Advanced Chiral Resolution of Indoline-2-Carboxylic Acid for High-Purity Perindopril Manufacturing
Advanced Chiral Resolution of Indoline-2-Carboxylic Acid for High-Purity Perindopril Manufacturing
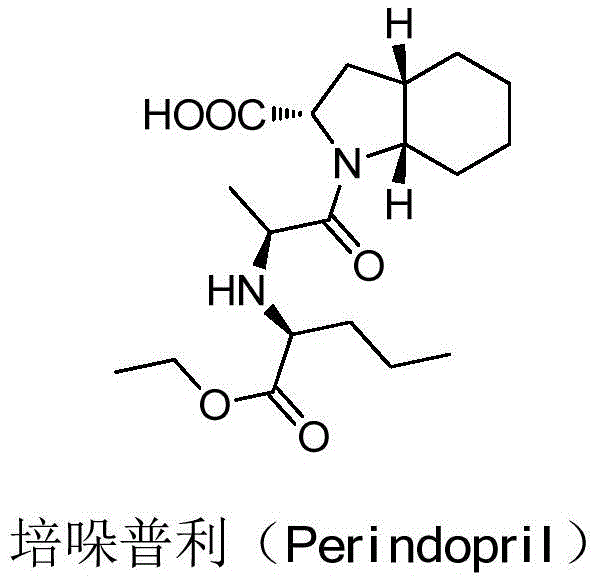
The global demand for high-quality antihypertensive agents continues to drive innovation in the synthesis of key pharmaceutical intermediates. Specifically, the production of Perindopril, a widely prescribed ACE inhibitor, relies heavily on the availability of optically pure S-indoline-2-carboxylic acid. A recent technological breakthrough, documented in patent CN109251163B, introduces a novel resolution method that fundamentally alters the economic and technical landscape of producing this critical chiral building block. This patent discloses a robust process utilizing chiral amino acid esters as resolving agents, offering a distinct departure from conventional amine-based resolution strategies. By leveraging diastereomeric salt formation with tailored amino acid derivatives, the method achieves exceptional separation efficiency, stability, and yield. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the manufacturing of complex chiral intermediates, ensuring a reliable supply of high-purity materials for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
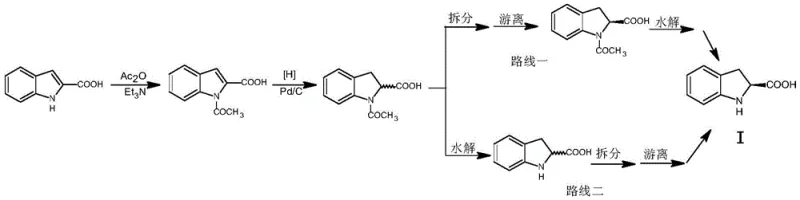
Historically, the industrial preparation of S-indoline-2-carboxylic acid has been plagued by inefficiencies inherent to traditional resolution techniques. As illustrated in the background art, conventional routes often rely on chiral organic bases such as R-alpha-methyl phenethylamine to separate the racemic mixture. However, this approach suffers from significant drawbacks, primarily poor resolution efficiency and the instability of the resulting compounds. The resolution salts formed with R-alpha-methyl phenethylamine frequently fail to achieve adequate separation in a single crystallization step, necessitating multiple, labor-intensive recrystallizations to reach acceptable optical purity. Furthermore, the recovery of the unwanted R-isomer from the mother liquor is problematic; attempts to racemize the recovered material often result in the formation of substantial tar and degradation products, leading to low overall yields and increased waste generation. These factors collectively inflate production costs and complicate the supply chain for this essential pharmaceutical intermediate.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN109251163B introduces a sophisticated resolution strategy centered on the use of chiral amino acid esters. Instead of simple chiral amines, the process employs resolving agents such as phenylglycine esters or phenylalanine esters (Compound II), which interact with the N-acyl indoline-2-carboxylic acid (Compound V) to form diastereomeric salts. This specific molecular recognition leads to a much larger solubility difference between the desired S-configured salt and the unwanted R-configured salt. Consequently, the desired isomer precipitates with high optical purity in a single step, eliminating the need for repetitive recrystallization. Moreover, the compounds involved in this new pathway exhibit superior chemical stability throughout the resolution process. This stability extends to the subsequent recycling steps, where the unwanted isomer can be efficiently racemized and reused, creating a closed-loop system that maximizes atom economy and minimizes raw material consumption.
