Advanced Asymmetric Synthesis of Diazepinoindolone Intermediates for Commercial PDE4 Inhibitor Production
Advanced Asymmetric Synthesis of Diazepinoindolone Intermediates for Commercial PDE4 Inhibitor Production
The pharmaceutical industry's relentless pursuit of potent Phosphodiesterase 4 (PDE4) inhibitors has driven significant innovation in the synthesis of complex heterocyclic scaffolds. Patent CN116668C introduces a groundbreaking methodology for the preparation of enantiomerically pure [1,4]diazepino[6,7,1-hi]indol-4-ones, a core structure critical for developing anti-inflammatory and anti-asthmatic therapies. Unlike traditional approaches that rely on the resolution of racemic mixtures—a process inherently limited by a maximum theoretical yield of 50%—this invention leverages a sophisticated weak Lewis acid catalyzed intramolecular cyclization. This strategic shift not only ensures the preservation of stereochemical integrity but also dramatically enhances the economic viability of producing these high-value pharmaceutical intermediates. By directly accessing the desired S-configuration from chiral starting materials, the process eliminates wasteful separation steps, positioning it as a superior choice for reliable pharmaceutical intermediate supplier networks aiming for efficiency.
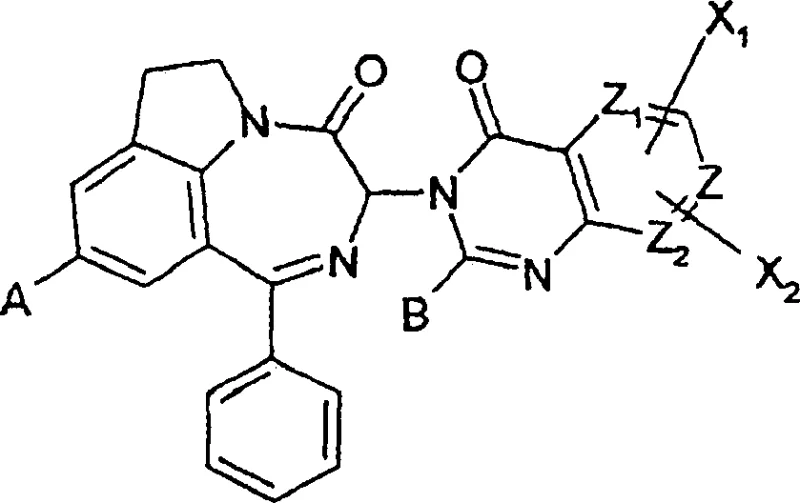
The structural complexity of these molecules, characterized by the fused diazepino-indole core shown in the general formula, demands precise control over reaction conditions to prevent racemization. The patent explicitly defines the substituents A, B, X1, and X2, allowing for a diverse library of analogues to be synthesized using this unified platform. This versatility is paramount for medicinal chemists exploring structure-activity relationships (SAR) in the quest for next-generation bronchodilators. Furthermore, the ability to introduce various functional groups without compromising the chiral center underscores the robustness of this synthetic route, making it an indispensable tool for the cost reduction in API manufacturing where purity and yield are the primary drivers of profitability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically International Patent Application WO 98/49169, disclosed the synthesis of these diazepinoindolone compounds but relied heavily on the preparation of racemic mixtures followed by chiral phase chromatography or salt formation with enantiomerically pure amines for separation. This conventional resolution strategy is fraught with inefficiencies, primarily due to the inherent loss of half the material during the separation of enantiomers, which drastically inflates the cost of goods sold (COGS). Additionally, chromatographic separation on an industrial scale is resource-intensive, requiring large volumes of solvents and specialized stationary phases, which complicates waste management and extends production lead times. The reliance on resolution also introduces variability in optical purity, often necessitating recrystallization cycles that further erode overall yield. For supply chain heads, these factors translate into unpredictable availability and higher inventory costs, creating a bottleneck in the production of high-purity PDE4 inhibitor intermediates needed for clinical and commercial supplies.
