Advanced Atmospheric Synthesis of (2S)-Indoline-2-Carboxylic Acid for High-Volume Pharmaceutical Production
The pharmaceutical industry's demand for high-quality angiotensin-converting enzyme (ACE) inhibitors continues to drive innovation in intermediate synthesis, particularly for blockbuster drugs like Perindopril. Patent CN101774960B introduces a transformative preparation method for (2S)-indoline-2-carboxylic acid, a critical chiral building block in this therapeutic class. This technology addresses long-standing manufacturing bottlenecks by replacing hazardous high-pressure conditions with a safe, atmospheric pressure reflux system. By leveraging a sophisticated racemization and resolution cycle, the process ensures that the absolute quantity of the desired (2S)-isomer is dominant while efficiently recycling the unwanted (2R)-enantiomer. For global supply chain leaders, this represents a pivotal shift towards more robust and scalable production capabilities that do not compromise on the stringent purity specifications required for modern cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of racemic indoline-2-carboxylic acid and its subsequent resolution relied heavily on aggressive reaction conditions that posed significant operational challenges. Prior art, such as the methods disclosed in China Patent No. CN100337996C, necessitated the use of aqueous NaOH solutions at temperatures ranging from 140°C to 200°C. Crucially, these reactions had to be conducted under pressures of 5 to 15 bar, mandating the use of specialized high-pressure autoclaves. This reliance on high-pressure equipment not only inflated capital expenditure (CAPEX) due to the need for reinforced reactors but also introduced substantial safety risks associated with high-temperature, high-pressure operations. Furthermore, the harsh thermal conditions often complicated the control of impurity profiles, requiring extensive downstream purification to meet the rigorous standards of pharmaceutical grade intermediates.
The Novel Approach
The methodology outlined in CN101774960B fundamentally reengineers this workflow by enabling racemization to proceed under atmospheric pressure reflux in an n-butyl alcohol solution of NaOH. This elimination of high-pressure requirements allows manufacturers to utilize standard glass-lined or stainless steel reactors, drastically lowering the barrier to entry for production and enhancing operational safety. The process involves tracking the reaction via liquid chromatography to ensure complete conversion to the racemic mixture before proceeding to the resolution step. By shifting to a milder thermal profile and ambient pressure, the novel approach not only mitigates safety hazards but also simplifies the engineering controls needed for commercial scale-up, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for consistent, large-volume output.
Mechanistic Insights into Base-Catalyzed Racemization and Chiral Resolution
The core chemical innovation lies in the efficient base-catalyzed racemization of the indoline ring system. In the presence of NaOH within the n-butyl alcohol solvent, the acidic proton at the C2 position of the indoline ring is abstracted, forming a planar carbanion or enolate intermediate. This planar geometry allows for reprotonation from either face with equal probability, effectively converting the enriched mixture of (2S) and (2R) isomers into a true racemate (Formula II). This step is critical because it allows the manufacturer to take the 'waste' stream—the unwanted (2R)-isomer from previous resolution batches—and reset it to a 50:50 mixture, thereby feeding it back into the resolution cycle. The use of n-butyl alcohol is particularly strategic, as its boiling point facilitates effective reflux without degrading the sensitive indoline scaffold, ensuring that the structural integrity of the molecule is maintained throughout the vigorous stirring and heating phases.
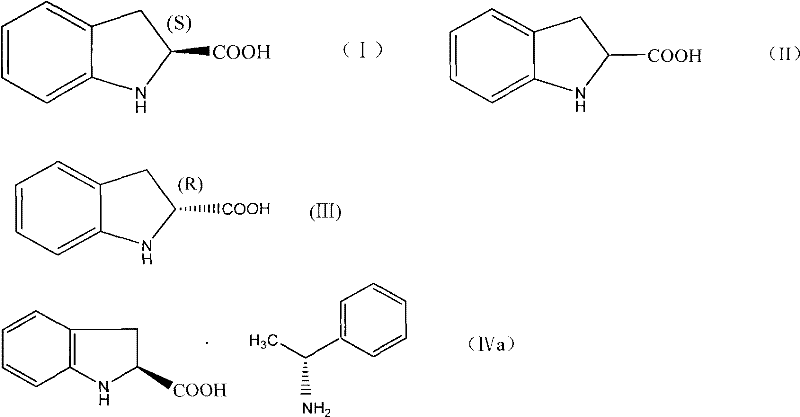
Following racemization, the process employs a classical diastereomeric salt formation for chiral separation. The racemic acid reacts with (R)-alpha-methylbenzylamine in ethanol, where the solubility differences between the resulting diastereomeric salts drive the crystallization. Specifically, the salt formed between the (2S)-acid and the (R)-amine (Formula IVa) exhibits lower solubility in the ethanol medium compared to its (2R)-counterpart, allowing it to precipitate as Filter Cake A. This solid is then isolated and acidified with hydrochloric acid to liberate the free (2S)-indoline-2-carboxylic acid with exceptional optical purity. The mother liquor, rich in the unwanted (2R)-isomer salt (Formula IVb), is not discarded but instead processed to recover the acid for re-racemization, creating a closed-loop system that maximizes atom economy and minimizes chemical waste.
