Advanced Non-Cyanide Synthesis of Febuxostat for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways for the production of critical gout therapeutics like Febuxostat (TMX-67). Patent CN102936230A introduces a groundbreaking preparation method that fundamentally restructures the synthetic logic of this xanthine oxidase inhibitor. Unlike conventional methodologies that rely on hazardous cyanide sources and expensive noble metal catalysts, this novel approach leverages a strategic sequence starting from 2-hydroxy-5-cyanobenzaldehyde. The process ingeniously bypasses the traditional nitro-reduction and diazotization steps, replacing them with a milder thioamide formation and an oxime-mediated nitrile introduction. This shift not only mitigates significant safety hazards associated with high-pressure hydrogenation and toxic heavy metals but also streamlines the purification workflow, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
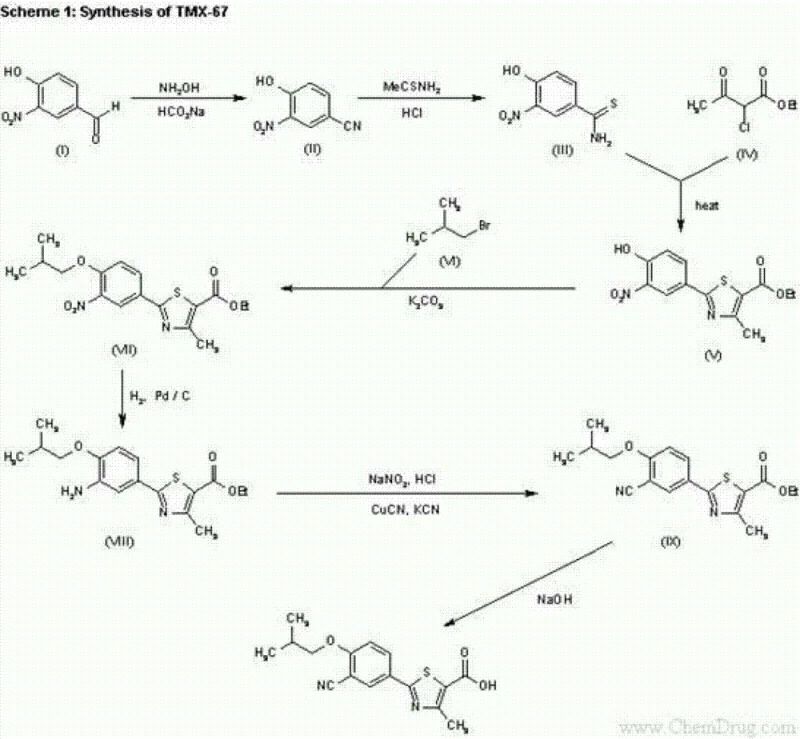
Historically, the industrial synthesis of Febuxostat has been plagued by severe operational bottlenecks and environmental liabilities. Traditional routes, often depicted in legacy literature, typically initiate from 3-nitro-4-hydroxybenzaldehyde, necessitating a multi-step transformation that includes a palladium-on-carbon (Pd/C) catalyzed hydrogenation to reduce the nitro group to an amine. This step imposes rigorous equipment standards due to the risks of high-pressure hydrogen handling and the pyrophoric nature of spent catalyst filtration. Furthermore, the subsequent introduction of the cyano group traditionally relies on a Sandmeyer reaction involving sodium nitrite and cuprous cyanide or potassium cyanide. These reagents are acutely toxic, posing grave dangers to personnel and generating complex cyanide-containing wastewater that requires costly and specialized treatment protocols. Additionally, alternative routes utilizing hexamethylenetetramine (HMTA) in polyphosphoric acid (PPA) for formylation introduce massive amounts of phosphorus-containing waste and suffer from poor mixing efficiency due to the high viscosity of PPA, severely hampering heat transfer and reaction control in large reactors.
The Novel Approach
In stark contrast, the methodology disclosed in CN102936230A presents a streamlined, five-step sequence that elegantly circumvents these historical pitfalls. By selecting 2-hydroxy-5-cyanobenzaldehyde as the starting material, the synthesis inherently possesses the required nitrile functionality, thereby completely eliminating the need for dangerous cyanation reactions post-cyclization. The core innovation lies in the direct conversion of the aldehyde-thioamide intermediate into the thiazole ring, followed by a mild dehydration of an oxime intermediate to secure the final nitrile positioning if rearrangement were needed, though here the nitrile is preserved or manipulated safely. This route avoids the use of corrosive strong acids like polyphosphoric acid and eliminates the requirement for transition metal catalysts like palladium or copper. The result is a process characterized by significantly reduced equipment corrosion, lower waste treatment burdens, and a dramatic simplification of the operational workflow, making it ideally suited for cost reduction in API manufacturing where safety and environmental compliance are paramount.
