Scalable Synthesis of Febuxostat Intermediates: Technical Upgrades for Commercial Manufacturing
Scalable Synthesis of Febuxostat Intermediates: Technical Upgrades for Commercial Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical active pharmaceutical ingredient (API) precursors, particularly for treatments addressing chronic conditions like hyperuricemia. Patent CN102086169B introduces a transformative preparation method for key febuxostat intermediates, specifically targeting ethyl 2-(3-formyl-4-hydroxyphenyl)-4-methyl-5-thiazolecarboxylate. This technical disclosure represents a significant leap forward in process chemistry, moving away from hazardous gaseous reagents toward safer, aqueous-based catalytic systems. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality. The innovation lies not just in the chemical transformation but in the holistic optimization of safety, yield, and environmental compliance, making it a cornerstone for modern cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazole-based intermediates has been plagued by significant operational hazards and inefficiencies that hinder large-scale industrial adoption. Traditional routes often rely heavily on hydrogen sulfide gas, a highly toxic and flammable substance that necessitates specialized high-pressure equipment and rigorous safety protocols, drastically increasing capital expenditure and operational risk. Furthermore, earlier methodologies frequently utilized polyphosphoric acid as a primary solvent, which tends to polymerize and become excessively viscous during reaction progress, creating severe agitation challenges and heat transfer issues that compromise yield consistency. These legacy processes also suffer from cumbersome post-treatment procedures, requiring extensive purification steps to remove metal salts and organic solvent residues, which inflates production costs and extends lead times. The cumulative effect of these drawbacks is a fragile supply chain vulnerable to regulatory scrutiny and production bottlenecks, making the search for high-purity pharmaceutical intermediates increasingly difficult for downstream manufacturers.
The Novel Approach
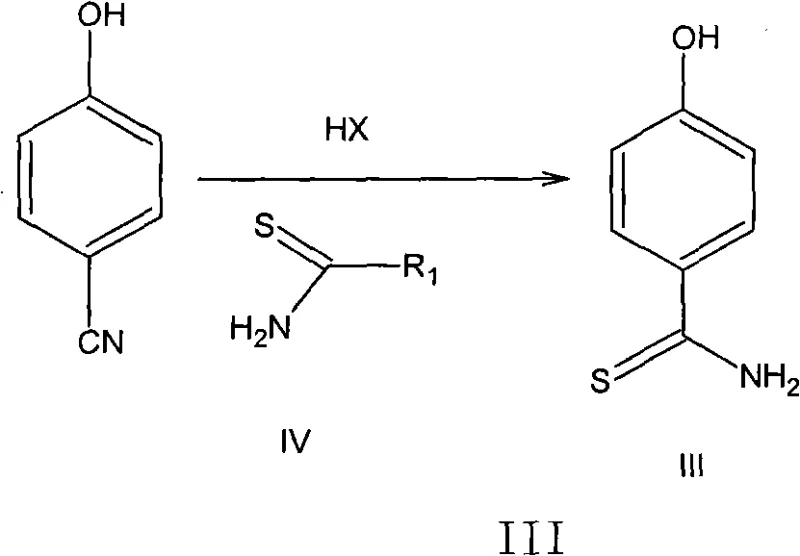
In stark contrast, the novel approach detailed in the patent data utilizes a streamlined, aqueous hydrochloric acid system that fundamentally alters the risk profile and economic viability of the synthesis. By employing thioacetamide and p-cyanophenol in a controlled acidic environment, the process successfully bypasses the need for dangerous hydrogen sulfide gas, thereby eliminating the requirement for high-pressure reactors and complex gas scrubbing systems. This shift to aqueous chemistry not only enhances operator safety but also simplifies waste treatment, as the effluent is easier to manage compared to heavy metal-laden organic streams. The method demonstrates exceptional stability, with reaction conditions that are mild enough to prevent thermal degradation yet robust enough to drive conversions to completion, ensuring that the commercial scale-up of complex pharmaceutical intermediates is both feasible and predictable. This technological pivot offers a clear pathway for reducing lead time for high-purity pharmaceutical intermediates while maintaining stringent quality standards required by global regulatory bodies.