Mechanistic Insights into Diastereomeric Salt Formation and Recycling
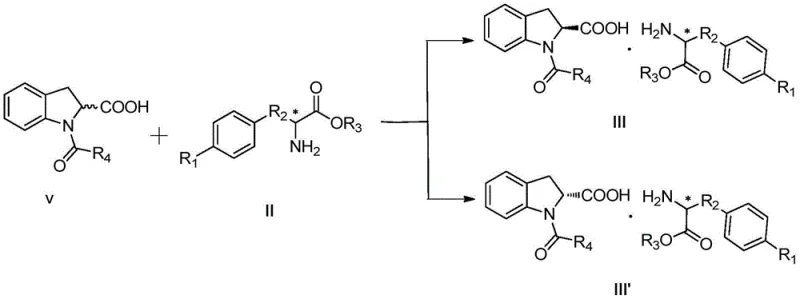
The core of this innovative process lies in the precise stereochemical control exerted during the salt-forming reaction. When the racemic N-acyl indoline-2-carboxylic acid (Compound V) reacts with a single enantiomer of a chiral amino acid ester (Compound II), two distinct diastereomeric salts are generated: Compound III (containing the S-acid) and Compound III' (containing the R-acid). Due to the specific three-dimensional arrangement of the amino acid ester side chains, these salts possess markedly different physical properties, particularly solubility in alcohol or aqueous solvents. Under optimized conditions—typically heating to reflux between 50°C and 100°C followed by controlled cooling—the less soluble salt (usually the S-configured complex) crystallizes out of the solution with high selectivity. This mechanistic advantage allows for the direct isolation of the target intermediate with optical purity often exceeding 98% ee, as demonstrated in the patent examples using agents like L-phenylglycine ethyl ester.
Beyond the initial separation, the true engineering brilliance of this method is found in its integrated recycling mechanism. The mother liquor, enriched with the unwanted R-isomer, is not discarded but instead processed to recover the chiral acid. Following liberation from the resolving agent using mineral acids like hydrochloric acid, the recovered R-indoline derivative undergoes a hydrolysis step to remove the N-acyl protecting group. Crucially, the resulting free acid is then subjected to a racemization reaction using acid anhydrides (e.g., acetic anhydride) at elevated temperatures (100°C to 120°C). This step effectively converts the unwanted enantiomer back into a racemic mixture, which can be re-acylated and fed back into the resolution cycle. This continuous loop ensures that nearly 100% of the starting material is eventually converted into the desired S-product, drastically reducing waste and enhancing the overall sustainability of the manufacturing process.
How to Synthesize S-Indoline-2-Carboxylic Acid Efficiently
The implementation of this resolution technology requires careful control of reaction parameters to maximize yield and optical purity. The process begins with the selection of an appropriate N-acyl protecting group (such as acetyl, propionyl, or benzoyl) and a matching chiral amino acid ester resolving agent. The salt formation is typically conducted in green solvents like water, ethanol, or methanol, aligning with modern environmental standards. Following the crystallization and filtration of the diastereomeric salt, the protocol involves an acid-mediated freeing reaction followed by hydrolysis to yield the final free acid. For a detailed, step-by-step breakdown of the experimental conditions, stoichiometry, and workup procedures described in the patent examples, please refer to the standardized synthesis guide below.
- Perform a salt-forming reaction between racemic N-acyl indoline-2-carboxylic acid (Compound V) and a chiral amino acid ester resolving agent (Compound II) in an alcohol or water solvent.
- Filter the precipitated diastereomeric salt (Compound III) which contains the desired S-configuration, separating it from the mother liquor containing the unwanted isomer.
