Advanced Manufacturing of Mitiglinide Calcium: A Cost-Effective Chiral Route for Global Supply Chains
The pharmaceutical landscape for type II diabetes treatment continues to evolve, driven by the demand for agents that offer rapid onset and minimal hypoglycemic risk, such as Mitiglinide Calcium (S 21403). A pivotal advancement in the manufacturing of this ATP-dependent potassium channel blocker is detailed in patent CN102101838A, which outlines a sophisticated preparation method that fundamentally alters the economic and technical feasibility of production. Unlike conventional pathways that struggle with inefficient raw material utilization, this disclosed methodology leverages a strategic early-stage chiral resolution to optimize the entire synthetic sequence. For global procurement leaders and R&D directors, understanding this shift is critical, as it represents a move towards more sustainable and cost-effective pharmaceutical intermediate sourcing. The patent describes a six-step sequence beginning with a Stobbe condensation, proceeding through catalytic hydrogenation and chiral resolution, and culminating in the formation of the final calcium salt, thereby establishing a robust framework for high-purity API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Mitiglinide Calcium has been plagued by significant inefficiencies related to the handling of chiral centers and expensive raw materials. Prior art, such as the method described in patent application 200510200127.9, typically involves the synthesis of a racemic mixture of the target molecule or its immediate precursors before attempting separation. This approach necessitates the coupling of cis-hexahydroisoindole with a racemic acid, meaning that 50% of this valuable and costly amine raw material is essentially wasted on the unwanted enantiomer which must subsequently be discarded or recycled with difficulty. Furthermore, separating enantiomers at the final amide stage is often technically challenging, requiring complex chromatography or multiple recrystallization steps that drastically reduce overall yield and increase processing time. These bottlenecks create substantial supply chain vulnerabilities, leading to higher costs and inconsistent availability of the high-purity pharmaceutical intermediate required for final drug formulation.
The Novel Approach
The innovative strategy presented in CN102101838A circumvents these traditional pitfalls by shifting the chiral resolution to an earlier stage in the synthetic pathway, specifically at the dicarboxylic acid level. By resolving DL-2-benzyl succinic acid using an inexpensive chiral amine like (R)-1-phenylethylamine prior to the coupling reaction, the process ensures that only the desired (S)-enantiomer proceeds to the expensive amide formation step. This strategic pivot not only conserves the precious cis-hexahydroisoindole raw material but also simplifies the purification profile of the final product. The result is a streamlined process that enhances the overall atom economy and reduces the burden on downstream purification units. For manufacturers aiming for cost reduction in API manufacturing, this approach offers a compelling alternative that aligns technical excellence with commercial viability, ensuring a more reliable supply of this critical antidiabetic agent.
Mechanistic Insights into Stobbe Condensation and Chiral Resolution
The foundation of this synthesis lies in the classic Stobbe condensation, a powerful carbon-carbon bond-forming reaction that constructs the core skeleton of the molecule. In this specific application, diethyl succinate reacts with benzaldehyde in the presence of sodium ethoxide, generated in situ from sodium metal and absolute ethanol. The mechanism involves the formation of an enolate from the succinate ester, which attacks the carbonyl carbon of the benzaldehyde, followed by intramolecular cyclization and ring opening to yield the mono-ester of benzylidene succinic acid. Subsequent hydrolysis under alkaline conditions converts the remaining ester group into a carboxylic acid, yielding benzylidene succinic acid. This step is crucial as it establishes the conjugated system that will later be reduced, and the conditions must be tightly controlled to prevent polymerization or side reactions that could compromise the purity of the intermediate.
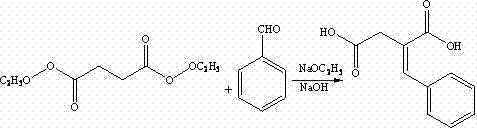
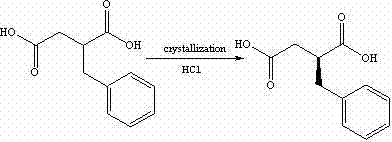
Following the construction of the carbon backbone, the process employs catalytic hydrogenation to saturate the double bond, converting benzylidene succinic acid into DL-2-benzyl succinic acid. The true brilliance of the process, however, is revealed in the subsequent resolution step. The racemic acid is treated with a chiral base, specifically (R)-1-phenylethylamine or its derivatives, in a solvent like methanol. This reaction forms diastereomeric salts between the chiral amine and the two enantiomers of the acid. Due to the differences in physical properties, particularly solubility, one diastereomeric salt precipitates preferentially while the other remains in solution. This allows for the isolation of the desired (S)-enantiomer with high optical purity through simple filtration and acidification. This mechanistic elegance ensures that the chirality is locked in before the introduction of the complex isoindole moiety.