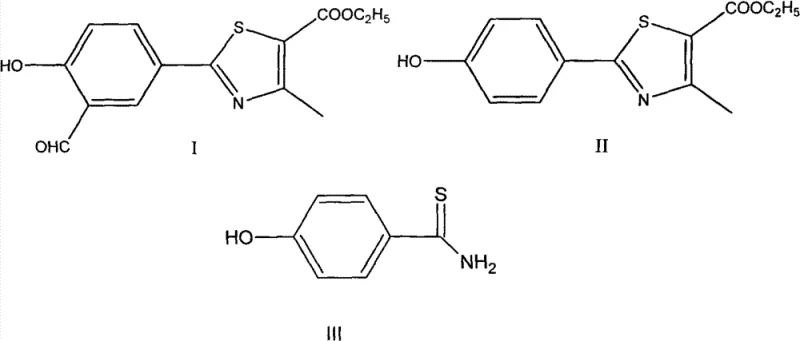
Mechanistic Insights into Aqueous Acid-Catalyzed Thioamide Formation
The core of this synthetic breakthrough lies in the efficient conversion of nitriles to thioamides using thioacetamide as a sulfur source in an aqueous acid medium. Mechanistically, the protonation of the nitrile group in p-cyanophenol by concentrated hydrochloric acid increases its electrophilicity, facilitating nucleophilic attack by the sulfur atom of thioacetamide. This reaction proceeds through a stable transition state that minimizes side reactions such as hydrolysis to the corresponding carboxylic acid, a common impurity in aqueous systems. The presence of water, typically seen as a detriment in organic synthesis, is here leveraged to solubilize the inorganic acid catalyst and facilitate heat dissipation, ensuring uniform reaction kinetics throughout the bulk solution. This precise control over the reaction environment allows for the selective formation of 4-hydroxythiobenzamide with minimal byproduct generation, setting a high-purity foundation for subsequent cyclization steps.
Following the initial thioamide formation, the subsequent cyclization with ethyl 2-chloroacetoacetate is equally critical for defining the impurity profile of the final API intermediate. The reaction mechanism involves the nucleophilic attack of the thioamide sulfur on the alpha-carbon of the chloroacetate, followed by intramolecular condensation to close the thiazole ring. The patent specifies that this step can tolerate a certain degree of moisture and residual acid from the previous step, which is a significant advantage for telescoping operations. By avoiding the isolation and drying of the intermediate thioamide, manufacturers can reduce solvent consumption and processing time, directly contributing to cost reduction in API manufacturing. The robustness of this cyclization ensures that the structural integrity of the thiazole ring is maintained, preventing the formation of open-chain impurities that are difficult to remove in later stages.
How to Synthesize Ethyl 2-(3-formyl-4-hydroxyphenyl)-4-methyl-5-thiazolecarboxylate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and reagent stoichiometry, to maximize the efficiency of each transformation. The process begins with the formation of the thioamide, followed by cyclization to the thiazole ester, and concludes with a formylation step using hexamethylenetetramine in a polyphosphoric acid and methanesulfonic acid mixture. Each stage is designed to be operationally simple, avoiding the need for exotic reagents or extreme conditions that complicate technology transfer. Detailed standard operating procedures regarding mixing rates, addition profiles, and quenching methods are essential for reproducing the high yields reported in the patent literature. The following guide outlines the critical operational milestones necessary for successful execution.
- React p-cyanophenol with thioacetamide in concentrated hydrochloric acid aqueous solution at 30-70°C to form 4-hydroxythiobenzamide.
- Condense the resulting 4-hydroxythiobenzamide with ethyl 2-chloroacetoacetate in ethanol solvent under reflux conditions to yield the thiazole ester.