How to Synthesize (2S)-Indoline-2-Carboxylic Acid Efficiently
The synthesis protocol described in the patent offers a streamlined pathway that balances high yield with operational simplicity. The procedure begins with the racemization of the starting material mixture in n-butyl alcohol, followed by solvent removal and acidification to isolate the racemic acid. This intermediate is then subjected to resolution with (R)-alpha-methylbenzylamine in ethanol. The key to maximizing efficiency lies in the iterative recycling of the mother liquor; by repeating the racemization and resolution steps 2 to 6 times on the recovered material, manufacturers can cumulatively harvest the desired (2S)-isomer from what would otherwise be waste streams. Detailed standardized operating procedures regarding specific stoichiometric ratios, temperature controls, and filtration parameters are essential for replicating the reported 98% chemical purity and >99% ee values.
- Perform racemization of the (2S)/(2R) mixture in n-butyl alcohol with NaOH under atmospheric reflux to generate racemic indoline-2-carboxylic acid.
- Conduct chiral resolution by reacting the racemic acid with (R)-alpha-methylbenzylamine in ethanol to precipitate the desired (2S)-isomer salt.
- Acidify the filter cake with hydrochloric acid to isolate the final product, and recycle the mother liquor to recover remaining isomers for reprocessing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from high-pressure autoclave methods to atmospheric reflux systems offers profound economic and logistical benefits. The most immediate impact is the drastic reduction in equipment dependency; facilities no longer need to allocate expensive high-pressure vessels for this specific step, freeing up critical reactor capacity for other processes. This flexibility enhances the overall agility of the manufacturing plant, allowing for better response times to market fluctuations. Additionally, the ability to recycle n-butyl alcohol significantly lowers the recurring cost of raw materials and reduces the volume of hazardous waste requiring disposal, aligning production with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of high-pressure autoclaves translates directly into lower capital investment and reduced maintenance costs. Standard atmospheric reactors are cheaper to purchase, easier to maintain, and have longer operational lifespans compared to high-pressure vessels. Furthermore, the recycling of the n-butyl alcohol solvent means that the consumption of fresh solvent per kilogram of product is significantly minimized, leading to substantial variable cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Operating at atmospheric pressure inherently reduces the risk of unplanned shutdowns due to equipment failure or safety interlocks triggered by pressure deviations. This stability ensures a more predictable production schedule, which is vital for maintaining the continuity of supply for downstream API manufacturers. The robustness of the chemistry, demonstrated by consistent purity levels across multiple recycling loops, guarantees that quality issues will not become a bottleneck in the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the engineering complexities associated with scaling high-pressure reactions. Heat transfer and mixing are more manageable in atmospheric vessels, facilitating a smoother transition from pilot plant to commercial tonnage. Moreover, the closed-loop recycling of solvents and the recovery of the (2R)-isomer reduce the environmental footprint, making the process compliant with green chemistry principles and reducing the regulatory burden associated with waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the process capabilities and limitations for potential partners.
Q: What is the primary advantage of this synthesis method over prior art?
A: Unlike previous methods requiring high-pressure autoclaves at 140-200°C, this process operates at atmospheric pressure, significantly reducing equipment costs and safety risks while maintaining high optical purity (>99% ee).
Q: How is the overall yield improved in this process?
A: The process incorporates a recycling loop where the mother liquor containing the unwanted (2R)-isomer is re-racemized and re-resolved. This iterative recovery increases the cumulative yield from an initial ~35% to over 60%.
Q: Is the solvent system environmentally sustainable?
A: Yes, the method utilizes n-butyl alcohol which can be distilled and recycled after the racemization step, minimizing solvent waste and reducing the environmental footprint of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S)-Indoline-2-Carboxylic Acid Supplier
As the global demand for cardiovascular therapeutics grows, securing a stable source of high-purity intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to leverage this advanced atmospheric pressure technology to deliver (2S)-indoline-2-carboxylic acid at commercial scale. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on our stringent purity specifications. With our rigorous QC labs and commitment to process safety, we provide a supply chain partnership that is both resilient and responsive to the dynamic needs of the pharmaceutical market.
We invite you to engage with our technical team to explore how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential efficiencies of switching to this atmospheric method. We encourage all prospective partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your production timelines.