How to Synthesize Mitiglinide Calcium Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and optical purity. The process begins with the rigorous exclusion of moisture during the formation of sodium ethoxide, followed by careful temperature management during the Stobbe condensation to ensure complete conversion. Once the resolved (S)-2-benzyl succinic acid is obtained, it is converted into its corresponding anhydride using acetic anhydride, a reactive intermediate that facilitates the subsequent amide bond formation. The coupling with cis-hexahydroisoindole is performed under mild conditions to preserve the stereochemical integrity of the chiral center. Finally, the free acid is converted to the calcium salt using calcium chloride and ammonia water, a step that requires controlled crystallization to obtain the stable dihydrate form of the drug substance. The detailed standardized synthesis steps are outlined in the guide below.
- Perform Stobbe condensation on diethyl succinate and benzaldehyde using sodium ethoxide, followed by hydrolysis to obtain benzylidene succinic acid.
- Execute catalytic hydrogenation to reduce the double bond, yielding DL-2-benzyl succinic acid, which is then resolved using (R)-chiral amine.
- Convert the resolved (S)-acid to its anhydride, couple with cis-hexahydroisoindole, and finally form the calcium salt to obtain Mitiglinide Calcium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis route described in CN102101838A translates directly into tangible operational benefits and risk mitigation. The primary advantage lies in the drastic reduction of raw material costs associated with the chiral amine component. By avoiding the waste of cis-hexahydroisoindole on the wrong enantiomer, manufacturers can significantly lower the bill of materials for every kilogram of API produced. This efficiency gain is compounded by the use of commodity chemicals like diethyl succinate and benzaldehyde, which are readily available in the global market, thereby reducing the risk of supply disruptions caused by reliance on exotic or proprietary starting materials. Furthermore, the simplified purification workflow reduces solvent consumption and waste generation, aligning with modern environmental compliance standards and lowering disposal costs.
- Cost Reduction in Manufacturing: The most significant financial impact of this novel route is the conservation of cis-hexahydroisoindole, a relatively expensive and structurally complex building block. In traditional racemic synthesis, half of this material is effectively discarded, representing a direct loss of capital. By resolving the acid precursor first, the new method ensures that nearly 100% of the coupled amine contributes to the final product, leading to substantial cost savings per batch. Additionally, the high yield of the hydrogenation and resolution steps minimizes the need for reprocessing, further driving down the cost of goods sold (COGS) and enhancing the overall profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on robust, well-understood chemical transformations that are less prone to failure than complex enzymatic or asymmetric catalytic processes. The reagents used, such as sodium metal, palladium on carbon, and common chiral amines, are sourced from a broad base of global suppliers, preventing single-source bottlenecks. The ability to produce the key chiral intermediate in-house with high consistency means that manufacturers are less dependent on external vendors for critical chiral building blocks, granting them greater autonomy and control over their production schedules and inventory levels.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations like reflux, filtration, and crystallization that are easily transferred from pilot plants to multi-ton commercial reactors. The absence of heavy metal catalysts in the resolution step (using organic amines instead) and the efficient use of solvents contribute to a greener manufacturing profile. This ease of scale-up ensures that commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet market demand, while the reduced waste stream simplifies regulatory compliance and environmental permitting, accelerating time-to-market for generic versions of the drug.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Mitiglinide Calcium using this advanced methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and quality assurance measures. Understanding these details is essential for partners evaluating the feasibility of integrating this supply source into their global procurement strategies.
Q: How does this new synthesis route improve cost efficiency compared to traditional methods?
A: Traditional methods synthesize the racemic amide first, requiring the use of expensive cis-hexahydroisoindole for both enantiomers before discarding half. This novel route resolves the chiral center at the acid stage, significantly reducing the consumption of the costly amine raw material.
Q: What represents the critical control point for ensuring high optical purity in this process?
A: The critical control point is the resolution step using (R)-chiral amines such as (R)-1-phenylethylamine. By forming diastereomeric salts at the benzyl succinic acid stage, the process achieves a high degree of chiral separation before the final coupling reaction.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process utilizes standard industrial reagents like diethyl succinate and benzaldehyde, and employs robust unit operations such as reflux, hydrogenation, and crystallization, making it highly scalable for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mitiglinide Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-quality synthesis routes in the competitive landscape of antidiabetic medications. Our technical team has thoroughly analyzed the pathway described in CN102101838A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are committed to delivering Mitiglinide Calcium with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for pharmaceutical applications. Our facility is equipped to handle the specific unit operations involved, from low-temperature crystallizations to precision hydrogenations, ensuring a seamless transition from development to full-scale manufacturing.
We invite global partners to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your Mitiglinide Calcium supply.