- Perform formylation using hexamethylenetetramine in a polyphosphoric acid and methanesulfonic acid mixture at 80-110°C to obtain the final aldehyde intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond mere chemical yield. The elimination of hazardous gases and high-pressure equipment significantly lowers the barrier to entry for manufacturing partners, expanding the pool of qualified suppliers and enhancing supply chain resilience. By simplifying the workup procedures and reducing the reliance on expensive organic solvents, the overall cost structure of the intermediate is optimized, allowing for more competitive pricing without sacrificing quality. Furthermore, the environmental benefits of using aqueous systems align with increasingly strict global sustainability mandates, reducing the regulatory burden associated with waste disposal and emissions. This holistic improvement in process design ensures a more reliable pharmaceutical intermediates supplier relationship, characterized by consistent delivery and reduced risk of production stoppages.
- Cost Reduction in Manufacturing: The substitution of hazardous hydrogen sulfide gas with solid thioacetamide removes the need for specialized gas handling infrastructure and safety monitoring systems, leading to substantial capital and operational savings. Additionally, the ability to use crude intermediates directly in subsequent steps without extensive purification reduces solvent consumption and energy usage associated with drying and distillation processes. The high conversion rates minimize raw material waste, ensuring that the input costs are efficiently translated into final product output. These factors collectively drive down the unit cost of production, providing a competitive edge in the marketplace.
- Enhanced Supply Chain Reliability: By utilizing readily available raw materials such as p-cyanophenol and thioacetamide, the process mitigates the risk of supply disruptions caused by the scarcity of specialized reagents. The mild reaction conditions reduce the likelihood of equipment failure or safety incidents that could halt production lines, ensuring a steady flow of materials to downstream API manufacturers. The robustness of the chemistry allows for flexible batch sizing, enabling suppliers to respond quickly to fluctuations in market demand. This reliability is crucial for maintaining continuous drug production schedules and avoiding costly stockouts.
- Scalability and Environmental Compliance: The aqueous nature of the primary reaction steps facilitates easier heat management and mixing on a large scale, overcoming the viscosity issues associated with polyphosphoric acid-only systems. This scalability ensures that the process can be seamlessly transferred from pilot plant to full commercial production without significant re-engineering. Moreover, the reduction in organic solvent usage and the elimination of toxic gas emissions simplify environmental compliance, lowering the cost of waste treatment and permitting. This alignment with green chemistry principles enhances the long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway, derived directly from the patent's background and beneficial effects sections. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers reflect the specific advantages of the aqueous acid method over traditional approaches, focusing on safety, yield, and operational simplicity. These insights are intended to clarify the technical value proposition for potential manufacturing partners.
Q: How does this synthesis method improve safety compared to traditional routes?
A: This method eliminates the use of hazardous hydrogen sulfide gas and high-pressure reactors, utilizing safer aqueous hydrochloric acid systems instead.
Q: What are the yield improvements for the key intermediates?
A: The process achieves yields exceeding 90% for 4-hydroxythiobenzamide, over 85% for the thiazole ester, and above 80% for the final formylated intermediate.
Q: Is the crude product suitable for direct use in subsequent steps?
A: Yes, the post-processing is simplified, and the crude product possesses high purity, allowing it to be fed directly into the next reaction stage without extensive refining.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 2-(3-formyl-4-hydroxyphenyl)-4-methyl-5-thiazolecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis route are fully realized in practice. We maintain stringent purity specifications across all batches, supported by rigorous QC labs equipped with advanced analytical instrumentation to verify identity and assay. Our dedication to technical excellence ensures that every kilogram of material delivered meets the exacting standards required for global API registration.
We invite you to collaborate with us to optimize your supply chain and leverage the cost efficiencies offered by this advanced manufacturing technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us help you secure a stable, high-quality supply of critical intermediates for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