Mechanistic Insights into Thioamide Formation and Thiazole Cyclization
The chemical elegance of this new process is anchored in the initial transformation of 2-hydroxy-5-cyanobenzaldehyde into 3-aldehyde-4-hydroxybenzothioamide. This step utilizes sodium hydrosulfide (NaSH) in the presence of magnesium chloride (MgCl2) within a DMF solvent system. The magnesium chloride acts as a crucial Lewis acid promoter, coordinating with the carbonyl oxygen and potentially the nitrile nitrogen to activate the substrate for nucleophilic attack by the hydrosulfide ion. This catalytic effect enhances the reaction rate and selectivity, ensuring high conversion at moderate temperatures ranging from 50°C to 80°C. Following this, the thioamide undergoes cyclization with ethyl 2-haloacetoacetate (where the halogen is preferably bromine or chlorine). This Hantzsch-type thiazole synthesis proceeds efficiently under reflux conditions in alcoholic solvents, constructing the critical 4-methyl-5-ethoxycarbonyl thiazole scaffold with high regioselectivity. The preservation of the aldehyde group during this cyclization is vital for subsequent functionalization, demonstrating the orthogonal reactivity managed by this specific reaction sequence.
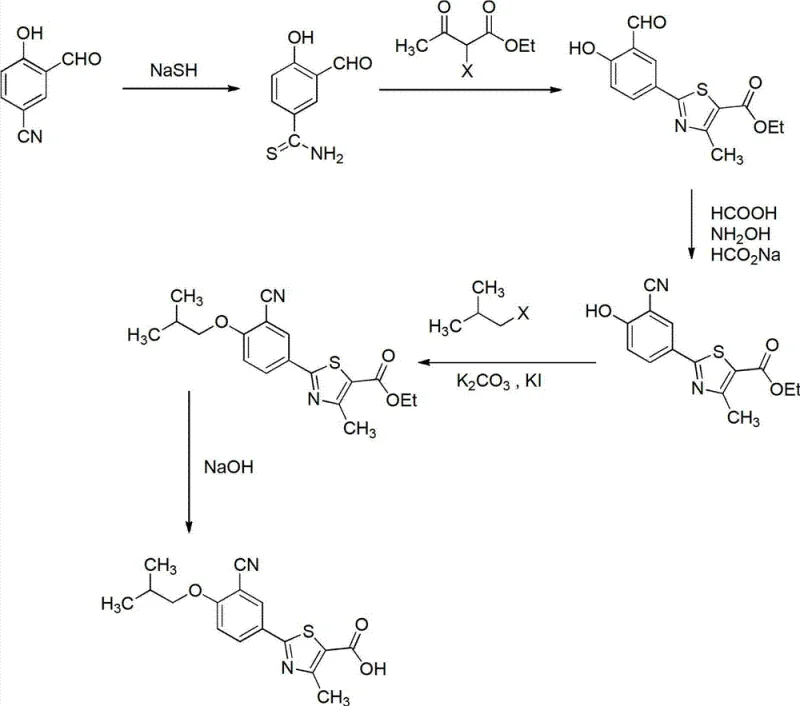
Furthermore, the strategy for impurity control is intrinsically built into the choice of reagents. By avoiding the Pd/C hydrogenation step, the process eliminates the risk of residual heavy metal contamination in the final active pharmaceutical ingredient (API), a critical quality attribute for regulatory approval. Similarly, the absence of cuprous cyanide removes the potential for copper impurities which can catalyze degradation pathways in the final drug product. The conversion of the aldehyde to the nitrile (in variations where the nitrile is introduced later) or the manipulation of the existing nitrile is handled under mild acidic conditions using hydroxylamine and formic acid. This dehydration protocol is far cleaner than traditional methods, producing water and volatile byproducts rather than inorganic salt sludges. The cumulative effect of these mechanistic choices is a product profile with exceptional purity, consistently reported above 99%, which minimizes the need for extensive recrystallization or chromatographic purification downstream.