The Novel Approach
In stark contrast, the novel approach detailed in CN116668C bypasses the resolution step entirely by utilizing optically active starting materials that undergo a stereospecific cyclization. The key innovation lies in the use of weak Lewis acid catalysts, such as Scandium Trifluoromethanesulfonate (Scandium Triflate), which promote the ring-closing reaction under mild conditions without inducing epimerization at the sensitive chiral center. This method allows for the direct conversion of linear precursors into the cyclic target with high fidelity, preserving the optical purity established in the earlier stages of synthesis. By avoiding the harsh conditions associated with strong Lewis acids like Aluminum Chloride, which are known to cause racemization, this process ensures consistent quality and significantly higher throughput. The elimination of resolution steps simplifies the workflow, reduces solvent consumption, and aligns perfectly with the goals of commercial scale-up of complex heterocycles, offering a streamlined path from laboratory bench to kilogram-scale production.
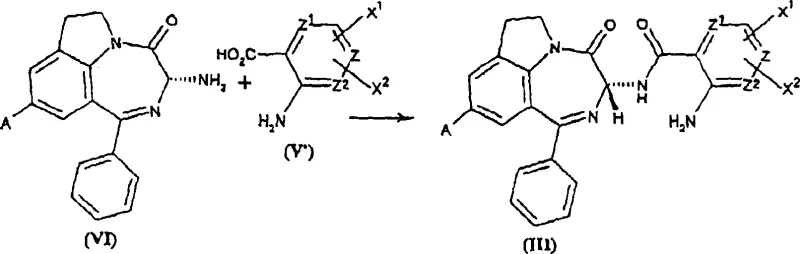
Mechanistic Insights into Weak Lewis Acid-Catalyzed Cyclization
The mechanistic elegance of this synthesis revolves around the careful selection of the catalyst to balance reactivity and stereoselectivity. The reaction involves the intramolecular attack of an aromatic ring onto an imidate or amidine intermediate, a transformation that typically requires acidic activation. However, strong protic or Lewis acids can protonate or coordinate too aggressively with the nitrogen atoms or the chiral center, facilitating the formation of a planar carbocation or enol intermediate that leads to racemization. The use of weak Lewis acids, particularly lanthanide triflates like Scandium Triflate or Ytterbium Triflate, provides just enough activation to facilitate the electrophilic aromatic substitution without disrupting the stereochemical environment. These catalysts function effectively in aprotic solvents such as methylene chloride or toluene at temperatures ranging from 0°C to 40°C, conditions that are mild enough to prevent thermal degradation or epimerization. This precise control is critical for maintaining the enantiomeric excess (ee) above 95%, a specification often required for regulatory approval of chiral drugs.
Furthermore, the process incorporates a protective strategy for the amine functionality prior to cyclization, typically involving the formation of an orthoester or imidate intermediate. This activation step, often performed using trimethyl orthoacetate, converts the amide into a more electrophilic species that is susceptible to cyclization under the influence of the weak Lewis acid. The subsequent removal of the catalyst is straightforward, as many lanthanide triflates are water-soluble and can be washed away during the aqueous workup, or they can be recovered and recycled, adding another layer of sustainability to the process. The robustness of this catalytic system is evidenced by experimental data showing that even after extended reaction times of up to 48 hours, the optical purity remains intact, demonstrating the stability of the chiral center under these specific reaction conditions. This reliability is a key factor for R&D directors evaluating the feasibility of transferring this chemistry to pilot and production plants.
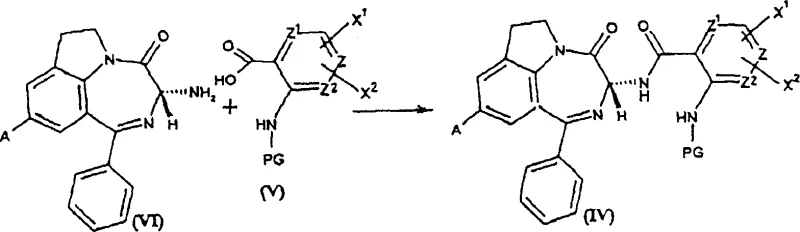
How to Synthesize Diazepinoindolone Efficiently
The synthesis of these valuable intermediates follows a logical sequence designed to maximize yield and purity while minimizing operational complexity. The process begins with the condensation of an optically active amino benzodiazepine with a protected anthranilic acid derivative, utilizing standard peptide coupling reagents such as DCC and HOBt to form the amide bond with high efficiency. Following this, the protecting group is removed under acidic conditions to reveal the free amine, which is then activated via reaction with an orthoester to form the cyclization precursor. The final and most critical step involves the addition of the weak Lewis acid catalyst in an inert solvent, where the mixture is stirred at controlled temperatures to effect ring closure. Detailed standardized synthesis steps see the guide below.