- Treat the salt with acid to liberate the free base, followed by acid hydrolysis to remove the acyl protecting group, yielding pure S-indoline-2-carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel resolution method translates into tangible strategic benefits that extend far beyond simple chemical yield. By replacing inefficient, multi-step recrystallization processes with a high-selectivity single-step resolution, manufacturers can significantly streamline production timelines. The ability to recycle the unwanted isomer through a robust racemization pathway means that raw material costs are drastically optimized, as the effective yield approaches theoretical maximums. Furthermore, the use of commercially available amino acid esters as resolving agents mitigates supply risk, as these materials are produced on a massive scale for the food and pharma industries, ensuring consistent availability and price stability compared to specialized chiral amines.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the elimination of yield losses associated with traditional mother liquor disposal. In conventional processes, up to half of the starting material (the unwanted enantiomer) is often lost or degraded during recovery. By implementing the patented racemization cycle, this material is continuously reintroduced into the process, effectively doubling the output per unit of raw material input without proportional increases in cost. Additionally, the high purity achieved in the first crystallization reduces the burden on downstream purification steps, lowering energy consumption and solvent usage. This holistic efficiency gain results in a substantially lower cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is critical for long-term API contracts, and this method enhances reliability by simplifying the dependency on exotic reagents. Traditional resolving agents like R-alpha-methyl phenethylamine can sometimes face supply bottlenecks or quality variability. In contrast, the amino acid esters utilized in this new method (e.g., phenylglycine esters) are commodity chemicals with robust global supply chains. Moreover, the process stability—characterized by the absence of tar formation and degradation during recycling—ensures consistent batch-to-batch quality. This predictability allows supply chain planners to forecast production capacity with greater accuracy, reducing the risk of stockouts for critical hypertension medications.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is exceptionally well-suited for large-scale commercial production. The reactions utilize standard unit operations such as reflux, cooling crystallization, and filtration, which are easily transferable from pilot plants to multi-ton reactors. The preference for alcohol and water solvents over hazardous organic solvents simplifies waste treatment and aligns with increasingly stringent environmental regulations. The high atom economy achieved through the recycling loop also minimizes the volume of chemical waste requiring disposal, supporting corporate sustainability goals and reducing the environmental footprint of pharmaceutical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and claims within patent CN109251163B, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What resolving agents are preferred for this indoline derivative resolution?
A: Unlike traditional methods using R-alpha-methyl phenethylamine, this patent utilizes chiral amino acid esters such as L-phenylglycine ethyl ester, L-phenylalanine methyl ester, or their derivatives. These agents provide superior resolution efficiency and higher optical purity in a single crystallization step.
Q: Can the unwanted R-isomer be recycled in this process?
A: Yes, a key advantage of this method is the ability to recycle the mother liquor. The unwanted R-isomer recovered from the mother liquor can be subjected to a racemization reaction using acid anhydrides, converting it back into a racemic mixture for re-entry into the resolution cycle, thereby maximizing material utilization.
Q: What represents the critical quality attribute for the final S-indoline-2-carboxylic acid?
A: The critical quality attribute is the optical purity (ee value). The described method consistently achieves high optical purity (often >98% ee) and low isomer content (<0.5%), which is essential for the subsequent synthesis of the ACE inhibitor Perindopril to meet regulatory standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoline-2-Carboxylic Acid Supplier
As the pharmaceutical industry demands ever-higher standards of purity and efficiency, NINGBO INNO PHARMCHEM stands at the forefront of delivering advanced chiral intermediates. We have closely analyzed the technological shifts represented by patents like CN109251163B and have integrated similar high-efficiency resolution strategies into our own manufacturing platforms. Our facility possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric needs of global generic and innovator drug companies. With our stringent purity specifications and rigorous QC labs, we guarantee that every batch of S-indoline-2-carboxylic acid meets the exacting standards required for Perindopril synthesis, minimizing impurity risks in your final drug product.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain. Our technical team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to our optimized intermediate can reduce your overall API production costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us partner with you to secure a sustainable, high-quality supply of this critical cardiovascular intermediate.