How to Synthesize Febuxostat Efficiently
The implementation of this novel synthesis route requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The process begins with the careful addition of sodium hydrosulfide and magnesium chloride to a DMF solution of the starting aldehyde, maintaining the temperature between 58°C and 62°C for optimal thioamide formation. Subsequent cyclization with ethyl 2-bromoacetoacetate is conducted under reflux in ethanol, followed by a workup that leverages the solubility differences of the product to isolate the thiazole intermediate as a solid. The final stages involve etherification with isobutyl bromide using potassium carbonate and a catalytic amount of potassium iodide, followed by saponification of the ester group. For a detailed, step-by-step breakdown of the exact molar ratios, temperature profiles, and workup procedures described in the patent, please refer to the standardized guide below.
- React 2-hydroxy-5-cyanobenzaldehyde with sodium hydrosulfide (NaSH) and magnesium chloride in DMF to form 3-aldehyde-4-hydroxybenzothioamide.
- Perform cyclization with ethyl 2-haloacetoacetate to construct the 4-methyl-5-ethoxycarbonyl thiazole core.
- Convert the aldehyde group to a nitrile using hydroxylamine in formic acid solution, followed by etherification and hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The primary economic driver is the elimination of expensive and hazardous reagents. By removing the need for palladium catalysts, the process avoids the capital expenditure associated with hydrogenation reactors and the recurring cost of noble metal recovery or disposal. Furthermore, the substitution of toxic cyanides with safer alternatives drastically reduces the regulatory burden and insurance costs associated with handling Schedule 1 chemicals. This shift allows for production in facilities with standard chemical processing capabilities rather than requiring specialized high-containment or high-pressure suites, thereby expanding the pool of eligible contract manufacturing organizations (CMOs) and enhancing supply chain resilience.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, driven primarily by the simplification of the unit operations. The removal of the Pd/C hydrogenation step eliminates a major bottleneck, reducing batch cycle times and energy consumption associated with high-pressure operations. Additionally, the avoidance of polyphosphoric acid removes the need for specialized glass-lined or Hastelloy equipment resistant to extreme corrosion and viscosity, allowing for the use of standard stainless steel reactors. The simplified workup procedures, which rely on pH adjustments and filtration rather than complex extractions or chromatography, lead to significant savings in solvent usage and labor hours. These factors collectively contribute to a lower cost of goods sold (COGS) without compromising the quality of the high-purity Febuxostat produced.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the raw materials for this route are commoditized and readily available on the global market. 2-hydroxy-5-cyanobenzaldehyde and ethyl 2-bromoacetoacetate are produced at scale by multiple vendors, reducing the risk of single-source dependency that often plagues specialized nitro-compounds or exotic catalysts. The robustness of the chemistry, characterized by high yields and reproducibility across a broad temperature range, ensures consistent output volumes. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, preventing stockouts that could disrupt the production of finished gout medications. The process stability also means fewer failed batches, further securing the supply continuity for long-term contracts.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this route is designed with industrial practicality in mind. The reactions are exothermic but manageable, and the absence of gas-evolving steps (like diazotization) or high-pressure requirements simplifies reactor design and safety interlocks. Environmentally, the process generates significantly less hazardous waste. The elimination of cyanide-containing effluents and heavy metal residues simplifies wastewater treatment, aligning with increasingly stringent global environmental regulations. This 'green' profile not only reduces disposal costs but also enhances the corporate social responsibility (CSR) standing of the manufacturer, making the supply chain more attractive to sustainability-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Febuxostat synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on the practical implications for R&D and production teams.
Q: How does this new process improve safety compared to traditional Febuxostat synthesis?
A: The novel process eliminates the use of highly toxic cuprous cyanide and potassium cyanide required in traditional Sandmeyer reactions, and avoids high-pressure hydrogenation with palladium on carbon, significantly reducing operational risks and waste treatment costs.
Q: What are the key purity advantages of the NaSH/MgCl2 catalytic system?
A: The use of magnesium chloride as a Lewis acid promoter in the thioamide formation step enhances reaction selectivity, minimizing side products and facilitating easier purification to achieve purity levels exceeding 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route utilizes readily available raw materials like 2-hydroxy-5-cyanobenzaldehyde and avoids corrosive reagents like polyphosphoric acid, making it highly scalable with simplified equipment requirements and shorter production cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Supplier
The technical advancements detailed in patent CN102936230A represent a significant leap forward in the manufacturing of gout therapeutics, offering a pathway that balances high purity with operational safety. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative laboratory protocols into robust industrial realities. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the versatile reactor systems necessary to handle the specific solvent and temperature requirements of this thiazole-based synthesis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs and advanced analytical capabilities.
We invite pharmaceutical partners to leverage our expertise to secure a stable and cost-effective supply of this critical intermediate. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this non-cyanide route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on the foundation of the most advanced and reliable chemistry available.