- Condense optically active amino benzodiazepine with protected anthranilic acid using coupling agents like DCC/HOBt to form the amide intermediate.
- Deprotect the amine group under acidic conditions to generate the free amine precursor suitable for cyclization.
- Perform intramolecular cyclization using a weak Lewis acid catalyst such as Scandium Triflate in an aprotic solvent to form the final chiral ring system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers transformative benefits that extend beyond mere technical superiority. The primary advantage lies in the substantial cost savings achieved by eliminating the resolution step, which traditionally consumes significant resources and results in the discard of the unwanted enantiomer. By starting with chiral materials and preserving that chirality throughout the synthesis, the overall material efficiency is nearly doubled compared to racemic routes, directly impacting the bottom line. Additionally, the use of recoverable catalysts and common organic solvents simplifies the supply chain logistics, reducing dependency on exotic reagents that may have long lead times or volatile pricing. This process stability ensures a consistent supply of high-quality intermediates, mitigating the risk of production delays that can plague complex pharmaceutical manufacturing campaigns.
- Cost Reduction in Manufacturing: The elimination of chiral resolution chromatography removes a major cost center, as this technique typically requires expensive columns and large solvent volumes. Furthermore, the high yields reported in the patent examples, often exceeding 80% for key steps, mean that less starting material is required to produce the same amount of final product. The ability to recycle the Lewis acid catalyst further contributes to cost optimization, reducing the consumption of metal salts which can be expensive, particularly for rare earth elements like Scandium. These cumulative efficiencies result in a significantly lower cost per kilogram of the active intermediate, enhancing the competitiveness of the final drug product in the marketplace.
- Enhanced Supply Chain Reliability: The robustness of the weak Lewis acid catalysis ensures that the reaction is less sensitive to minor fluctuations in temperature or moisture compared to sensitive organometallic reactions. This tolerance makes the process more reliable for large-scale manufacturing, where perfect control is harder to maintain than in the laboratory. Moreover, the starting materials, such as substituted anthranilic acids and chiral amines, are commercially available or easily synthesized from commodity chemicals, ensuring a stable upstream supply. This reduces the risk of bottlenecks caused by the scarcity of specialized reagents, allowing for more accurate forecasting and inventory management for reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions, operating between 0°C and 40°C, do not require energy-intensive cryogenic cooling or high-pressure equipment, making the process easier to scale from grams to tons. The use of standard solvents like methylene chloride and ethyl acetate facilitates solvent recovery and recycling, aligning with modern environmental, health, and safety (EHS) standards. Additionally, the avoidance of heavy metal catalysts that are difficult to remove from the final product simplifies the purification process and ensures compliance with strict residual metal limits imposed by regulatory agencies. This environmental friendliness not only reduces waste disposal costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and comparative examples provided in the patent documentation, offering clarity on the practical aspects of the method. Understanding these nuances is essential for technical teams evaluating the transfer of this technology to their own facilities.
Q: Why is Scandium Triflate preferred over strong Lewis acids like Aluminum Chloride?
A: Strong Lewis acids like Aluminum Chloride promote epimerization at the chiral center, leading to racemic mixtures. Scandium Triflate is a weak Lewis acid that facilitates cyclization while maintaining high enantiomeric excess (ee > 95%).
Q: What are the typical yields for this cyclization process?
A: Experimental data indicates high efficiency, with cyclization yields reaching up to 88% and overall optical purity consistently exceeding 94% ee without the need for chiral resolution.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the process utilizes stable catalysts and standard solvents like methylene chloride, avoiding cryogenic conditions. The robustness of the weak Lewis acid system supports scalable commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazepinoindolone Supplier
The technological advancements described in CN116668C represent a significant leap forward in the synthesis of PDE4 inhibitor intermediates, offering a pathway to higher purity and lower costs. NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative methodologies, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of weak Lewis acid catalysis, ensuring that stringent purity specifications are met with every batch. With our rigorous QC labs and commitment to process excellence, we guarantee the delivery of high-quality intermediates that meet the demanding standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this enantioselective process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of reliability, quality, and innovation.
