrystallization from suitable solvent systems such as chloroform/methanol or ethanol/ether to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described offers significant logistical and economic benefits. The reliance on commodity chemicals such as sodium hydride, dimethylformamide, and common alkyl halides ensures a stable supply chain不受 geopolitical disruptions affecting rare earth metals or specialized catalysts. This accessibility translates directly into enhanced supply chain reliability, as raw material sourcing becomes predictable and less volatile. Furthermore, the elimination of complex catalytic systems simplifies the manufacturing infrastructure, reducing the capital expenditure required for specialized reactor setups. These factors collectively contribute to a more resilient production model capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The process eliminates the need for costly transition metal catalysts and ligands, which are often expensive and difficult to remove to trace levels. By utilizing standard organic synthesis techniques involving nucleophilic substitution and acylation, the overall cost of goods sold is significantly optimized. The avoidance of heavy metal removal steps also reduces the burden on wastewater treatment facilities, leading to substantial cost savings in environmental compliance and waste management operations.
- Enhanced Supply Chain Reliability: The starting materials, including substituted imidazoles and biphenyl derivatives, are commercially available from multiple global vendors. This multi-sourcing capability mitigates the risk of single-supplier dependency, ensuring continuous production even if one vendor faces shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, further enhances operational flexibility, allowing for consistent output regardless of minor variations in raw material specifications.
- Scalability and Environmental Compliance: The synthetic pathway is designed for scalability, utilizing solvents that can be easily recovered and recycled through distillation. The absence of toxic heavy metals simplifies the disposal of chemical waste, aligning with green chemistry principles. This environmental compatibility not only reduces regulatory hurdles but also improves the corporate sustainability profile, making the manufacturing process more attractive to environmentally conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these imidazopyridine derivatives. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions regarding technology adoption and partnership opportunities.
Q: How does the fused-ring structure improve upon traditional single-ring imidazole antagonists?
A: The fused imidazopyridine ring system provides enhanced metabolic stability and potentially higher binding affinity to angiotensin II receptors compared to traditional single-ring imidazole compounds, leading to improved therapeutic efficacy.
Q: What are the critical purification challenges associated with these derivatives?
A: Separation of positional isomers formed during the N-alkylation step is a key challenge, typically addressed through precise silica gel column chromatography and selective recrystallization techniques to isolate the desired regioisomer.
Q: Is the synthesis process scalable for commercial production?
A: Yes, the described methodology utilizes standard industrial solvents and reagents like sodium hydride and dimethylformamide, allowing for straightforward scale-up from laboratory to commercial manufacturing without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazopyridine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to navigate the complexities of fused-ring heterocycle synthesis, ensuring that every batch meets stringent purity specifications. We understand that consistency is key in pharmaceutical manufacturing, which is why our rigorous QC labs employ advanced analytical techniques to verify identity and assay. By partnering with us, clients gain access to a supply chain that prioritizes quality, safety, and reliability above all else.
We invite potential partners to engage with our technical procurement team to discuss your specific requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new projects, we are equipped to provide comprehensive support. Our commitment to transparency and technical excellence ensures that we can meet the demanding needs of the global pharmaceutical market, delivering high-quality intermediates that drive your success.
